Abstract
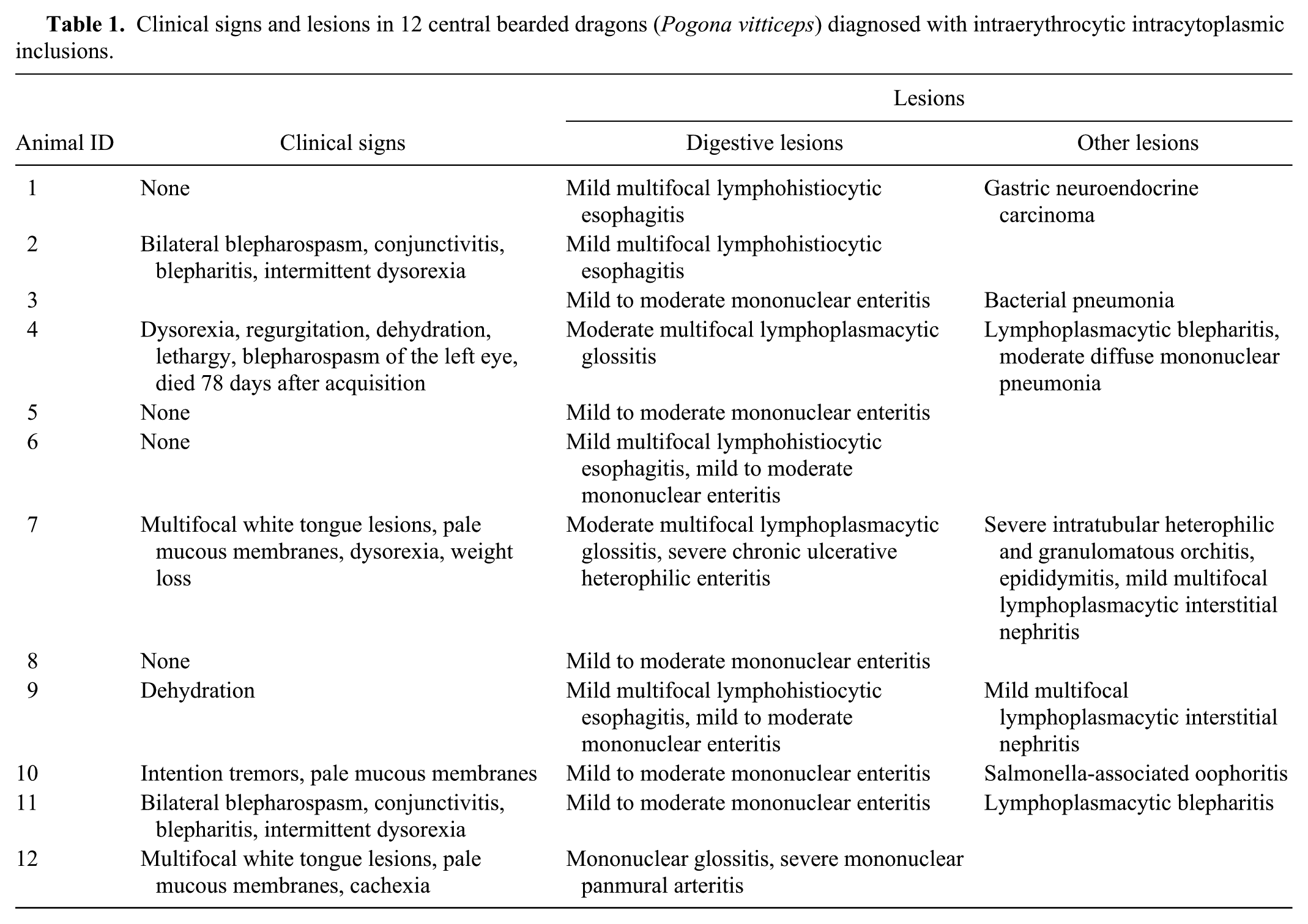
Three adult central bearded dragons (Pogona vitticeps) originating from a commercial breeding facility presented with clinical signs, including anorexia, dehydration, white multifocal lesions on the dorsal aspect of the tongue, blepharospasm, and weight loss. In 1 of 3 lizards, a marked regenerative anemia was noted, and all 3 bearded dragons had erythrocytic intracytoplasmic inclusion bodies. Nine bearded dragons housed in contact also had identical, but fewer intraerythrocytic inclusions. Inclusion bodies examined by electron microscopy had particles consistent with iridoviruses. Attempts to culture the virus were unsuccessful; however, amplification and sequencing of regions of the viral DNA polymerase by polymerase chain reaction confirmed the presence of an iridovirus. One of the bearded dragons died, while the 2 others showing clinical signs were euthanized. The remaining 9 infected bearded dragons of the teaching colony were also euthanized. Postmortem examination revealed a moderate, multifocal, lymphoplasmacytic or mononuclear adenitis of the tongue in the 3 bearded dragons, and a lymphohistiocytic hepatitis with bacterial granulomas in 2 lizards.
Introduction
Reptile erythrocytic iridoviruses (EIVs), historically misidentified as the protozoans Pirhemocyton, Toddia, and possibly Cytamoeba, have been previously reported in squamates within the families Gekkonidae, Phyllodactylidae, Scincidae, Cordylidae, Lacertidae, 34 Pythonidae, Colubridae,6,44 Viperidae, Varanidae, Iguanidae, Phrynosomatidae, Agamidae, and Chamaeleonidae. 37 Within the family Agamidae, EIVs have been found in the Bibron’s agama (Agama impalearis) 2 and the common agama (A. agama).18,26 In some cases, erythrocytic inclusions have been associated with marked anemia,17,26 but experimental infection has failed to reproduce anemia unless the lizards were maintained in a cold environment. 3 Many viral diseases of poikilothermic vertebrates are highly temperature dependent, including iridoviruses, and the current data does not differentiate among the relative roles of host and virus on temperature-dependence of disease.19,30 Therefore, the pathogenicity of EIVs in reptiles is not well defined. The current report describes a novel erythrocytic iridovirus associated with intracytoplasmic erythrocytic inclusion bodies in central bearded dragons (Pogona vitticeps) in California; only insect iridovirus DNA has been previously identified by polymerase chain reaction (PCR) from bearded dragons. 20
Materials and methods
Animals
Eleven adult captive-bred bearded dragons, including 5 males and 6 females, were obtained in January 2012 from a private breeder in California as part of a teaching colony and, based on caging, were identified by numbers 1–11. An additional sick juvenile male bearded dragon was requested and obtained from the same breeder in November 2012 and identified as no. 12. The project was carried out according to protocols approved by the Institutional Animal Care and Use Committee of the University of California, Davis (IACUC protocol no. 16899).
Housing and diet at the breeding facility
At the breeding facility, the bearded dragons were maintained in a greenhouse, where wildlife contact was possible, including but not limited to fence lizards (syn. spiny lizards, Sceloporus spp.), western gopher snakes (Pituophis catenifer), and house mice (Mus musculus). Captive amphibians, Lawson’s dragons (syn. dwarf bearded dragon, Pogona henrylawsoni), and various species of geckos were also housed in the same facility, but were never in direct contact with the bearded dragons; researchers did not directly observe husbandry protocols at the facility. The floor of the greenhouse was planted with live clover, intended to serve as both substrate and calcium supplement for the bearded dragons. In addition, the lizards were fed house crickets (Acheta domesticus), yellow mealworms (Tenebrio molitor), and various leafy greens. At the time of acquisition, the lizards were in brumation. The temperature of the environment was progressively increased. All bearded dragons showed normal appetite a week after acquisition.
Quarantine
Quarantine protocol on arrival at the University of California, Davis included a complete physical examination, repeated fecal parasitologic examinations, and venipuncture from the ventral coccygeal vein for packed cell volume and total solid measurement for each bearded dragon. Cutaneous mites were identified during the initial physical examination of the animals, and each dragon was treated with ivermectin a at 0.2 mg/kg subcutaneously, repeated after 14 days.
Housing and diet in the teaching colony
At the University of California, Davis, the bearded dragons were housed individually in 60-cm wide, 40-cm high, 40-cm deep glass aquaria equipped with full-spectrum fluorescent lighting b set on a 12-hr daily cycle, with infrared lamps c providing a temperature gradient ranging from 28°C to 31°C. The housing during quarantine was identical as no other reptile was kept in the facility; thus, no specific quarantine room was needed. The diet consisted of chopped fresh dandelion leaves, cabbage, and occasionally romaine lettuce offered ad libitum daily; crickets and mealworms acquired from a commercial distributor were gut-loaded with calcium and vitamin supplement d and offered once daily. The type and quantity of food items provided were the same for each dragon.
Blood samples
One bearded dragon (no. 4) died 78 days following acquisition. A 1-ml blood sample was obtained from the ventral coccygeal vein of the remaining 11 lizards of the teaching colony 3 months after acquisition. A volume of 0.4 ml was stored in ethylenediamine tetra-acetic acid (EDTA) for complete blood cell count in 11 bearded dragons, and 0.5 ml was stored in lithium heparin in 10 bearded dragons for the evaluation of serum biochemistries. e The remaining bearded dragon (no. 12) did not have a biochemistry panel because of insufficient sample volume. Biochemistry panel included uric acid, blood urea nitrogen, glucose, total proteins, aspartate aminotransferase, glutamate dehydrogenase, cholesterol, creatinine kinase, phosphorus, and total calcium. Blood samples were obtained in April, therefore results were interpreted based on seasonal reference intervals from bearded dragons housed in the northern hemisphere in February (vs. reference intervals obtained in August). 35 Hematocrit and blood smears were obtained from each animal (n = 12). A postmortem heart-blood sample was obtained from lizard no. 4, stored in EDTA, and examined for intraerythrocytic inclusions.
Light microscopic examination
Blood films were fixed and stained with Wright–Giemsa for differential leukocyte counts and morphologic evaluation of erythrocytes, leukocytes, and thrombocytes. A leukocyte estimate was performed by counting all leukocytes in 10 fields under a 500× objective lens. The average number of cells per field was multiplied by 1,600 to obtain the estimated count. One hundred leukocytes were counted to determine the leukocyte differential.
Transmission electron microscopic examination
Whole blood collected from dragon no. 7 was stored in Karnovsky fixative prior to electron microscopic examination. Because of financial constraints, a single blood sample was examined by electron microscopy. The cells were rinsed in buffer for 40 sec, and then postfixed in 1% aqueous OsO4, dehydrated through a graded acetone series, infiltrated, and embedded in epoxy resin. Ultrathin sections (70–90 nm) were obtained with an ultramicrotome f and stained with 4% uranyl acetate in ethanol and lead citrate prior to examination by electron microscopy. g
Euthanasia protocol
Because of declining health of the surviving lizards despite supportive treatment, euthanasia was elected (n = 11). All dragons were sedated with midazolam at 1 mg/kg intramuscularly h just prior to euthanasia and euthanized by intravenous injection of 100 mg/kg of pentobarbital sodium into the ventral coccygeal vein. i One milliliter of blood was sampled from the cranial vena cava for PCR and viral culture prior to injection of 2.0 mEq/kg of KCl into the cranial vena cava.
Gross and histologic examination
Complete necropsies were performed on all animals immediately after death or euthanasia (n = 12). A complete set of tissues, including esophagus, stomach, trachea, liver, gall bladder, urinary bladder, spleen, kidney, heart, skeletal muscle, lung, tongue, and brain were collected from each animal and rapidly fixed in 10% neutral buffered formalin, routinely processed, sectioned at 4 µm thick, stained with hematoxylin and eosin, and examined by board-certified pathologists. In addition, samples of the skin, liver, spleen, heart, kidney, and lung were frozen for molecular diagnostics and viral culture.
Viral culture
Samples of blood, skin, liver, spleen, kidney, and lung were obtained from each lizard, and a portion of those tissues were homogenized for virus isolation following standard procedures. 39 Samples were inoculated onto EPC (epithelioma papulosum cyprini derived from fathead minnow, Pimephales promelas) j cells at 20°C for 2 passages as previously described.4,39,47 In addition, blood was inoculated onto BF-2 k (derived from bluegill sunfish, Lepomis macrochirus) at 20°C and viper heart cells l at 26°C. Cultures were examined for signs of cytopathic effect for a minimum of 1 month.
Polymerase chain reaction and sequencing
DNA was extracted from frozen-thawed samples of the skin, liver, spleen, heart, and lung of each lizard using a commercial extraction kit m as previously described. 44 Consensus PCR amplification for a partial sequence of the DNA-dependent DNA polymerase gene of large DNA viruses was performed using previously described methods.6,11,44 In addition, PCR was performed with consensus primers for the genus Ranavirus 25 and consensus primers for Rickettsiales groESL. 29
An additional PCR was performed with remaining heparinized blood from lizard no. 12. Pan-EIV primers EryIridF (5′-TGYAAYATHTTYTRYTGGGARGAYCA-3′) andEryIridR (5′-GTCATNGCNCCNGGCATRTAIGG-3′) were designed based on conserved areas of the available DNA-dependent DNA polymerase sequences from Thamnophis sauritus erythrocytic virus (TsEIV) 44 and Iberolacerta monticola (syn. Lacerta monticola) erythrocytic virus (LmEIV), as well as other members of the Iridoviridae family. The primers were used in 2 hemi-nested protocols with previously reported consensus primers for large DNA viruses. 11 Two separate first rounds were used, one using EryIridF and ConR (5′-CCCGAATTCAGATCTCNGTRTCNCCRTA-3′), and the other using ConF (5′-CGGAATTCTAGAYTTYGCNWSNYTNTAYCC-3′) and EryIridR (Fig. 1). The reaction mixture for the PCR amplifications consisted of 0.13 µl of Taq DNA polymerase, n 2 µl of 10× PCR buffer, 0.8 µl of 50 mM MgCl2, 0.4 µl of 10 mM deoxyribonucleotide triphosphates, 1 µl of 20 µM of forward and reverse primers, and 2 µl of DNA template. The reaction conditions were: 93°C for 1 min for 1 cycle followed by 93°C, 30 sec; 45°C, 2 min; and 72°C, 3 min for 35 cycles, followed by a single cycle at 72°C for 4 min. For the second round reactions, 2 µl of each round 1 product were used, and primers EryIridF and EryIridR were used with reaction conditions identical to round 1. The PCR products were resolved in 1.5% agarose gels. After electrophoresis, bands of interest were cut from the gel, and their DNA was extracted using a commercial gel extraction kit. m The products were then submitted for sequencing to the University of Florida Interdisciplinary Center for Biotechnology Research (Gainesville, Florida) and analyzed on commercial DNA sequencers. o Amplicons were sequenced twice in each direction, and primer sequences were edited out prior to constructing contiguous sequences.

Diagram of the primers, which were designed based on conserved areas of the available DNA-dependent DNA polymerase sequences from Thamnophis sauritus erythrocytic virus 44 and Iberolacerta monticola (syn. Lacerta monticola) erythrocytic virus, 1 as well as other members of family Iridoviridae, and genomic position of these primers.
Phylogenetic analysis
Predicted homologous 131–204 amino acid polymerase sequences were obtained from GenBank, with the RefSeq database (http://www.ncbi.nlm.nih.gov/refseq/) selected whenever possible, and were aligned using MAFFT. 21 Bayesian analyses of each alignment were performed using MrBayes 3.1 on the CIPRES server (Miller M, Pfeiffer W, Schwartz T: 2010, Creating the CIPRES Science Gateway for inference of large phylogenetic trees. In: Proceedings of the Gateway Computing Environments Workshop, pp. 1–8. New Orleans, Louisiana), with gamma distributed rate variation and a proportion of invariant sites, and amino acid substitution model jumping. 31 Statistical convergence was assessed by the standard deviation of split frequencies as well as potential scale reduction factors of parameters and was stopped when statistical convergence was reached. Two independent Bayesian analyses were run to avoid entrapment on local optima.
Maximum likelihood (ML) analyses of each alignment were performed using RAxML on the CIPRES server, 33 with gamma distributed rate variation and a proportion of invariant sites. The amino acid substitution model with the highest posterior probability in the Bayesian analysis was selected. Trichoplusia ni Ascovirus (GenBank accession no. AAC54632), a noniridoviral member of the nucleocytoplasmic large DNA viruses, was used as the outgroup. Family Ascoviridae has been found to be the sister group to the Iridoviridae family. 49 Bootstrap analysis was used to test the strength of the tree topology, with 1,000 subsets. 8
Results
Clinical presentation
Clinical signs are summarized in Table 1. Twenty-eight days after arrival at the University of California, Davis, a male bearded dragon (no. 7) presented with decreased appetite, chronic wasting, pale mucous membranes, and white, opaque, nonraised foci on the dorsal aspect of the tongue. Within a 2-month period, this bearded dragon lost approximately 25% of its body weight, which decreased from 362 to 290 g despite supportive care.
Clinical signs and lesions in 12 central bearded dragons (Pogona vitticeps) diagnosed with intraerythrocytic intracytoplasmic inclusions.
An adult female bearded dragon (no. 4) presented 7 days after acquisition with decreased appetite, regurgitation, dehydration, lethargy, and blepharospasm of the left eye. The animal had a mild blepharitis and an ulcer of the left cornea that was confirmed by a positive fluorescein test. Despite supportive care including antibiotic ophthalmic drops, p blepharospasm persisted, and the lizard died 78 days after acquisition in January.
Two other bearded dragons (nos. 2 and 11) presented with bilateral blepharospasm and intermittent dysorexia, and had mild conjunctivitis, blepharitis, and negative fluorescein staining on ophthalmologic examination. The same treatment protocol as dragon no. 4 was initiated.
An additional juvenile male bearded dragon (no. 12), acquired in November from the same breeder, presented with pale mucous membranes, cachexia, and white multifocal lesions on the dorsal aspect of the tongue similar to the lesions observed in bearded dragon no. 7.
Biochemistry results
Biochemistry results (n = 10) are summarized in Table 2. Biochemical abnormalities were variable depending on the lizard and included hypercholesterolemia in 9 lizards, slight to severe hyperglycemia in 2 lizards (390 and 1,045 mg/dl, respectively; reference [ref.] interval: 218–356 mg/dl 35 ) and mild to moderate hypoglycemia in 7 lizards. Two females (nos. 6 and 8) were markedly hypercalcemic (31.2 and 32.6 mg/dl, respectively; ref. interval: 12.3–16.5 mg/dl 35 ).
Biochemistry results of 10 central bearded dragons (Pogona vitticeps) from a teaching colony infected with an erythrocytic iridovirus.*
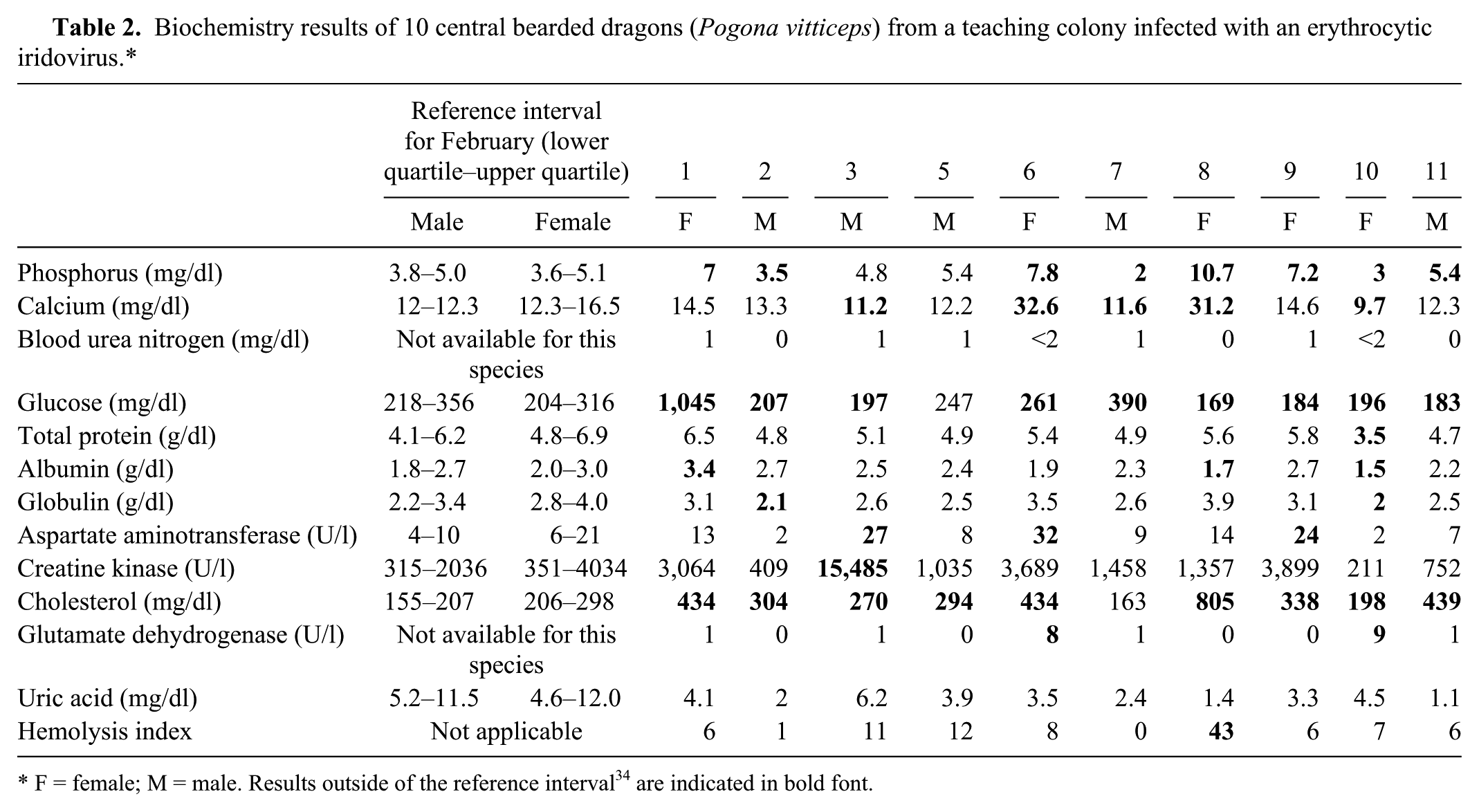
F = female; M = male. Results outside of the reference interval 34 are indicated in bold font.
Blood smear and complete blood cell count
In all bearded dragons (n = 11), intracytoplasmic erythrocytic inclusion bodies of various blue staining intensities were identified (Fig. 2A, 2B). Inclusions varied in size between 1.4 and 2.8 μm. In 3 of the 12 bearded dragons (nos. 4, 7, and 12), inclusions were present in approximately 40–50% of the mature and immature red blood cells. The majority of affected erythrocytes contained a single inclusion, although 2 inclusions were occasionally observed in the same cell. The hematocrit of lizard no. 7 decreased progressively from 26% to 9% over a 2-month period (ref. interval: 17–43% 7 ). The hematocrit of the other bearded dragons also decreased over 2 months, but remained within the reference interval during the same period. Complete blood cell count results are summarized in Table 3.
Complete blood cell count results of central bearded dragons (Pogona vitticeps) infected with an erythrocytic iridovirus.*
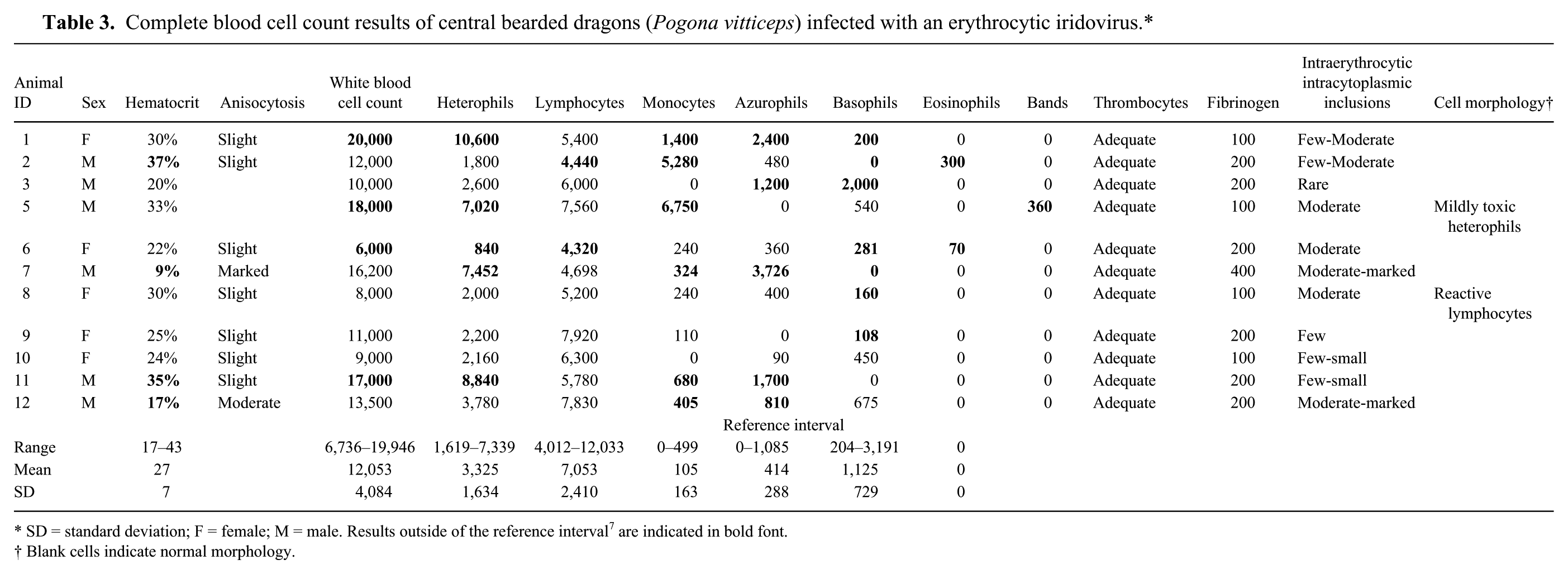
SD = standard deviation; F = female; M = male. Results outside of the reference interval 7 are indicated in bold font.
Blank cells indicate normal morphology.
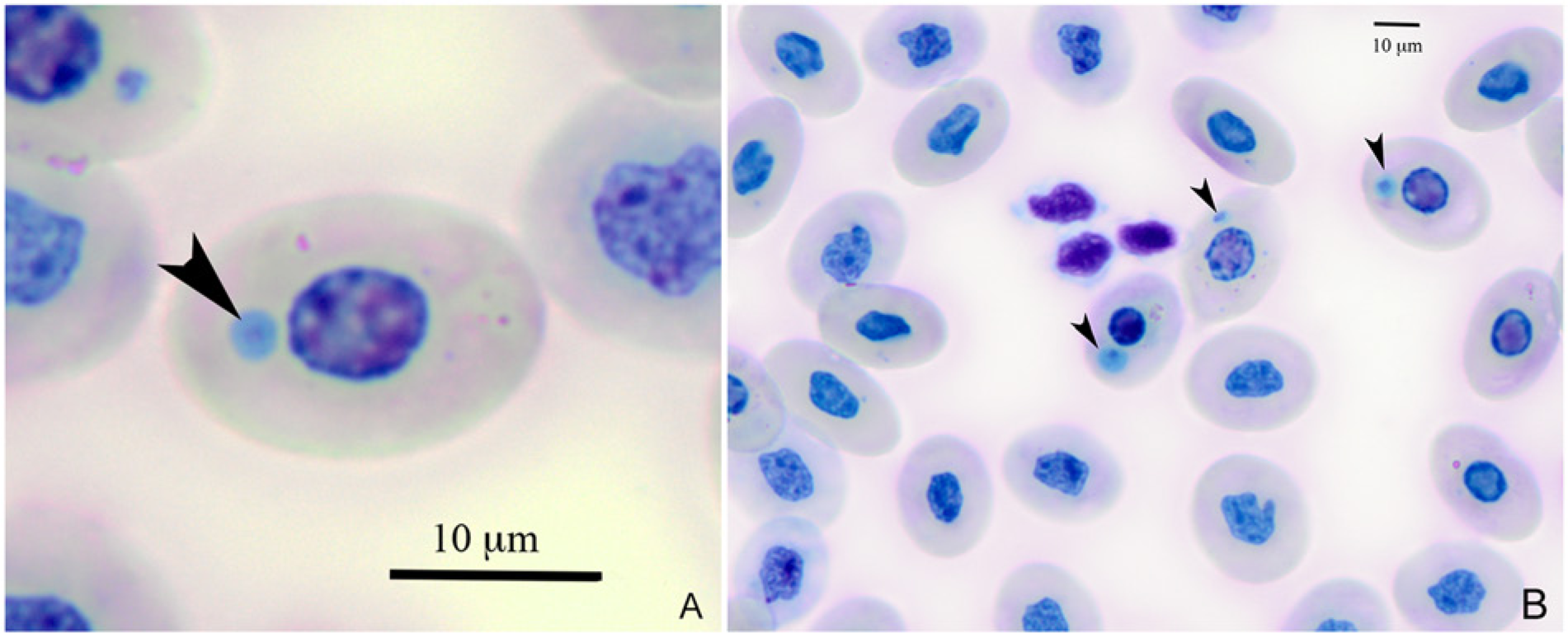
Blue-stained intraerythrocytic inclusion (arrowheads) observed with light microscopy from a central bearded dragon (Pogona vitticeps; no. 8).
Transmission electron microscopy
Intracytoplasmic viral inclusions were observed using transmission electron microscopic examination in the single individual whose blood sample was submitted (no. 7). Specifically, the intracytoplasmic inclusions were composed of enveloped icosahedral viral particles with a size of 141–193 nm (Fig. 3), which was consistent with an iridovirus. 13
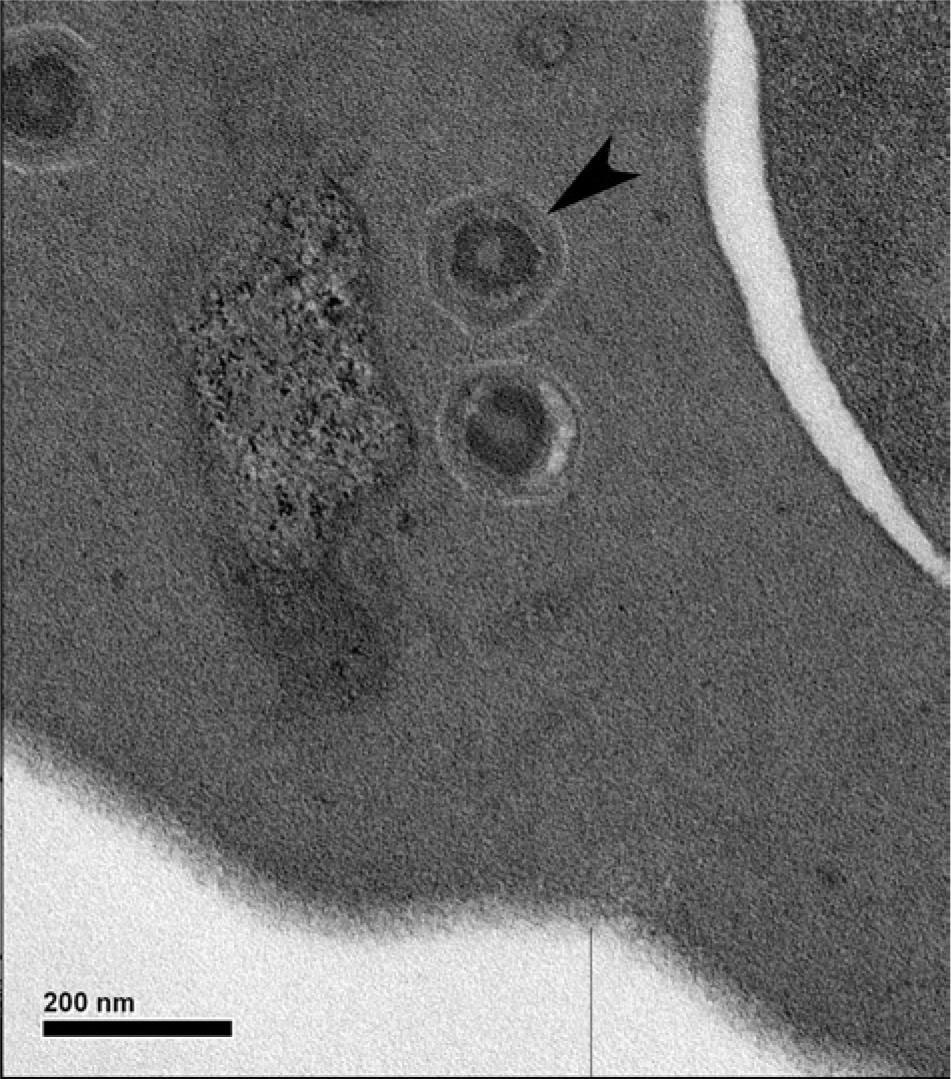
Viral particle (arrowhead) in a central bearded dragon (Pogona vitticeps; no. 7) by transmission electron microscopy. Bar = 200 nm.
Gross and histologic examination
Erythrocytic intracytoplasmic inclusions were noted in all 12 lizards. In 3 lizards (nos. 4, 7, and 12) with large numbers of inclusions detected on hematological examination, findings also included a moderate, multifocal, lymphoplasmacytic (nos. 4 and 7) or mononuclear (no. 12) glossitis, and a mild to moderate, multifocal, lymphohistiocytic hepatitis in 2 animals (nos. 4 and 5). Additional lesions were as follows: a severe, chronic, focally extensive, ulcerative, heterophilic enteritis with mixed intraluminal bacteria (no. 7); a mild to moderate, mononuclear enteritis (nos. 3, 5, 6, and 8–11); a mild, multifocal, lymphohistiocytic, esophagitis (nos. 1, 2, 6, and 9); and a mild, multifocal, lymphoplasmacytic, interstitial nephritis (nos. 7 and 9). Lymphoplasmacytic blepharitis was observed in 2 cases, which had previously presented with blepharospasm (nos. 4 and 11). Whether these lesions are associated with iridoviral infection is undetermined. Other lesions were found in a single individual and thought to be likely unrelated to the virus. Histologic results are summarized in Table 1. Inclusions were only observed in the red blood cells, and there was no definitive microscopic evidence of an extraerythrocytic viral infection.
Viral culture, PCR, and sequencing
Cultures in EPC cell line, BF-2, and in viper heart cells did not exhibit cytopathic effect. Generic bacterial PCR was positive for Delftia sp., which is considered a probable contaminant. The consensus Ranavirus PCR 24 was negative for all samples including blood, skin, liver, spleen, heart, and lung. Large DNA virus PCR did not generate any virus-specific product. The prey items were not tested for iridovirus presence.
The pan-erythrocytic iridoviral heminested PCR amplifications resulted in identical products of 439 base pairs after primers were edited out. A BLASTX search (http://blast.ncbi.nlm.nih.gov/Blast.cgi) of the database at GenBank found 60% homology with both TsEIV and LmEIV DNA polymerases. The sequence was submitted to GenBank under accession number KF767871.
Phylogenetic analysis
The Bayesian tree is shown in Figure 4. Bayesian phylogenetic analysis found that the WAG model of amino acid substitution was most probable with a posterior probability of 1.000, and this was used for ML analysis. 46 Bootstrap values as percentages from ML analysis are shown on the Bayesian tree (Fig. 4). There was a 96% Bayesian posterior probability value and a 64% ML bootstrap value supporting this virus clustering with the other squamate erythrocytic iridoviruses. The genetic distance seen between this and other characterized iridoviruses is consistent with placement of this virus as a novel species, and is hereafter referred to as Pogona vitticeps erythrocytic iridovirus (PvEIV).
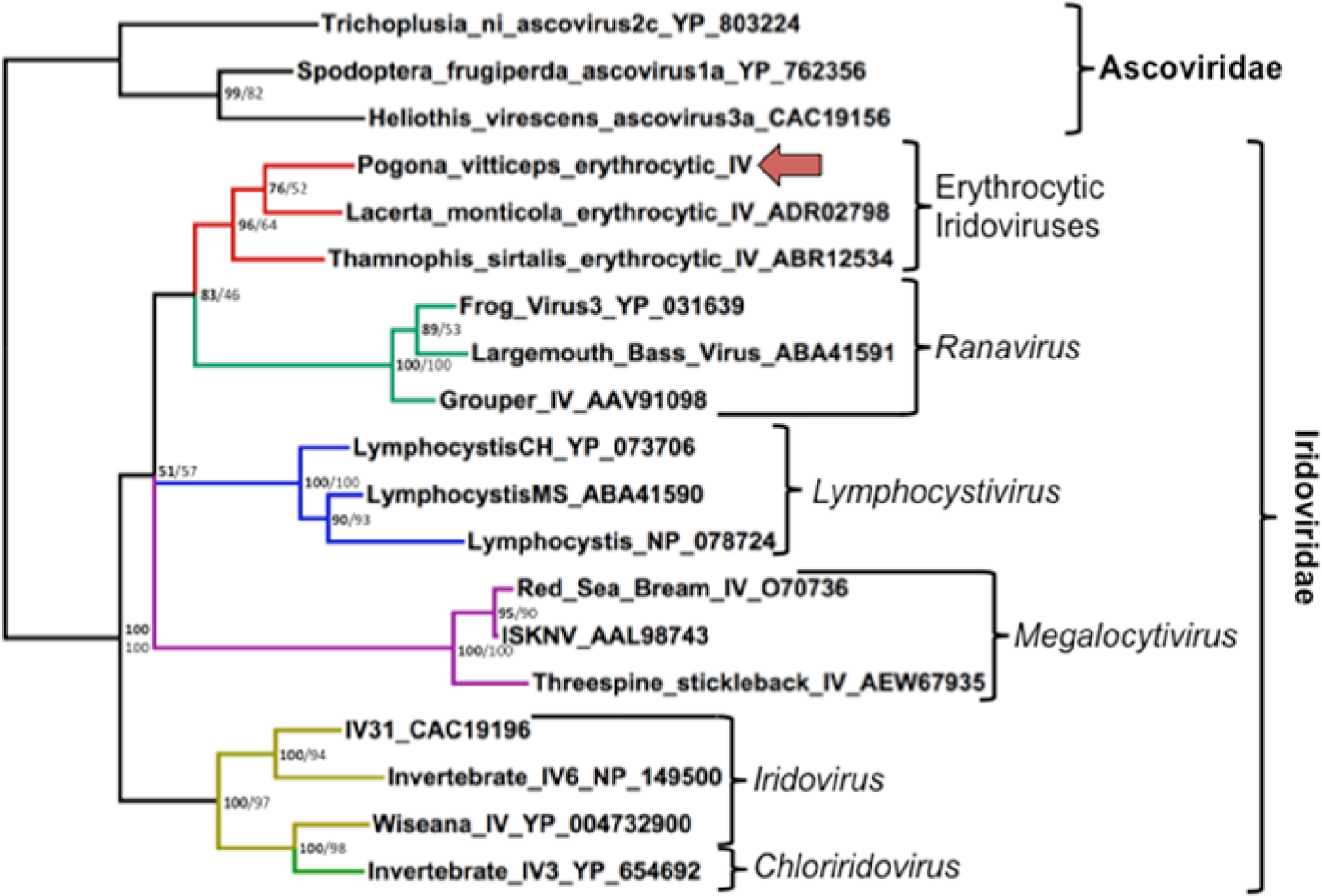
Bayesian phylogenetic tree of predicted amino acid sequences of iridoviral DNA-dependent DNA polymerase sequences based on MAFFT alignment. Bayesian posterior probabilities of clusters as percentages are in bold, and maximum likelihood bootstrap values for clusters based on 450 resamplings are given to the right. Trichoplusia ni Ascovirus was used as an outgroup. Pogona vitticeps erythrocytic iridovirus is indicated with an arrow. Brackets demarcate genera. Accession numbers of sequences retrieved from GenBank are given after the name.
Discussion
A novel erythrocytic iridovirus, PvEIV, was described in the current study in a colony of bearded dragons. Initially, the differential diagnoses for the intraerythrocytic inclusions included viral inclusions, parasites, bacteria, hemoglobin crystals, or staining artifacts. A combination of electron microscopy and molecular techniques confirmed the diagnosis of an iridoviral infection.
Iridoviruses are icosahedral double-stranded DNA viruses,13,20 measuring 120–300 nm in diameter. 39 The iridoviruses can occur as enveloped or nonenveloped forms, 13 although the envelope is not necessary for infection, thus rendering them more environmentally persistent than other enveloped viruses.13,28,41 For example, ranavirus isolates from tortoises have been shown to be more persistent in the environment than tortoise herpesviruses and picornaviruses, likely because their envelope is not required for infectivity and because double-stranded DNA is more stable than single-stranded RNA found in picornaviruses. 28
Family Iridoviridae is divided into 5 genera: Iridovirus, Chloriridovirus, 28 Ranavirus, Megalocytivirus, and Lymphocystivirus. 40 Members of the genera Iridovirus and Chloriridovirus are primarily known from arthropod hosts. Similar to previous analyses, the current study found that Chloriridovirus and Iridovirus do not segregate, making the genus Iridovirus paraphyletic. 48 Members of the genus Iridovirus found in crickets have also been reported in multiple tissues of several lizard species, including bearded dragons.20,22
Iridoviruses of the genera Lymphocystivirus and Megalocytivirus are only known to infect teleosts, 40 whereas members of the genus Ranavirus can infect diverse poikilothermic vertebrates (e.g., teleosts, frogs, turtles,15,45 and squamates12,42). Interestingly, a Ranavirus sp. described in a gecko (Uroplatus fimbriatus) was also associated with granulomatous glossitis and hepatitis, 23 similar to the findings in the bearded dragons in the present study. The relationship of iridoviral infection to the described histological changes was uncertain both in the previous report in a gecko and in the present case. 42 Iridoviruses have not been reported to naturally infect homoeothermic species, although this appears to be primarily a limitation of temperature; the viruses can infect avian and mammalian cells incubated at lower temperatures.9,47
When considering viral phylogeny, it is also important to consider host phylogeny. The geckos represent the earliest divergence of the extant squamates with reported EIVs, followed by skinks and cordylids, and then lacertids, which all represent more basal divergences within the squamates. The iguanids, snakes, varanids, chameleons, and agamids form a large monophyletic clade known as the Toxicofera, which includes nearly 60% of extant squamate species. 38 It has been a common fallacy that snakes are distinct from lizards, when there is not even a particularly basal divergence. The nomenclature that has been used in the literature of “Lizard erythrocytic virus” and “Snake erythrocytic virus” is therefore incorrect nomenclature based on genetic evolution. Reptile EIVs have been grouped as unclassified iridoviruses, and analysis in the current study supports this classification.1,27,42,44 Sequence information has previously been available from TsEIV and LmEIV.1,27,42,44 Snakes and agamids are both members of the Toxicofera, whereas lacertids are not. The phylogenetic analysis in the present study found weak support (76% posterior probability, 52% ML bootstrap value) that PvEIV clusters with LmEIV, suggesting that the branching order of these viruses differs from that of their host, consistent with a host-jumping event. This is not unexpected, as DNA viruses that replicate in the cytoplasm, such as iridoviruses, are associated with an increased ability to switch hosts. 27 Members of the genus Ranavirus, which may be a sister group to the EIVs, are capable of infecting hosts in diverse vertebrate classes. 15
Reptile EIVs have been associated with intracytoplasmic inclusions in erythrocytes of infected animals, polychromasia, and severely hypochromic erythrocytes, consistent with the present report in bearded dragons. 27 Reptile EIVs have been reported in diverse squamate species.18,36,37 Similarly in amphibians, Frog erythrocytic virus, which causes intraerythrocytic intracytoplasmic inclusions, is an iridovirus that does not belong to the genus Ranavirus.5,10,32 Frog erythrocytic virus has been shown to measure 293–312 nm 32 ; this is larger than PvEIV, which measures 141–193 nm. However, the frog EIV has not been sequenced or isolated in cell culture. 16 The mechanism of cellular death in cells infected with EIVs has not been definitively determined. 10 Increased osmotic fragility of infected erythrocytes and lytic viral infection have been demonstrated in some fish and amphibian infections, and has also been suspected in reptiles. 3 However, in other EIV infections, osmotic fragility was not observed. 10 Immune-mediated phagocytosis of infected cells has also been demonstrated in EIV infection in reptiles. 3
Severe anemia as well as infection of other cell types including leukocytes, hepatocytes, and endothelial cells may occur as demonstrated in LmEIV infection trials in lizards. 3 In the dragons in the current study, inclusion bodies were only detected in erythrocytes.
To the authors’ knowledge, reptile erythrocytic viruses have never been cultured in vitro.1,44 Therefore, the negative culture result in the current study is not surprising. In this context, development of a bearded dragon erythroid cell line may be useful for future culture attempts. It cannot be ruled out that the drugs used for sedation and euthanasia (pentobarbital and midazolam) may have interfered with viral culture, as previously described for a viral culture obtained from a human patient. 14
Not surprisingly, PvEIV was not amplified by PCR with primers targeting the ranaviral major capsid protein.6,44 The phylogenetic analysis in the present study clearly demonstrates that EIVs are distinct from ranaviruses. Although primers targeting a partial sequence of the DNA-dependent DNA polymerase gene of large DNA viruses have previously been used to amplify TsEIV from a peninsula ribbon snake, the primers were unsuccessful for amplification of PvEIV. 44 The sequence of the PCR product was clearly a noncoding sequence and did not represent a viral polymerase. The EIV-specific primers reported herein successfully amplified PvEIV and may be useful for further EIV discovery efforts.
Biochemical abnormalities varied depending on the lizard. Hypercholesterolemia, detected in 9 bearded dragons, and mild hyperglycemia (390 mg/dl; ref. interval: 218–356 35 ) in 1 bearded dragon (no. 7) could have been due to postprandial samples and reproductive activity in 4 out of the 5 females (nos. 1, 6, 8, and 9), as confirmed by postmortem examination of their reproductive tract.
During the acclimation period, an ectoparasitic mite infection was diagnosed and treated; no evidence of active mite infection was identified during postmortem examination. Hematophagous vectors may play a role in the virus transmission, as has been suspected in reports of other iridoviral infections.3,10 Although this hypothesis requires further investigation, infected erythrocytes could be transmitted by hematophagous vectors. Erythrocytic inclusions are also seen with rhabdoviral infections of squamates, and these viruses have been isolated from hematophagous insects as well as squamates. 43 A similar transmission mode should be considered for PvEIV. A possible role for insects or leeches in mechanical transmission of iridoviruses has been suggested, although infection by ingestion is also possible. 24
The current report describes a novel erythrocytic iridovirus associated with intracytoplasmic erythrocytic inclusion bodies in bearded dragons in California. The inclusions were associated with marked anemia in 1 out of 12 bearded dragons. Infected animals showed nonspecific clinical signs and death in 1 animal. It is unknown whether this newly described iridovirus originated from another reptile or amphibian species. Further studies are needed to fulfill Koch’s postulates for PvEIV in bearded dragons, and to elucidate the origin, mode of transmission, and host range of the virus.
Footnotes
Acknowledgements
The authors thank Dr. Foley’s laboratory team, Dr. Matthew Allender and Debbie Cassout, from the University of Illinois for providing the reptile cell line; Dr. Patricia Conrad for her evaluation of the intraerythrocytic inclusions; Grete Adamson, Patricia Kysar, and Emma Lee from the Electron Microscopy Laboratory, Department of Medical Pathology and Laboratory Medicine, School of Medicine, University of California at Davis (UC Davis); Dr. David Sanchez-Migallon Guzman for his contribution to this case; the technicians of the Companion Avian and Exotic Pet Medicine Department of UC Davis for logistical assistance; and Robin Houston from the Veterinary Medical Teaching Hospital Parasitology Department at UC Davis.
a.
Ivomec (10 mg/ml solution), Merial Ltd., Duluth, GA.
b.
Desert 50 UVB fluorescent bulb, Zilla 20 W; Zilla, Franklin, WI.
c.
Zilla 50 W, 120 V; Zilla, Franklin, WI.
d.
Fluker’s High Calcium Cricket Diet, Fluker Farm, Port-Allen, LA.
e.
Roche Cobas C501 Chemistry Analyzer, Roche Diagnostics, Indianapolis, IN.
f.
Leica Ultracut UCT, Leica, Vienna, Austria.
g.
Transmission microscope: Philips CM120 Biotwin Lens, FEI Company, Hillsboro, OR, made in Eindhoven, The Netherlands.
h.
Midazolam, Novaplus, Irving, TX.
i.
Pentasol powder, Virbac Animal Health, Fort Worth, TX.
j.
University of Zagreb, Croatia.
k.
American Type Culture Collection, Manassas, VA.
l.
Viper heart cells; kindly provided by Debbie Cassout, University of Illinois, Veterinary Diagnostic Laboratory, Urbana, IL.
m.
QIAquick, Qiagen Inc., Valencia, CA.
n.
Platinum Taq DNA polymerase, Invitrogen Corp., Carlsbad, CA.
o.
ABI 3130, Applied Biosystems Inc., Foster City, CA.
p.
Ofloxacin (0.3%), Bausch and Lomb Pharmaceuticals, Irvine, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
