Abstract
A 16-month-old Wagyu heifer calf presented for depression, inappetence, and polyuria/polydipsia. Physical examination revealed that the heifer calf was mentally dull, subjectively small for her age, bradycardic, and hypothermic and had bilateral nasal discharge. Laboratory tests revealed marked serum and cerebrospinal fluid hypernatremia and hyperchloremia with increased cerebrospinal fluid protein. The heifer calf was treated with Ringer solution intravenously for dehydration and electrolyte abnormalities, and with 1 dose each of thiamine and penicillin. Clinical deterioration prompted the owner to elect humane euthanasia. Necropsy revealed a mass lesion in the suprasellar region. Histopathology was consistent with a suprasellar germ cell tumor; the mass stained positive on immunohistochemistry for cytokeratin, vimentin, and c-kit. Suprasellar germ cell tumors have previously been reported in human beings and dogs.
Primary intracranial neoplasms are rare in the bovine species and are typically diagnosed on postmortem examination due to the physical and financial constraints of antemortem diagnosis. Suprasellar germ cell tumor (GCT) has been reported rarely in human beings1,8,14,16,21 and dogs.13,20 In general, masses arising from the suprasellar region cause clinical signs consistent with pituitary dysfunction 14 ; blindness, exophthalmos, obtundation, mydriasis, ptosis, and polyuria have also been reported.13,20 The following report describes the case of a suprasellar GCT in a bovid as well as the histological and immunohistochemical findings of this rare neoplasm.
A 16-month-old Wagyu heifer calf was presented to The Ohio State University Department of Veterinary Clinical Sciences Food and Fiber Animal Medicine and Surgery Section (Columbus, Ohio) for a 3-week history of a head tilt, generalized poor well-being, and a 1-week history of inappetence. Three weeks before presentation, the owner noticed a head tilt and an infection associated with a fly tag; direction of the head tilt and the affected ear were unknown to the owner. Removal of the fly tag did not improve the head tilt. The heifer calf was given a magnet 7 days prior to arrival, and subsequently the calf became inappetent. She had been eating well previously, although not gaining weight. She was polyuric and polydipsic. She had bilateral nasal discharge of 2 days duration. Previous treatment with oxytetracycline, flunixin meglumine, vitamin B12, sulfa-antibiotics, and dexamethasone did not improve her condition.
On presentation, the heifer calf was estimated to weigh 317.5 kg. The calf was depressed, but responsive, hypothermic (37°C), and tachycardic (68 bpm), with a normal respiratory rate (16/min). Heart and lungs auscultated normally. A mucopurulent nasal discharge was present bilaterally, which was worse on the right side. Ruminations were absent. Oral mucous membranes were light pink with a prolonged capillary refill time of 2.5 sec.
Initial work-up included a complete blood cell count (CBC), biochemical profile, dipstick urinalysis, fecal flotation, and abdominal ultrasound. The CBC showed mild polycythemia (40%, range: 23–35%). a Biochemical profile revealed hyperphosphatemia (9.1 mg/dl, range: 3.8–7.7 mg/dl), hypernatremia (158 mEq/l, range: 133–143mEq/l), hyperchloremia (115.2 mEq/l, range: 98–108 mEq/l), increased aspartate aminotransferase (384 IU/l, range: 50–120 IU/l), and elevated creatine kinase (1350 IU/l, range: 90–310 IU/l). Results of a urine dipstick were within normal limits, and urine specific gravity was isosthenuric (1.012). Fecal exam showed occasional Eimeria spp. An abdominal ultrasound revealed hypomotile intestines with no other significant findings.
Supportive treatment was started for dehydration and electrolyte abnormalities; Ringer solution and dextrose were administered intravenously. Shortly after hospitalization, the heifer calf became weak and obtunded. She was unable to stand and remained recumbent. She ate only small amounts of hay and did not drink any water. Twelve hours later, her mental status remained unchanged. A blood gas was performed, and the calf was noted to be markedly hypernatremic (180.2 mEq/l, range: 133–143 mEq/l). An electrolyte panel confirmed hypernatremia (181 mEq/l), hyperchloremia (140.6 mEq/l, range: 98–108 mEq/l), and hyperphosphatemia (9.0 mg/dl, range: 3.8–7.7 mg/dl). A lumbosacral cerebrospinal fluid (CSF) collection was performed due to her mental status. The CSF was colorless, the cell count was within normal limits, the protein level was 270 mg/dl, and analysis showed mild blood contamination. An electrolyte panel was performed on the CSF to compare values to peripheral blood. The CSF was hypernatremic (184 mEq/l) and hyperchloremic (153.4 mEq/l), similar to serum values. The heifer calf was given 1 treatment each of thiamine and penicillin to address differential diagnoses of polioencephalomalacia and listeriosis, respectively. Her mental status continued to deteriorate and she became increasingly hypothermic and bradycardic. The owner elected euthanasia due to worsening of clinical signs and a poor prognosis. Necropsy was requested to determine the cause of her clinical signs.
Following humane euthanasia, the heifer calf had a body condition score of 3 out of 5 and was in good postmortem condition. The liver had multifocal fibrous white tags on the surface that extended into the parenchyma as firm white nodules. A spherical, extra-axial mass was ventral to the brain at the level of the mesencephalon and hypothalamus and dorsal to the sella turcica, with compression of surrounding parenchyma. The mass was approximately 3 cm in diameter, yellow–tan in color, with a soft yellow center (Fig. 1).
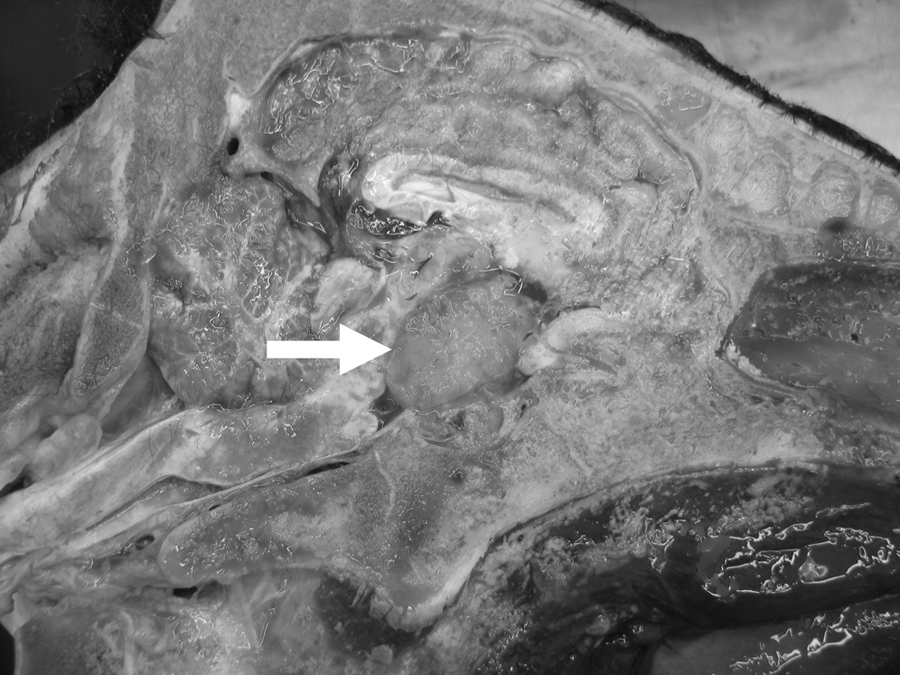
Suprasellar mass (arrow) in a 16-month-old Wagyu heifer calf.
A section of the brain mass was submitted for aerobic bacterial culture. There was no significant bacterial growth. Histologically, the multifocal lesions in the liver were determined to be focal organizing granulomas, an incidental finding.
The brain mass was composed of solid sheets of poorly differentiated cells that ranged from large polygonal cells with abundant cytoplasm that was often vacuolated, to small round cells with small amounts of amphophilic cytoplasm. There were also sporadic areas with epithelial differentiation (cuboidal cells devoid of cilia or microvilli) and tubule formation (Fig. 2). Cells were supported by a delicate fibrovascular stroma, but there was no appearance of neuroendocrine packeting. Mitotic figures were rare.
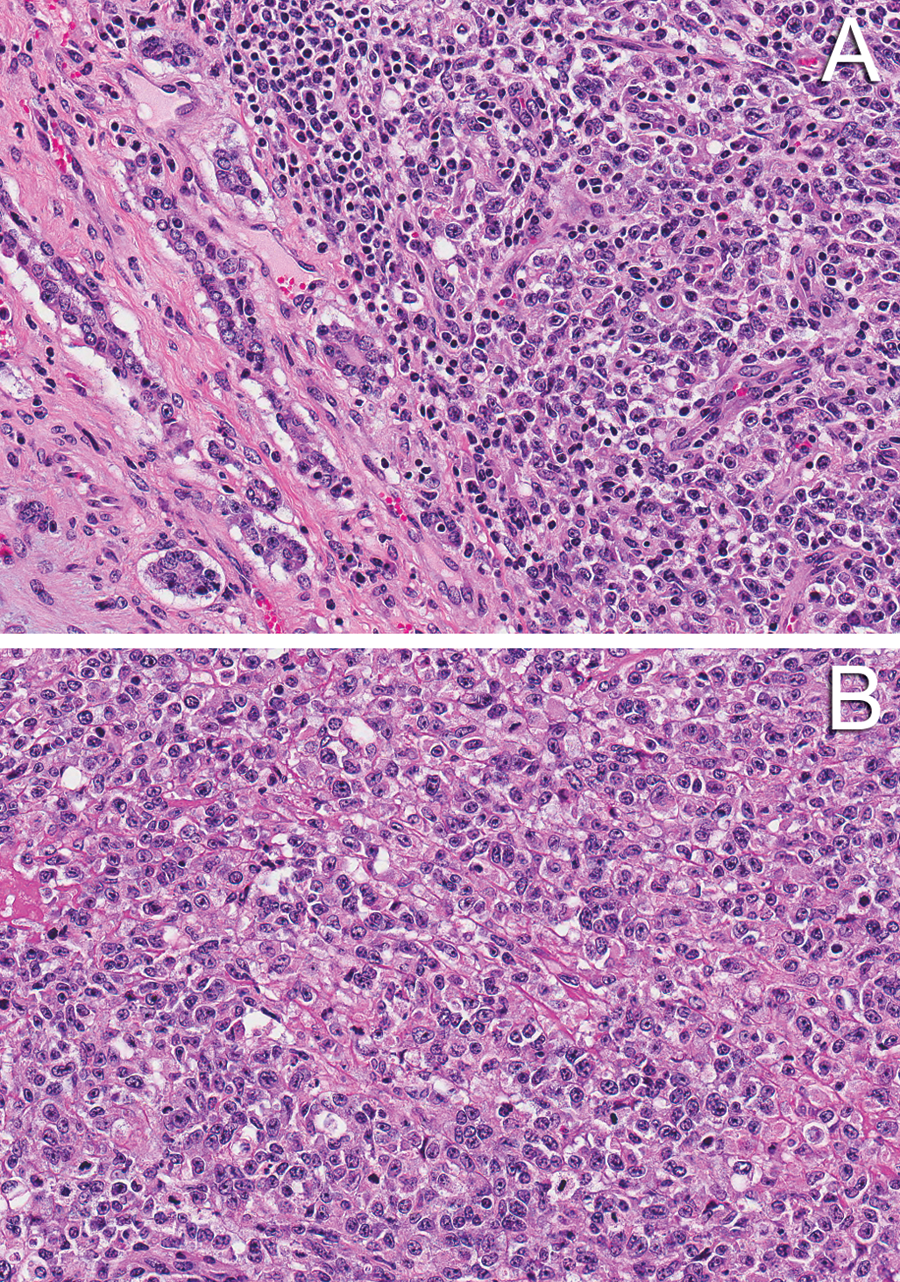
Suprasellar germ cell tumor, mixed type; 16-month-old Wagyu heifer calf.
Immunohistochemistry was performed to further classify the tumor. a Tissues did not stain with antibody against alpha-fetoprotein (AFP; rabbit polyclonal antibody, 1:4,000 dilution), b-human chorionic gonadotropin (b-HCG; rabbit polyclonal antibody, 1:800 dilution), or placental alkaline phosphatase (PLAP; rabbit polyclonal antibody, 1:25 dilution). Cytoplasmic c-kit immunoreactivity (rabbit polyclonal antibody, 1:500 dilution) was observed in portions of the neoplasm, particularly in the small cell type (Fig. 3A). The epithelial component exhibited strong cytokeratin immunoreactivity (rabbit polyclonal antibody, 1:50 dilution; Fig. 3B). Vimentin (rabbit polyclonal antibody, 1:50 dilution) staining in round cells, as well as in the stromal components of the neoplasm, was also observed (Fig. 3C). Tumor morphology and immunoreactivity profile were consistent with a diagnosis of suprasellar GCT.
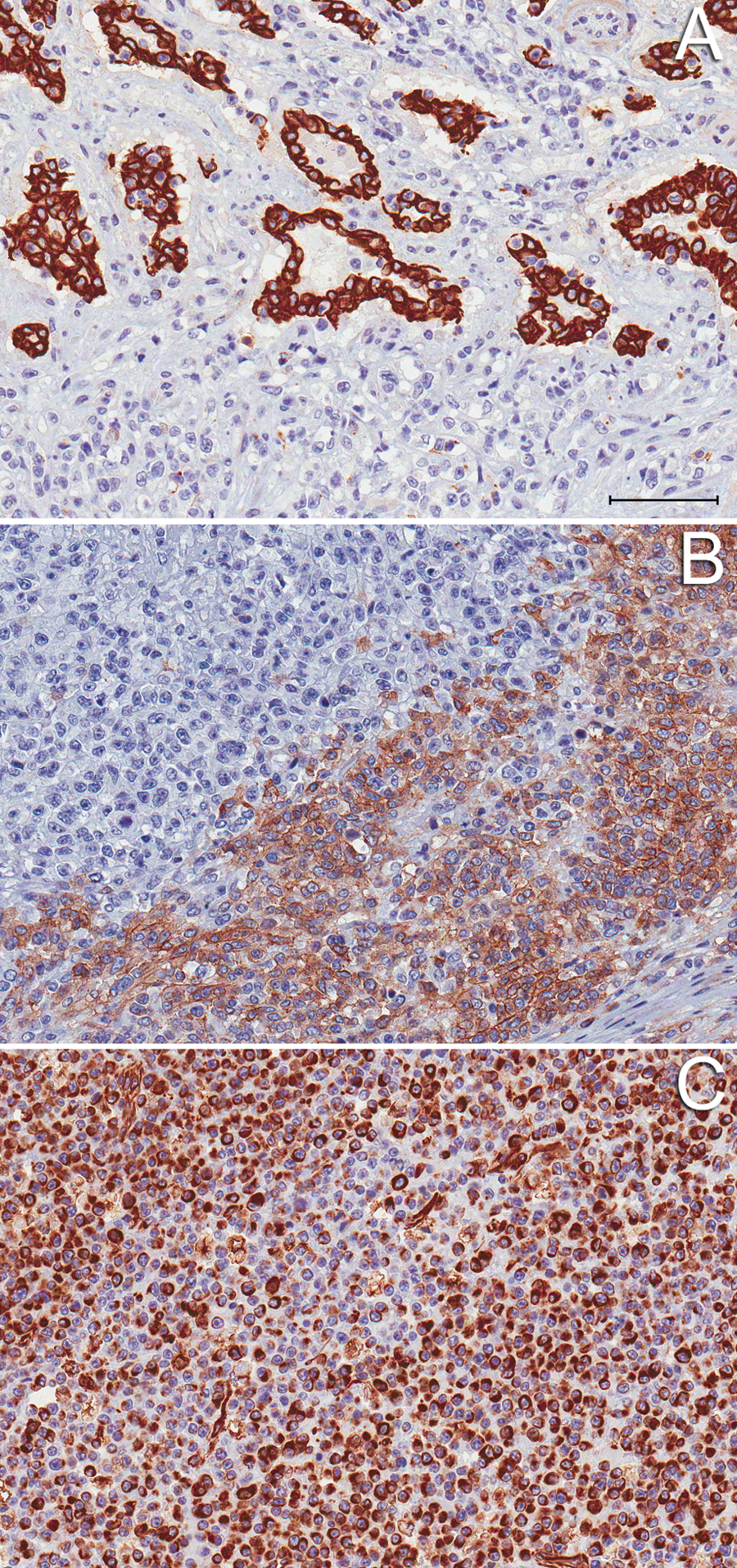
Immunohistochemical staining profile of the suprasellar germ cell tumor, mixed type; 16-month-old Wagyu heifer calf.
Primary intracranial tumors have been reported rarely in cattle, including choroid plexus papilloma, 2 primitive neuroectodermal tumor,7,19 diffuse intracranial oligodendroglioma, 5 nonfunctional pituitary adenoma, 18 and an anaplastic gangliocytoma. 4 The majority of localized central nervous system lesions in the cow are infectious by nature. 17
Intracranial GCTs have been described rarely in human beings and other animals, 20 representing approximately 1% of primary brain tumors in human beings. 6 Human patients are most commonly young adults. 20
Intracranial GCTs typically arise from the suprasellar region, 21 which is located in the most ventral region of the brain on midline, near the hypothalamus–pituitary region, and caudal to the optic chiasm. Germ cell tumors in this region can present similarly to pituitary adenomas, or with blindness, due to proximity or local extension into the pituitary gland or optic chiasm, respectively. 21
Five cases of suprasellar GCT in the dog were reported in a retrospective study; these cases presented with depression, mydriasis, ptosis, and visual deficits. 20 The masses in these dogs were classified as malignant and locally invasive, accounting for diverse clinical signs, such as bradycardia, polyuria, and polydipsia. Increased CSF protein was found in 2 of the cases. All masses were diagnosed on necropsy. Additionally, an intracranial GCT in a Miniature Pinscher, also diagnosed on necropsy, was reported; clinical signs included exophthalmos, conjunctivitis, polyuria, and eventually, complete blindness. 13
In human beings, intracranial GCTs are divided into different classifications based on histologic appearance (World Health Organization classification): germinoma, teratoma, embryonal carcinoma, choriocarcinoma, yolk sac tumor, and mixed tumor.6,8,16,20 Typically, germinoma and teratoma carry a better prognosis following treatment with radiation therapy and surgical excision, respectively. 6 Germinoma is the most common histopathologic subtype, 16 representing 65% of all intracranial GCTs. 6 This classification system has not been adapted for use in veterinary medicine; however, a similar classification scheme may be indicated, as cases of dogs with intracranial GCTs are now more frequently recognized and reported. According to histologic description reported in canine GCTs, tumors appear to be consistent with a mixed tumor type, having components of both germinoma and teratoma.13,20
Histologically, germinoma will display a 2-cell pattern consisting of large neoplastic cells and small lymphocytic cells.9,16 The neoplastic cells are typically polygonal in shape and have central nuclei with prominent nucleoli. Necrosis is rare, although mitotic figures are frequently seen. 16 Grossly, germinoma will be a solid, well-circumscribed mass that appears gray–pink on cut surface. 16 Yolk sac tumors, on the other hand, are frequently composed of epithelial cells, ranging from cuboidal to columnar in appearance, and organized in sheets, cords, or other various patterns. 16
Immunohistochemistry is used to differentiate between the types of GCT for a more definitive diagnosis as well as treatment guidance in human patients. Common markers tested for include AFP, b-HCG, and PLAP.6,9,12,16,20,21 Serum titers for AFP and HCG can be performed in lieu of immunohistochemistry. 8 Up to 70% of germinomas stain positive for PLAP, while only some will be positive for AFP or b-HCG. Conversely, yolk sac tumors will more frequently stain positive for AFP, while rarely staining positive for PLAP and generally negative for b-HCG. 16 Mutations in c-kit have been identified in approximately 25% of intracranial germinomas of human beings, resulting in increased levels of KIT protein expression on the cell surface.3,11,15,16
The diagnosis of suprasellar GCT was made in the current case based on the location of the mass and histologic appearance. Immunohistochemical phenotype combined with the tumor morphology based on hematoxylin and eosin staining support a mixed tumor type. Placental alkaline phosphatase, AFP, and b-HCG were all negative, and cytokeratin and c-kit were positive, indicating epithelial 22 (as seen in yolk sac tumors) and germinoma 16 components to the tumor, respectively. Vimentin immunoreactivity has been described in human cases of intracranial and gonadal GCTs, 10 and therefore supports a diagnosis of GCT in the current case.
Germ cell tumors arising from the suprasellar region will commonly result in clinical signs consistent with hypothalamic/pituitary dysfunction, or more specifically, diabetes insipidus 14 due to local compression or invasion of the mass. The heifer in the present case had clinical signs consistent with diabetes insipidus: polyuria, polydipsia, isosthenuria in the face of relative dehydration, serum hypernatremia, and CSF hypernatremia. Additionally, the heifer appeared subjectively small for her age, which may have been a sign of secondary hypopituitarism. Interestingly, the total protein in the heifer’s CSF was increased, which was also seen in 2 of the 5 dogs with suprasellar GCTs. 20 Such changes are consistent with effects associated with intracranial neoplasms.
In conclusion, primary intracranial masses are rare in cattle; therefore, an antemortem diagnosis of suprasellar GCT was not obvious in the present case. Definitively diagnosing intracranial masses in cattle is challenging due to physical and financial constraints and is more likely to occur on post mortem examination, if at all. This finding, along with the relatively recent findings in dogs, indicates a more broad species incidence than previously thought. Therefore, intracranial GCT, though rare, should be included as a clinical differential diagnosis for an intracranial mass.
Footnotes
a.
Antibodies (AFP, HCG, c-kit, PLAP, cytokeratin, and vimentin), Dako North America Inc., Carpinteria, CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
