Abstract
Inclusion bodies (IBs) and multinucleate cells can be associated with viral infections; however, IBs and multinucleate cells have been described in normal tissue and with non-viral disease processes in multiple species. We examined fundic stomach from 50 callitrichids histologically for bi- and multinucleate parietal cells and cytoplasmic IBs in gastric epithelial cells. Callitrichids represented included 6 genera: Saguinus (4 spp.), Leontopithecus (1 sp.), Mico (3 spp.), Cebuella (1 sp.), Callithrix (1 sp.), Callimico (1 sp.), and 13 unspecified marmosets. Gastric epithelial IBs were present in 46 of 47 (98%) of the callitrichids from which the stomach was sufficiently well preserved to identify IBs. Cytoplasmic IBs were identified in gastric surface pit epithelial cells (43 of 44, 98%), mucous neck cells (43 of 44, 98%), parietal cells (43 of 44, 98%), and chief cells (43 of 44, 98%). The IBs were eosinophilic, ovoid, round, elongate, or variably indented, sometimes slightly refractile, and 1–6 × 1–13 µm. IBs were sometimes perinuclear and molded around the nucleus. Electron microscopy of the gastric epithelium of one marmoset indicated that IBs were composed of intermediate filaments. The IBs did not stain with immunohistochemical markers for cytokeratin AE1/AE3 or vimentin. Binucleate parietal cells were found in 49 of 50 (98%) callitrichids, and multinucleate parietal cells were observed in 40 of 49 (82%) callitrichids. Gastric epithelial cytoplasmic IBs and bi- and multinucleate parietal cells are likely a normal finding in callitrichids, and, to our knowledge, have not been reported previously.
The Callitrichidae are a family of New World monkeys distributed throughout South and Central America. The 6 extant genera of callitrichids include tamarins (Saguinus), lion tamarins (Leontopithecus), Atlantic Forest and dwarf marmosets (Callithrix), Amazonian marmosets (Mico), pygmy marmosets (Cebuella), and Goeldi’s marmoset (Callimico).5,20 These animals are popular in zoo exhibits, are used as disease models in research, and some species are kept as pets. Knowledge of the normal histology of callitrichids is necessary to facilitate differentiation between incidental or normal findings and pathologic changes.
Inclusion bodies (IBs) are defined as any stainable aggregate of material in the nucleus or cytoplasm of a cell. 2 Pathologic IBs can be caused by a variety of viral infections. Given the zoonotic implications, the measles virus, a morbillivirus of the Paramyxoviridae family, is of particular concern in primates. 17 This virus can form intranuclear and cytoplasmic IBs in numerous tissues.1,16,17 In marmosets, viral IBs associated with the measles virus infection have been identified in epithelial cells of the gastrointestinal tract, bronchial epithelium, alveolar epithelial cells of the lung, liver parenchyma, and pancreatic ducts. 1 Non-viral IBs consisting of intermediate filaments have been described in various disease conditions in humans, including neurodegenerative diseases 23 and rhabdoid tumors. 25
IBs can also be an incidental finding. Cytoplasmic filamentous IBs, composed of tonofilaments, have been described in the urinary bladder epithelium of rhesus monkeys. 6 Cytoplasmic hyaline IBs composed of microtubule aggregates are commonly identified in the hepatocytes of stranded cetaceans. 27 Neuronal cytoplasmic IBs that appear to be non-pathologic have been described in the dorsal horn and supraoptic nucleus of squirrel monkeys, 4 in the lateral vestibular nucleus of rats, 26 and in the lateral geniculate nucleus of cats. 19
Multinucleate cells may represent syncytia formed as a consequence of viral infection. Syncytia can be observed in primates infected with measles virus, parainfluenza virus, various herpesviruses, and simian immunodeficiency virus.1,3,16,17 Multinucleate cells can occasionally be the result of medical treatment or non-viral disease processes. Multinucleate parietal cells have been observed in the gastric mucosa of children treated with omeprazole. 9 Additionally, multinucleate gastric parietal cells have been identified in human patients that have uremia 10 or are on immunosuppressive therapy after renal transplantation. 11
In some species, multinucleation can be a normal or incidental finding in tissues in which cells are not typically multinucleate. Multinucleate renal collecting duct epithelial cells in macaques and multinucleate hepatocytes in gorillas, chimpanzees, and macaques are considered incidental. 15 Porcine cardiac myocytes commonly contain 4–16 nuclei. 24 Multinucleate cells can be present in the seminiferous tubules and epididymis of normal Beagles, with younger dogs having more multinucleate cells than older ones. 12 In humans, multinucleate stromal cells are commonly identified in normal anal mucosa. 13 Binucleate gastric parietal cells are a normal finding in humans and domestic animals.14,28
Given that IBs and multinucleate epithelial cells can be manifestations of viral infections and other pathologic processes, knowledge of the occurrence of normal or incidental IBs and multinucleation is essential for accurate assessment of disease. Prior postmortem examinations of callitrichids submitted to the Anatomic Pathology Service at the University of Florida Veterinary Diagnostic Laboratories (APS-UF-VDL) had identified multinucleate parietal cells and cytoplasmic IBs in gastric epithelial cells. However, detailed survey and investigation into the cause or origin and significance of these observations has not been reported previously. Thus, our goals were to 1) describe and document the prevalence of gastric epithelial IBs and bi- and multinucleate parietal cells in callitrichids; 2) determine whether there is an association between the presence of IBs or bi- and multinucleate parietal cells with signalment; and 3) determine whether there is an association between the presence or number of multinucleate parietal cells and renal disease.
Materials and methods
For case identification, the APS-UF-VDL postmortem archives (1976–2019) were searched for the keywords “marmoset”, “tamarin”, and “primate”. Cases were classified according to taxonomy (marmoset, tamarin, and Goeldi’s monkey), and demographic information, including genus, age, and sex, was collected from the pathology reports and medical records, where available. Animals were assigned groups by age: neonate (<1 wk), infant (≥1 wk and <6 mo), juvenile ( ≥6 mo and <1 y), and adult (≥1 y); however, given that there were only 1–4 animals in each of the non-adult categories, animals were categorized as adult and non-adult for statistical analysis.
All archival tissues had been fixed in 10% neutral-buffered formalin, processed routinely, and sections stained with hematoxylin and eosin (H&E). Each section of fundic stomach was examined by 2 of the authors, including one American College of Veterinary Pathologists boarded veterinary pathologist, for the presence of bi- and multinucleate parietal cells and the maximum number of nuclei seen in the parietal cells. The total number of bi- and multinucleate parietal cells in ten 400× fields was also assessed; counts were made from 10 consecutive fields; fields with autolysis that prohibited identification of features were not counted. The presence of IBs in surface pit epithelial cells, mucous neck cells, parietal cells, and chief cells was subjectively evaluated and scored: 0 = none, 1 = few to low, 2 = moderate, 3 = many. Given an association between multinucleate parietal cells and uremia in humans, kidney sections, where available, were evaluated for lesions in the glomeruli, tubules, interstitium, and vasculature. Any renal lesions (e.g., inflammation, degeneration, necrosis, etc.) identified in those areas were graded by the following scale: 0 = none, 1 = minimal, 2 = mild, 3 = moderate, 4 = marked.
Electron microscopy
Formalin-fixed tissue from the stomach of a female marmoset (species and age not specified) was minced and post-fixed in Karnovsky fixative overnight, rinsed in phosphate buffer, then post-fixed in 1% osmium tetroxide, stained en bloc with 0.5% aqueous uranyl acetate, dehydrated in a series of ethanol, acetone, and propylene oxide washes, and embedded in Epon–Araldite epoxy (composed of Araldite 502, Embed-812, DDSA, and DMP-30; Electron Microscopy Sciences). Thick plastic sections stained with toluidine blue were examined. Areas were selected for 65–75 nm thin sections, which were then post-stained with uranyl acetate and lead citrate and examined with a transmission electron microscope (JEM-1011; Jeol).
Immunohistochemistry
Immunohistochemistry (IHC) for cytokeratin (CK) AE1/AE3 (AE1/AE3-L-CE, 1:100; Leica) and vimentin (PA0640, ready-to-use; Leica) was performed on formalin-fixed, paraffin-embedded fundic stomach from one marmoset, one tamarin, and one Goeldi’s monkey (Bond RX automated stainer, Bond polymer refine detection kit; Leica). Heat-induced antigen retrieval was performed at 100°C for 20 min in pH 6.0 buffer. The chromogen was 3,3′-diaminobenzidine tetrahydrochloride hydrate. Slides were counterstained with either a standard H&E stain (CK) or hematoxylin alone (vimentin).
Statistical analysis
Chi-square tests were used to assess the correlation between the number of binucleate parietal cells, number of multinucleate parietal cells, and maximum number of nuclei in parietal cells versus sex, age group (adult or non-adult), and taxonomy (marmoset, tamarin, or Goeldi’s monkey). Wilcoxon and Kruskal–Wallis tests were used to assess the correlation between the presence of multinucleate parietal cells and grade of IBs in the different gastric cell types versus sex, age group (adult or non-adult), and taxonomy (marmoset, tamarin, or Goeldi’s monkey). The correlation between the number of multinucleate parietal cells and lesion grade in the kidney was evaluated with a chi-square test. A Spearman correlation analysis was used to assess correlation between age and number of bi- and multinucleate parietal cells, maximum number of nuclei in parietal cells, and grade of IBs in the different gastric cell types. A value of p ≤ 0.05 was considered significant.
Results
The search of archived postmortem materials yielded 124 callitrichid cases. Of these cases, 56 cases had a fundic stomach sample available for histologic examination. However, 4 cases were excluded because of autolysis, and 2 cases were excluded because they lacked required demographic information.
Callitrichid cases in our study included 34 marmosets, 13 tamarins, and 3 Goeldi’s monkeys. These animals comprised 6 genera: 10 Saguinus (4 spp.), 3 Leontopithecus (1 sp.), 3 Mico (3 spp.), 9 Cebuella (1 sp.), 9 Callithrix (1 sp.), and 3 Callimico (1 sp.). The remaining 13 cases were identified as marmosets with no further taxonomic information. There were 27 males, 22 females, and 1 of unknown sex. The age groupings included 1 full-term fetus, 2 neonates, 4 infants, 3 juveniles, 38 adults, and 2 callitrichids of unspecified age. Histologic sections of kidneys were available for examination in 48 of 50 cases. All callitrichids were privately owned or from a zoologic institution.
Gastric epithelial IBs were eosinophilic, ovoid, round, elongate, or variably indented, were exclusively located in the cytoplasm, and 1–6 × 1–13 µm (Figs. 1–5). The IBs were often found adjacent to the nucleus of the cell and sometimes exhibited molding around the nucleus (Fig. 1). Gastric epithelial IBs were present in 46 of 47 (98%) of the callitrichids that had a gastric fundic sample sufficiently well preserved to identify IBs. IBs were seen in all cell types (Fig. 2), including surface pit epithelial cells (Fig. 1), mucous neck cells (Fig. 3), parietal cells (Fig. 4), and chief cells (Fig. 5) in all animals with IBs in which all cell types could be assessed. IBs were typically single within cells, but occasional cells contained 2–4 smaller IBs (Fig. 4). Given that autolysis in some cases affected the ability to identify IBs in some cell types, not every cell type could be assessed for every case. Each cell type had IBs in 43 of 44 cases (98%). The mean grade for IBs was 2.8 ± 0.7 (±SD) for surface pit epithelial cells, 2.0 ± 0.9 for mucous neck cells, 1.3 ± 0.6 for parietal cells, and 1.5 ± 0.9 for chief cells (Table 1). The one animal without IBs was a neonatal Goeldi’s monkey. IBs were present in all cell types in the term fetus and in the other neonate in the study (Saguinus sp.). Although not specifically assessed in our study, given that sections of stomach were typically only fundic, similar IBs were identified in the surface pit and glandular epithelium in the cardiac and/or pyloric regions of the stomach in a few animals. IBs were identified in the cardiac region of the stomach in 2 of 3 marmosets and 2 of 3 tamarins, and in the pyloric region in 6 of 6 marmosets and 4 of 4 tamarins.
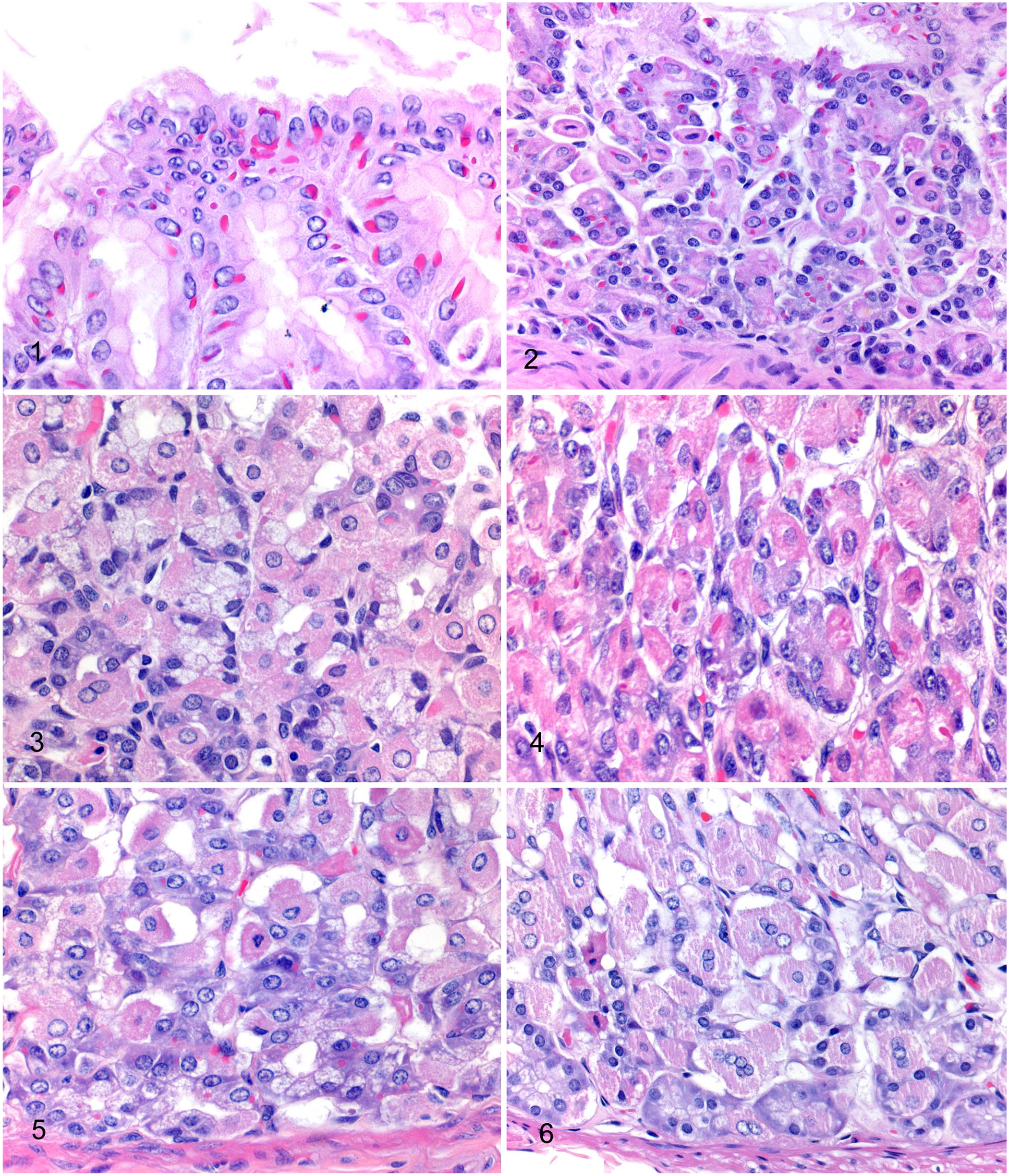
Gastric fundic mucosa of callitrichids. H&E.
Number of bi- and multinucleate parietal cells, maximum number of nuclei in parietal cells, and grade of cytoplasmic inclusion bodies in gastric epithelial cells in callitrichids.
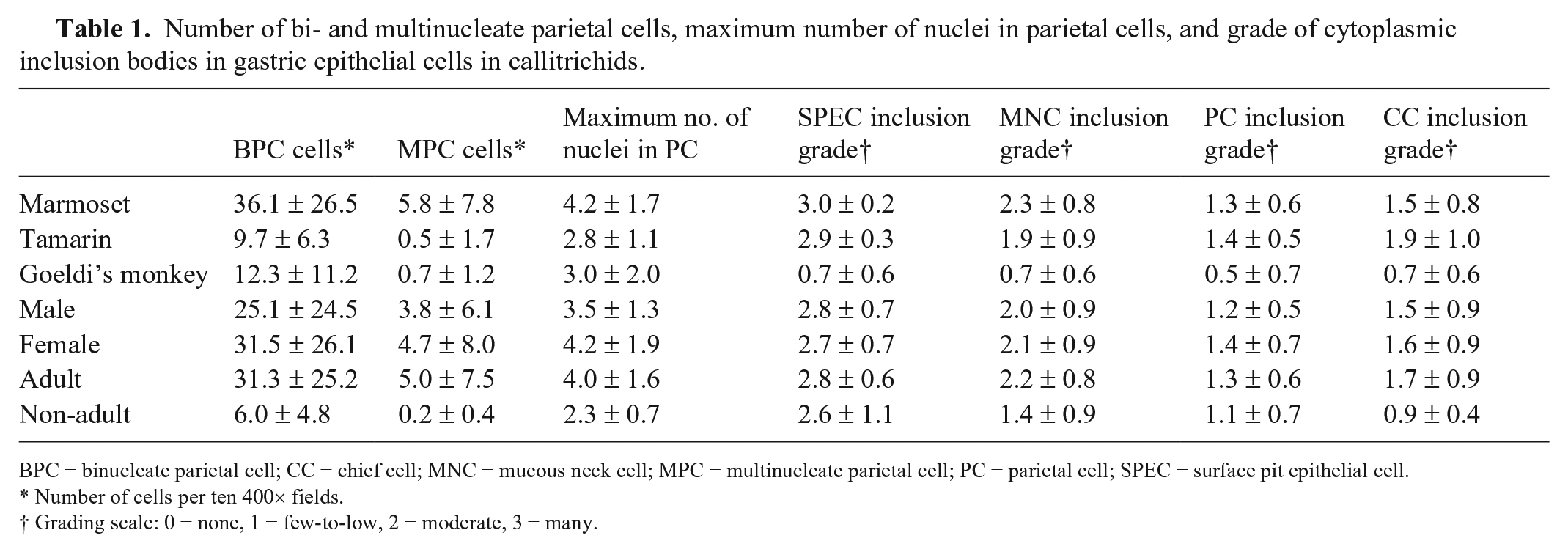
BPC = binucleate parietal cell; CC = chief cell; MNC = mucous neck cell; MPC = multinucleate parietal cell; PC = parietal cell; SPEC = surface pit epithelial cell.
Number of cells per ten 400× fields.
Grading scale: 0 = none, 1 = few-to-low, 2 = moderate, 3 = many.
Binucleate parietal cells (Fig. 6) were present in 49 of 50 cases (98%), with a range of 3–91 binucleate cells in ten 400× fields in cases in which bi- and multinucleate cells could be quantified (n = 48; Table 1). Autolysis sometimes allowed identification of these cells, but limited quantification of these cells to < 10 fields. Multinucleate parietal cells (Fig. 6) were present in 40 of 49 cases (82%), with a range of 1–32 multinucleate cells in ten 400× fields in cases in which multinucleate cells were quantifiable. Multinucleate parietal cells contained a maximum of 3 (n = 20 animals), 4 (n = 6), 5 (n = 7), 6 (n = 4), 7 (n = 1), 8 (n = 1), or 9 (n = 1) nuclei, and sometimes exhibited prominent cytomegaly compared to mono- and binucleate parietal cells.
Marmosets had significantly more binucleate parietal cells (36.1 ± 26.5) than did tamarins (9.7 ± 6.3; p = 0.0045), but there was no significant difference between either marmosets or tamarins and Goeldi’s monkeys (12.3 ± 11.2; Table 1). Marmosets (5.8 ± 7.8) had significantly more multinucleate parietal cells than tamarins (0.5 ± 1.7; p = 0.0004), but there was no significant difference between either marmosets or tamarins and Goeldi’s monkeys (0.7 ± 1.2).
Goeldi’s monkeys had a significantly lower grade of IBs in surface pit epithelial cells (0.7 ± 0.6) than did tamarins (2.9 ± 0.3; p = 0.0012) and marmosets (3.0 ± 0.2; p < 0.0001; Table 1). Goeldi’s monkeys had a significantly lower grade of IBs in mucous neck cells (0.7 ± 0.6) than did marmosets (2.3 ± 0.8; p = 0.0116), but there was no significant difference between either Goeldi’s monkeys and tamarins (1.9 ± 0.9) or marmosets and tamarins. There was no significant difference in the grade of IBs in parietal cells or chief cells between types of monkey.
Adult callitrichids had significantly more binucleate parietal cells (31.3 ± 25.2) than did non-adults (6.0 ± 4.8; p = 0.0002), and adults (5.0 ± 7.5) also had significantly more multinucleate parietal cells than did non-adults (0.2 ± 0.4; p = 0.0095; Table 1). Adults had significantly higher maximum number of nuclei in parietal cells (4.0 ± 1.6) than did non-adults (2.3 ± 0.7; p = 0.0012). Adults had a significantly higher grade of IBs in mucous neck cells (2.2 ± 0.8) than did non-adults (1.4 ± 0.9; p = 0.0304). Adults had significantly higher grade of IBs in chief cells (1.7 ± 0.9) than did non-adults (0.9 ± 0.4; p = 0.0174). There was no significant difference in the grade of IBs in surface pit epithelial cells or parietal cells between adult and non-adult animals.
The neonatal Goeldi’s monkey without IBs also had no bi- or multinucleate parietal cells. The other neonatal monkey (Saguinus sp.) had a low number of binucleate parietal cells (7 in ten 400× fields), 1 parietal cell with 3 nuclei, moderate numbers of parietal cell IBs, and few-to-low numbers of IBs in all remaining gastric epithelial cell types. The fetus (Leontopithecus sp.) had a few binucleate (4 in ten 400× fields) but no multinucleate parietal cells and few-to-low numbers of IBs in all gastric epithelial cell types. There was a significant correlation between age and the grade of IBs in mucous neck cells (p = 0.0438) and chief cells (p = 0.0076), but no significant correlations between age and the number of bi- or multinucleate parietal cells, the maximum number of nuclei in parietal cells, or the grade of IBs in surface pit epithelial cells or parietal cells.
No significant association was identified between sex and the grade of IBs in any epithelial cell type or the number of bi- or multinucleate parietal cells. There was also no association between grade of renal lesions and the presence or number of multinucleate parietal cells.
No immunoreactivity for CK AE1/AE3 or vimentin was identified in IBs. For CK AE1/AE3 in the sections from the marmoset, immunoreactivity was strong in pancreatic duct epithelium, mesothelial cells, gastric surface pit epithelial cells, and gastric mucous neck cells; moderate-to-strong in parietal cells; weak in chief cells; and absent in pancreatic acinar cells. In the Goeldi’s monkey tissue sections stained with CK AE1/AE3, immunoreactivity was strong in all gastric epithelial cell types, mesothelium, and all epithelial cells in the large intestine and lung. In the sections from the tamarin, immunoreactivity for CK AE1/AE3 was strong in gastric surface pit epithelial cells, gastric mucous neck cells, esophageal epithelium, pancreatic duct epithelium, mesothelial cells, and urinary bladder epithelium; moderate-to-strong in small intestinal epithelial cells; and moderate in gastric chief and parietal cells and pancreatic acinar cells. Vimentin immunoreactivity was strong in fibroblasts, vascular smooth muscle cells, vascular endothelial cells, nerves or ganglia (neurons were not immunoreactive), adipocytes, and mucosal and submucosal round cells in all 3 monkeys. In mesothelial cells, vimentin immunoreactivity was strong in the marmoset, moderate-to-strong in the Goeldi’s monkey, and weak-to-moderate in the tamarin. Various amounts of clumped, moderate-to-strong vimentin immunoreactivity was present in 25–50% of the smooth muscle cells of gastrointestinal muscularis in the marmoset. Vimentin immunoreactivity was sparse in the muscularis of the stomach and intestine in the tamarin and Goeldi’s monkey, and present in 5–10% and < 5% of smooth muscle cells, respectively. Vimentin immunoreactivity was not present in the muscularis of the urinary bladder in the tamarin.
Ultrastructural examination identified circumscribed, non–membrane-bound, intracytoplasmic accumulations of electron-dense tangled filaments in gastric mucosal epithelial cells (Fig. 7). IBs were in all epithelial cell types, but most often in mucous cells. IBs were usually perinuclear with occasional nuclear molding, ovoid-to-round, occasionally had an irregular outline, and were 2–3.5 µm × 1.5–2 µm. Filaments were 10–17 nm diameter and had a faint regularly spaced banded pattern.
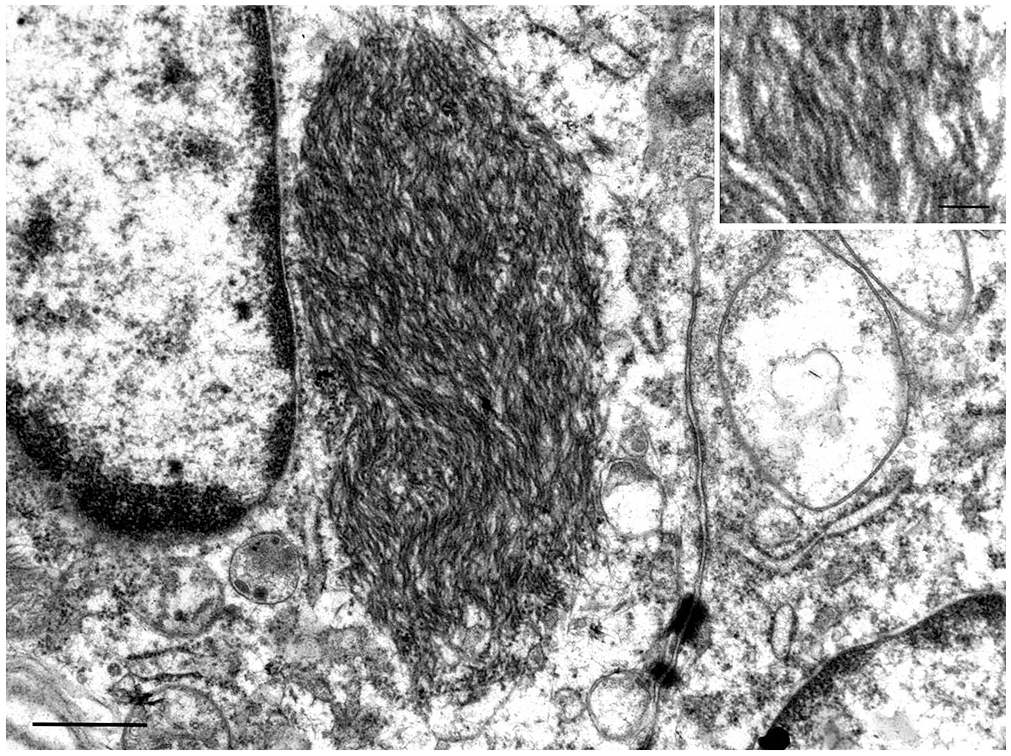
Transmission electron micrograph of an epithelial cell in marmoset (species unknown) gastric mucosa with an ovoid perinuclear, unencapsulated, intracytoplasmic inclusion body composed of a tangle of filaments. Bar = 600 nm. Inset: filaments are 10–17 nm diameter and have faint regular banding. Bar = 100 nm.
Discussion
Gastric epithelial IBs and bi- and multinucleate parietal cells are likely an incidental finding in callitrichids. Causes of morbidity and mortality were varied in the callitrichids and included infectious and noninfectious conditions affecting a variety of organ systems. No apparent association with other disease processes was identified with either IBs or multinucleate parietal cells. The presence of IBs in nearly every animal and the presence of multinucleate cells in most animals would also not be expected if these findings were linked to a particular disease process.
IBs were identified in every animal except for one. IBs were present in a term fetal tamarin and a neonatal tamarin, hence IBs can be present from birth. IBs may become more numerous with age, at least in some gastric epithelial cell types. The one animal that lacked IBs was a neonatal Goeldi’s monkey. There were only 3 Goeldi’s monkeys in our study, and the other 2 were adults. Both of the adult Goeldi’s monkeys had the lowest grade (few-to-low numbers) of IBs in all cell types that were assessed. There was a significant difference between the number of IBs in surface pit epithelial cells in Goeldi’s monkeys and both marmosets and tamarins, and a significant difference in the number of IBs in mucous neck cells in Goeldi’s monkeys compared to marmosets. Although it does appear that these IBs are less numerous in Goeldi’s monkeys, statistical analysis was hindered by the low number in our study.
IBs were overall most numerous in surface pit epithelial cells, followed by mucous neck cells, chief cells, and then parietal cells. All mature gastric epithelial cell types develop from a common stem cell, which is located in the gastric pit. Progeny cells migrate bidirectionally in the glands. 18 Formation of these IBs would be unlikely to be related to increasing age within these cells, given that the lifespan of surface mucous cells in the mouse is only 3–5 d, whereas chief cells may live for several months. 18 Speculatively, these IBs could originate in the stem cells, pass into the mature cells, and are then removed or broken down over time. Cell function could also speculatively have some impact on the presence of IBs.
Binucleate parietal cells have been described in normal gastric mucosa in humans and domestic species.14,28 Binucleate parietal cells were present in 49 of 50 animals; the 1 animal without binucleate parietal cells was the Goeldi’s monkey neonate that had no identified IBs. In callitrichids, binucleate parietal cells appear to be an incidental finding and may increase with age. Multinucleate cells were present in 40 of 49 cases. Again, adults had significantly more multinucleate cells than non-adults, as well as a higher maximum number of nuclei in parietal cells; however, a correlation was not seen between age and the number of multinucleate cells or maximum number of nuclei in parietal cells. Age was only known for 35 animals, and this may have been a factor in statistical correlation. Alternatively, the number of multinucleate parietal cells and the number of nuclei in those cells may increase in early adulthood and then plateau.
An increase in the number of multinucleate cells with age in an organ is not unprecedented. In humans, multinucleation of testicular stromal cells, germ cells or spermatids, and Leydig cells all appear to be associated with aging.8,21,22 In rodents and primates, multinucleate hepatocytes are observed in older animals.7,29 Possible mechanisms for formation of multinucleate cells include nuclear division without cell division, and cell fusion. It has been proposed that cell fusion is the mechanism for the formation of multinucleate spermatids in humans. 21 The mechanism for multinucleation in parietal cells in callitrichids is unknown.
Although adult animals had more renal lesions, the presence of multinucleate cells was not associated with the grade of renal lesions. Multinucleate parietal cells have been identified in human patients with uremia. 10 Given that few animals had recent chemistry panels, it was not possible to determine if azotemia was a factor. Although renal function is not always correlated with histopathology, it seemed unlikely that multinucleation of parietal cells was related to renal disease given the lack of correlation with renal lesions. Additionally, the presence of multinucleation in 82% of cases would also suggest that renal disease was unlikely to be a factor.
Interestingly, marmosets had more bi- and multinucleate parietal cells than tamarins. The reason for this is unknown. A smaller percentage of marmosets, for which the age class was known, were non-adults (6 of 32 non-adults, 19%) than tamarins (3 of 13 non-adults, 23%), hence age would not appear to have been a significant contributing factor.
By electron microscopy on one marmoset, IBs were consistent with intermediate filaments. No immunoreactivity was identified in the IBs for CK AE1/AE3 or vimentin in sections from a tamarin, a marmoset, and a Goeldi’s monkey. Although expected internal controls were generally strongly immunoreactive, there was some variation in staining in normal tissue, with weaker immunoreactivity for CK AE1/AE3 of some gastric epithelial cells, lack of CK AE1/AE3 immunoreactivity of pancreatic acinar cells in the marmoset (although moderate immunoreactivity of pancreatic acinar cells was identified in the tamarin), and variable, often sparse, and sometimes absent immunoreactivity for vimentin in smooth muscle of the gastrointestinal and urinary bladder muscularis. Therefore, these markers may not stain all epithelial and mesenchymal cells in callitrichids, as was expected. It is also possible that there is modification of the intermediate filaments, such as changes in structure or folding or immunologic modification, which would alter immunoreactivity to these antibodies. Alternatively, these IBs could be composed of an intermediate filament other than cytokeratin or vimentin. Additional immunohistochemical stains for different cytokeratins and other intermediate filaments could be considered in future studies.
Footnotes
Acknowledgements
We thank the UF-VDL Histology Laboratory for their assistance in preparing slides for our study, and Dr. Michael Garner for his consultation on the first case in which IBs were identified.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
