Abstract
Bovine viral diarrhea virus (BVDV) causes significant economic loss in cattle. Detection of persistently infected (PI) animals is an important control measure, but persistence of maternal antibodies may result in false-negative test results. We assessed the sensitivity and specificity of 2 antigen ELISAs (Idexx BVDV Ag/Serum Plus and BVDV PI X2) and a reverse-transcription real-time PCR (RT-rtPCR; Idexx RealPCR BVDV) assay for detecting PI calves. Ear notch samples were collected from 1,030 calves ~3, 10, 24, and 38 d old (days 3, 10, 24, and 38). All day 38 samples were tested using 2 antigen ELISAs and RT-rtPCR, and any calf that tested positive by any of these tests was blood sampled at ~100 d old (day 100) for antigen and antibody testing by ELISA; samples collected on days 3, 10, and 24 were tested using the antigen ELISAs and PCR. Calves were defined as PI if they were test-positive on day 38 by either ELISA or PCR and were antigen-positive on day 100. Twenty-six calves were PCR BVDV test–positive and one was BVDV PI X2 ELISA–positive at day 38. Five calves were defined as PI, and all tested positive by ELISAs and RT-PCR assay on days 3, 10, and 24. The sensitivity and specificity were 100% for both antigen ELISAs and 96.7% and 100%, respectively, by RT-rtPCR. Test results were not affected by calf age, suggesting that testing for PI calves can be undertaken at any age.
Introduction
Bovine viral diarrhea virus (BVDV) infection causes significant morbidity and economic loss in dairy cattle.20,26 When non-immune dams are exposed to BVDV, their fetuses become infected, but the effect on the fetus depends on fetal age. Infection in the first trimester, before the fetus becomes immunocompetent, may result in calves becoming persistently infected (PI).16,20 These animals excrete virus continuously in large amounts in all body fluids. Hence, they act as a source of infection for naïve animals, and if they survive to adulthood and conceive, they will in turn produce a PI calf. The prevalence of PI calves or yearlings prior to commencement of eradication programs was 0.3% in Belgium, 31 0.5% in Germany, 35 and 0.6% in Ireland. 1 An important part of BVD control is to identify and remove PI animals. 17 Where BVDV is detected in a herd, testing of newborn calves to detect PI animals is an important control measure.1,27
Tests that have been evaluated for detecting PI calves include antigen ELISA, immunohistochemistry, reverse-transcription PCR (RT-PCR) assays, and virus isolation.2,9,13,29 Samples may include plasma, serum, skin biopsy, or swabs of nasal passages or other tissues. False-negative antigen test results may occur when maternal antibodies to BVDV are present in calves.13,25 Maternal antibodies have been reported to have a half-life of 21 d 3 and to persist for up to 30 wk after birth.5,36 Following artificial challenge of dams with BVDV, all calves blood sampled within 30 min of calving and prior to colostrum intake were found to be positive on virus isolation, BVDV NS3 antigen ELISA, and RT-PCR testing. 36 When tested at 7 d old and following colostrum feeding, 21 of 25 calves were BVDV-negative, 15 of the 25 were antigen ELISA–negative; all 25 animals remained RT-PCR–positive. 36 Similarly, in colostrum-fed PI calves, antigen was undetectable in serum for 1–2 wk after colostrum feeding. 11 Therefore, maternal antibodies may interfere with testing for antigen in serum using ELISA in calves. Some authors recommend that the minimum age for testing using the antigen ELISA should be 10 or 12 wk old,19,36 and the current recommendation in New Zealand is that testing not be done before 35 d old.
Use of tissue samples collected by ear notch sampling reduces the likelihood of maternal antibody interference.14,18 In one study, 11 PI calves tested negative using an Erns-antigen ELISA on serum samples 1 and 2 d after colostrum intake, 7 of 11 were negative at 4 d, and all were positive by 35 d after colostrum intake. However, all were positive at all times using an Erns antigen ELISA on ear tissue samples. 18 In colostrum-fed PI calves, titers for the Erns antigen dropped by 10- to 100-fold in skin biopsies but were detectable in all samples at all times. 11 In the same study, the BVDV NS3 antigen was not detected in biopsy samples following colostrum feeding. 11
Calves may test positive by ELISA or immunohistochemistry at a young age but be test-negative at an older age. This is interpreted as evidence of being transiently infected (TI). 6 In a study of feedlot cattle, 2 of 88 calves that initially tested positive, based on antigen ELISA, were subsequently found to be test-negative to antigen ELISA, immunohistochemistry, and RT-PCR testing, demonstrating that these calves were likely TI. 9 It is generally presumed that PI animals are antibody-negative, whereas TI animals develop antibodies. However, PI animals may be antibody test–positive if they have ingested maternal antibodies or been exposed to a heterologous BVDV strain. 10 In one case, calves were found to have serum neutralization antibodies for BVDV-1 following vaccination but became infected with BVDV-2. 28 Hence repeat testing is required to differentiate TI from PI animals. TI animals have been reported to excrete virus for 4–15 d after infection, 8 therefore retesting should occur 3 or more weeks after an initial positive test to differentiate PI and TI calves. In New Zealand, the risk that a PI animal would be antibody-positive after maternal immunity has waned appears low, given that only BVDV-1 is present and only BVDV-1a and -1c killed vaccines are available. 24
This study was designed to assess the sensitivity (Se) and specificity (Sp) of ear notch samples analyzed using antigen ELISA and RT-PCR testing at 4 times after birth to detect calves subsequently definitively defined as PI with BVDV.
Materials and methods
This study was conducted with the prior approval of the animal ethics committee of AgResearch, Ruakura, Hamilton, New Zealand (application 14788). Calves were enrolled from 11 spring calving, pasture-based, dairy herds from which BVDV had been detected in bulk tank milk by PCR in spring 2018 (
All female calves born in spring 2019 following artificial insemination, and which were designated to be kept as replacement animals for the dairy herd, had 3 ear notch samples collected at weekly visits to each farm. Three samples were collected from each calf when they were ~3, 10, 24, and 38 d old (days 3, 10, 24, and 38, respectively).
Tissue samples were collected using commercial tissue-sampling pliers (Datamars) and were ~3 mm in diameter. One mL of a topical anesthetic and antiseptic solution containing 40.6 g/L lignocaine, 4.2 g/L bupivacaine, 24.8 g/L adrenaline, and 5.0 g/L cetrimide was applied to the ear following notching (Bayer). The pliers were rinsed in detergent between calves if dirty, and washed and rinsed in 70% methanol between farms. The identity of the calves was recorded against a code number on the sample vials, and the sample code was scanned into a purpose-built database (Access v.16; Microsoft), into which the herd and calf identity were entered.
Ear notch samples were stored at −20°C at the premises of Cognosco. At approximately weekly intervals, ear notches collected on day 38 were submitted for testing (Idexx Laboratories, Palmerston North, New Zealand). If one or more of the test results from day 38 reported insufficient tissue for testing, an attempt was made to locate the calf and re-collect ear notches. All previous samples (i.e., days 3, 10, and 24, where available) collected from calves that were test-positive at day 38 were analyzed by antigen ELISA and RT-rtPCR assay (see below). Additionally, earlier samples from calves that had not been sampled at day 38 (as a result of being missed, having died, or having been sold) were also submitted for antigen ELISA and RT-rtPCR assay.
Calves that tested positive by antigen ELISA or by RT-rtPCR assay at day 38 were also blood sampled, with samples collected from the jugular vein into an evacuated blood tube without anticoagulant (Becton Dickinson), at an average of 102 (SD: 13; range: 76–127) d old (day 100), for determination of antibodies to BVDV and BVDV antigen. This sample was collected on average 63 (SD: 12; range: 39–78) d after day 38. Blood samples were centrifuged (15 min, 1,500 ×
A short questionnaire was administered to each herd owner to obtain information on BVD management, including grazing, and subsequent testing, of animals off the farm, culling of PI animals, use of BVDV vaccines, and testing of bulls. Two of the farms were managed by one individual and therefore responses were obtained from 10 herd owners.
To assess colostrum quality and the efficacy of colostrum delivery, a 10-mL jugular vein blood sample was collected for assessment of serum IgG concentrations from 10–15 calves in each herd managed in the same way as those that were ear notched. Note that no calves were available to be sampled in one herd for IgG. Calves had a median age of 4 (interquartile interval: 2–6; range: 1–11) d at the time of sampling.
Laboratory procedures
The 3 ear notch samples collected from each calf were separately analyzed using 2 antigen ELISAs that detect the Erns protein (BVDV PI X2 ELISA, [PI X2 ELISA], and BVDV Ag/Serum Plus ELISA [Ag Plus ELISA]; Idexx) and a quantitative RT-rtPCR assay (RealPCR BVDV; Idexx) following the manufacturer’s instructions. PCR was undertaken upon individual samples using the high confidence analysis mode on a LightCycler 480 instrument II system (Roche). The cycle threshold (Ct) cutoff was 40 cycles. The analytical sensitivity is ≤ 15 copies/reaction for BVDV-1 and -2.
The serum samples collected on day 100 were tested for the presence of BVDV antibodies (BVDV Total Ab ELISA, BVDV p80 Ab ELISA; Idexx) and BVDV antigen (PI X2, Ag Plus ELISAs). Results of the assays were recorded as test-positive or -negative based on the manufacturer’s recommended cut-points (Table 1). Where available, the numerical results (S/P, sample-to-negative control [S/N] ratio, or Ct) for each test were also recorded.
Summary of Idexx tests used for the detection of antigens or antibodies to BVDV in different samples (matrix), with the manufacturer’s recommended cut-points for test results.
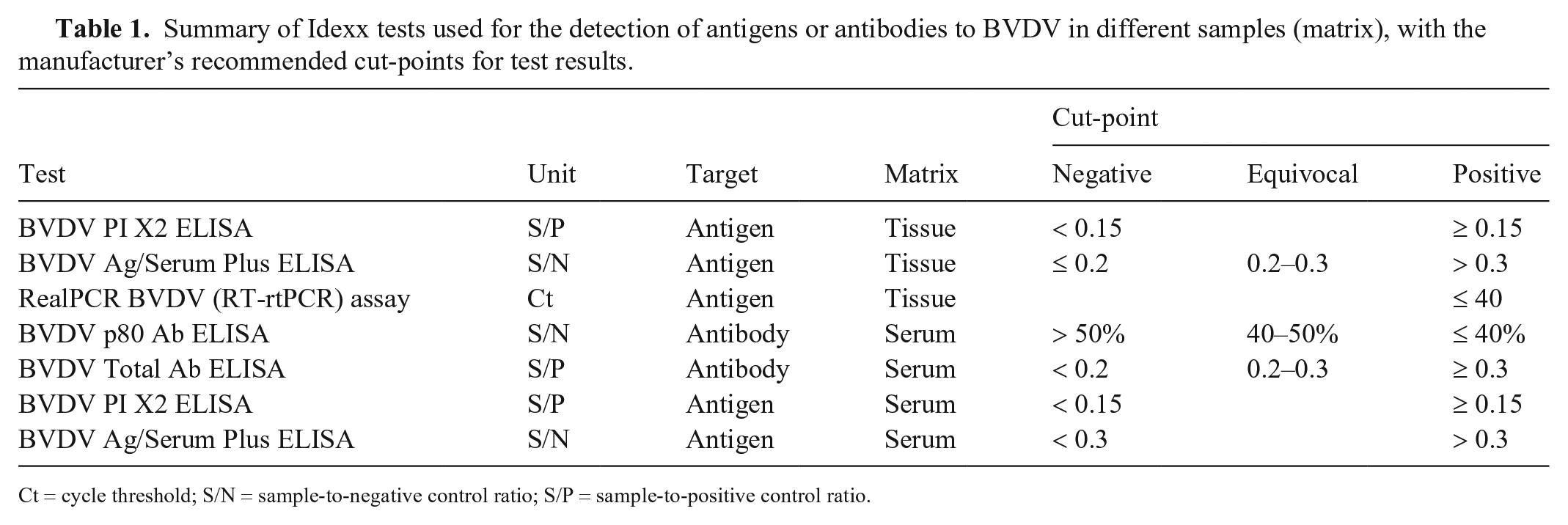
Ct = cycle threshold; S/N = sample-to-negative control ratio; S/P = sample-to-positive control ratio.
Concentrations of IgG in serum were determined using a turbidimetric immunoassay, which is based on the principle of immunologic agglutination and light scattering of the agglutination products. Tests were performed (AU680 analyzer; Beckman Coulter) using commercial reagents (Midland BioProducts). Failure of passive transfer (FPT) was defined as concentrations of IgG in serum < 10 g/L. 12
All testing was undertaken by Idexx Laboratories using the standard Idexx methodologies and quality assurance systems. Data were emailed in spreadsheets from the laboratory and loaded into the purpose-built Access database.
Data analysis
Individual calves were defined as PI, TI, or not infected (NI) based on all test data available for each animal. Animals were defined as PI if they tested positive in ear notch samples at day 38 on either ELISA or RT-rtPCR testing and tested positive for antigen in serum at day 100 on ELISA. Animals were defined as TI if they tested positive at day 38, by either ELISA or RT-rtPCR assay, and tested antigen-negative at day 100. Animals that tested negative at day 38 using both ELISA and RT-rtPCR assay were defined as NI, and no further testing was undertaken.
The Se of the day 3, 10, and 24 samples for detecting PI calves were calculated as the number of samples testing positive on each day divided by the total number of test-positive samples on day 38. The 95% confidence intervals (CIs) for the Se were calculated.
Numerical test results were examined using violin plots 15 in which the median, interquartile range, full range, and kernel density for the data were plotted, stratified by sampling day (days 3, 10, 24, and 38) and BVDV status of the calf.
The maximum of the sum of Se and Sp of the PI X2 and Ag Plus ELISAs and the RT-rtPCR assay, with PI status or not (TI and NI calves were combined for this analysis) as the categorical variable, were determined using receiver operator characteristic (ROC) curves. These were generated following linear regression, which accounted for the clustering of multiple samples within calf. For the RT-rtPCR testing, Ct values were only available for test-positive animals, therefore the ROC curve analysis of the RT-rtPCR assay data could only include the TI and PI animals.
Changes over time in the numerical values of the PI X2 and Ag Plus ELISAs were modeled using generalized linear mixed models with calf status (PI or TI), times (days 3, 10, 24, or 38), and the first-order interaction of status by time as fixed effects. Calf was included as a random effect. Estimated marginal means were calculated from the models and were compared using multiple comparisons adjusted by the Bonferroni correction factor. The Ct values from the RT-rtPCR were not normally distributed, and the use of generalized linear mixed models resulted in violations of assumptions of normal distribution. Hence, the Ct data were analyzed using nonparametric Kruskal–Wallis tests, with separate tests run to assess the effect of status (i.e., PI vs. TI), time (i.e., day 3, 10, 24, and 38), and time within status.
All statistical analyses were undertaken using STATA v.16.0 (StataCorp).
Power analysis
The main objective of the study was to determine the Se of testing for BVDV by Erns antigen ELISA or RT-rtPCR assay to detect calves that were PI with BVDV and potentially exposed to maternal antibodies for BVDV. Using an exact binomial CI, if 20 PI animals were detected at 38 d old, and all tested positive at the final test, then the 95% CI for Se would be 0.83–1. Assuming that we wanted 20 PI animals and that the within-herd prevalence of PI calves was ~2%, then 1,000 calves needed to be enrolled to achieve the required precision.
Results
Herd-level management of BVD and efficacy of colostrum delivery
All 10 herd owners provided information on their BVD management. Two herd owners reported using BVDV vaccination within the last 12 mo. Eight herd owners reported having found and removed a PI animal during the preceding lactation, with 6 having removed rising 2-y-old animals, 1 had removed an adult cow, and 1 had removed a calf. All 10 herd owners tested any purchased or leased bulls for BVDV. Eight herd owners grazed young replacement or other stock off the dairy farm, but none of them did any testing of the stock for BVDV when they returned to the farm. Of 145 calves sampled in 10 herds at 4 d old, 44 (30.3%) had concentrations of IgG in serum < 10 g/L, indicating FPT. The within-herd prevalence of FPT was 13–53% (Table 2).
Proportion (

Sampling and loss to follow-up
A total of 1,166 calves were sampled on day 3 (Tables 3 and 4). Some of these calves died, were sold, or not presented for sampling, therefore the number of calves sampled varied over time (Table 3). Of the calves sampled on day 3, 91 did not have a recorded birth date, 36 had recorded birth dates after the day of sampling, and 104 had birth dates > 7 d prior to initial sampling. It is likely that errors in animal identification, or recording or transcribing birth dates, occurred in some of these cases; for example, the animal must have been alive at the time it was sampled so having a birth date after sampling date is implausible. Note that calves that were > 7 d old (
Number of calves in 11 dairy herds that had ear notch samples collected 3, 10, 24, and 38 d after birth for BVDV antigen testing and had serum samples collected 100 d after birth for BVDV antigen and antibody testing. Note some calves were re-sampled after day 38 as a result of insufficient sample size at initial day 38 sampling.
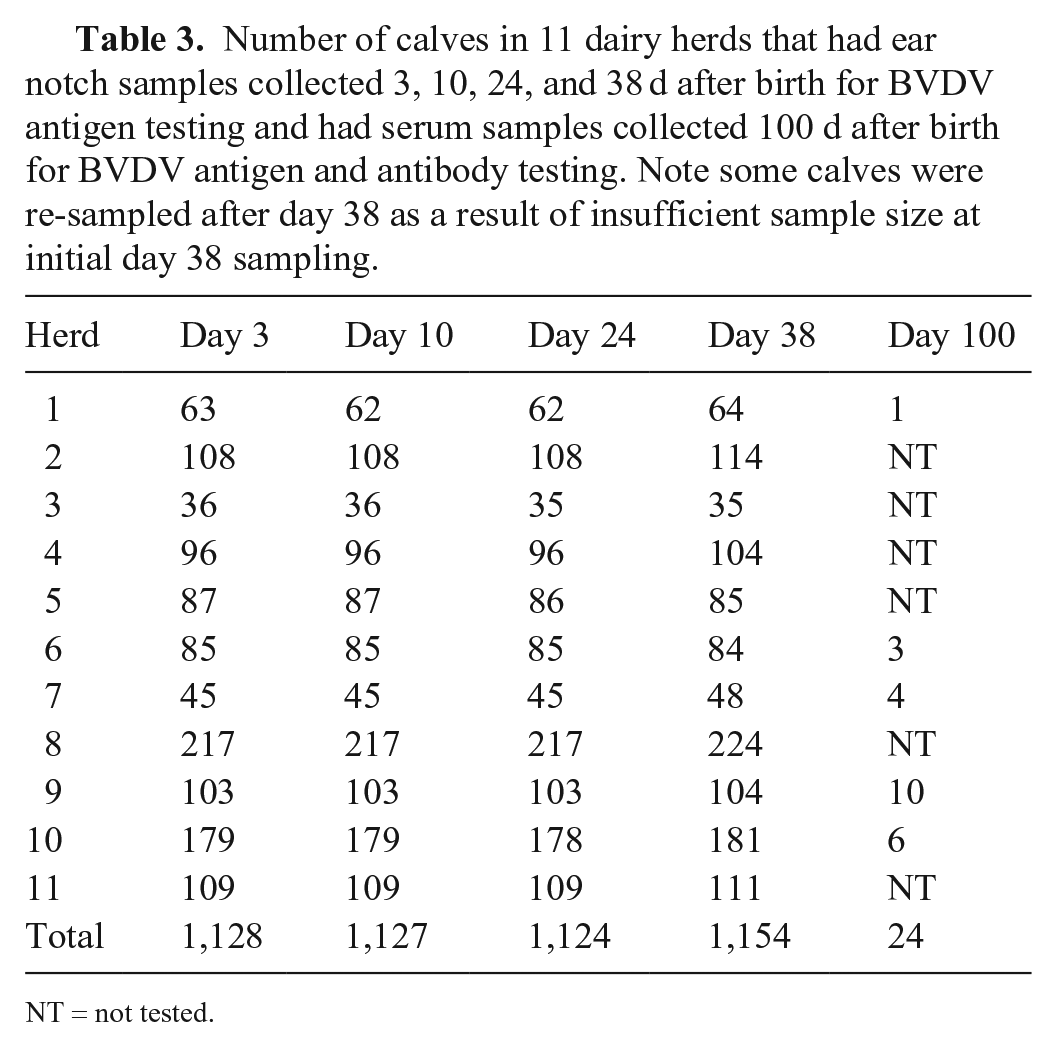
NT = not tested.
Average, standard deviation (SD), and minimum and maximum age of calves (d) at each sampling day. Note that birth dates were not available for some calves.
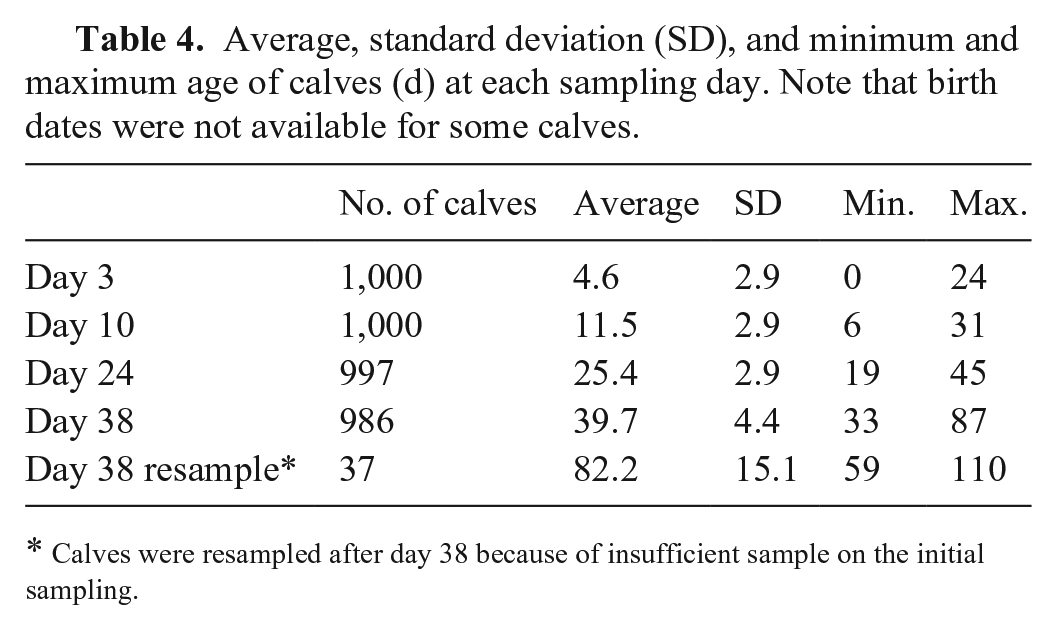
Calves were resampled after day 38 because of insufficient sample on the initial sampling.
Day 38 test results
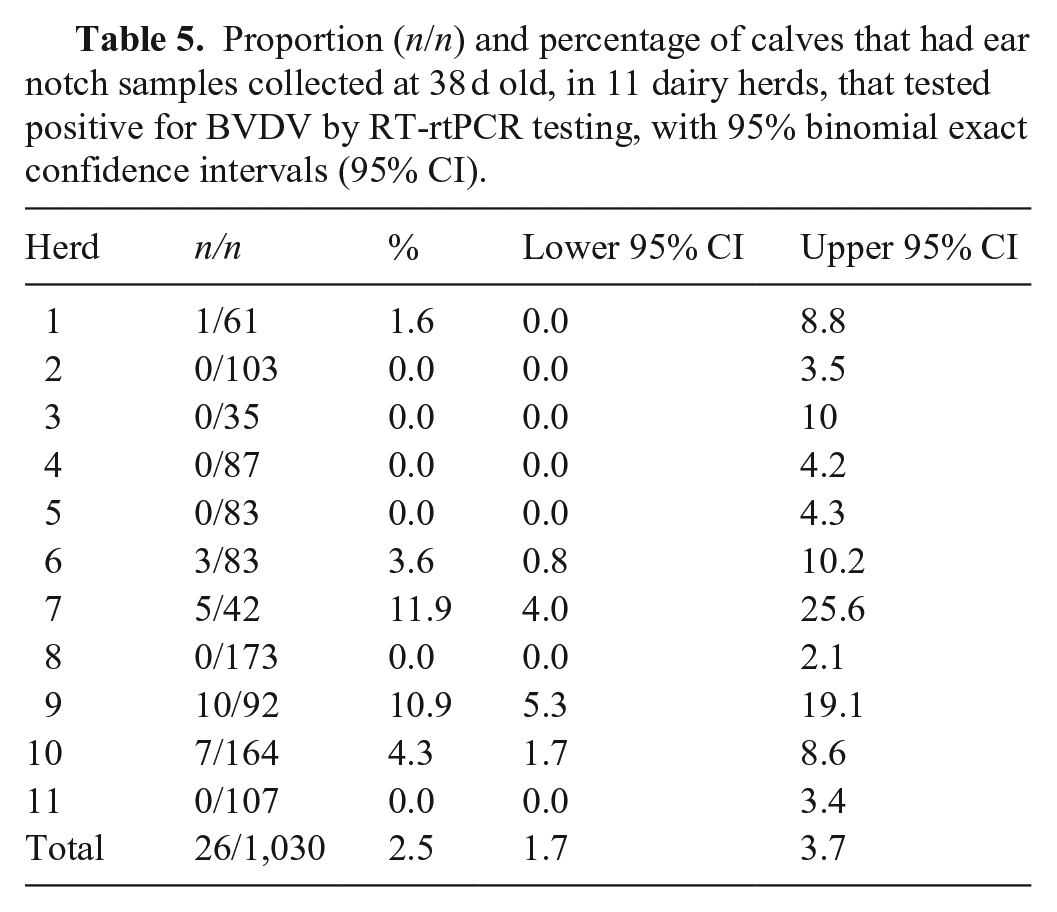
A total of 3,315 ear notch samples collected on day 38 were submitted for analysis; of these, 53 (1.6%) had insufficient tissue collected for testing. Of the 1,030 calves with test results on day 38, 26 (2.5%) were positive for BVDV by RT-rtPCR testing (Table 5). Of these, 6 of 26 (23%) and 5 of 26 (19%) tested positive with the PI X2 and Ag Plus ELISAs, respectively. One animal tested positive on the PI X2 ELISA that was negative with both the Ag Plus ELISA and RT-PCR assay, therefore 27 calves had positive test results on day 38. The prevalence of test-positive calves varied among herds, with 5 of 11 (45%) herds having 1 or more antigen ELISA–positive or RT-PCR test–positive calves at day 38 (Table 5).
Proportion (
PI and TI calves
Of the 27 calves that were test-positive on day 38, 5 (19%) were defined as PI, and 22 (81%) were defined as TI, based on assessment of the test results available (Table 6). Hence, of the total sample of calves assessed, 5 of 1,030 (0.5%) were PI. These animals came from 3 of the 11 farms. On 2 farms, only TI calves were located.
Test results for calves that tested positive for BVDV, using ear notch samples collected at 38 d old (day 38) by either RT-rtPCR testing or antigen ELISA (PI X2 or Ag Plus), and results of antigen testing of ear notch samples collected at ~3, 10, and 24 d old (days 3, 10, 24) and blood samples collected at ~100 d old (day 100) tested for BVDV antibodies (Ab) and antigen (Ag).
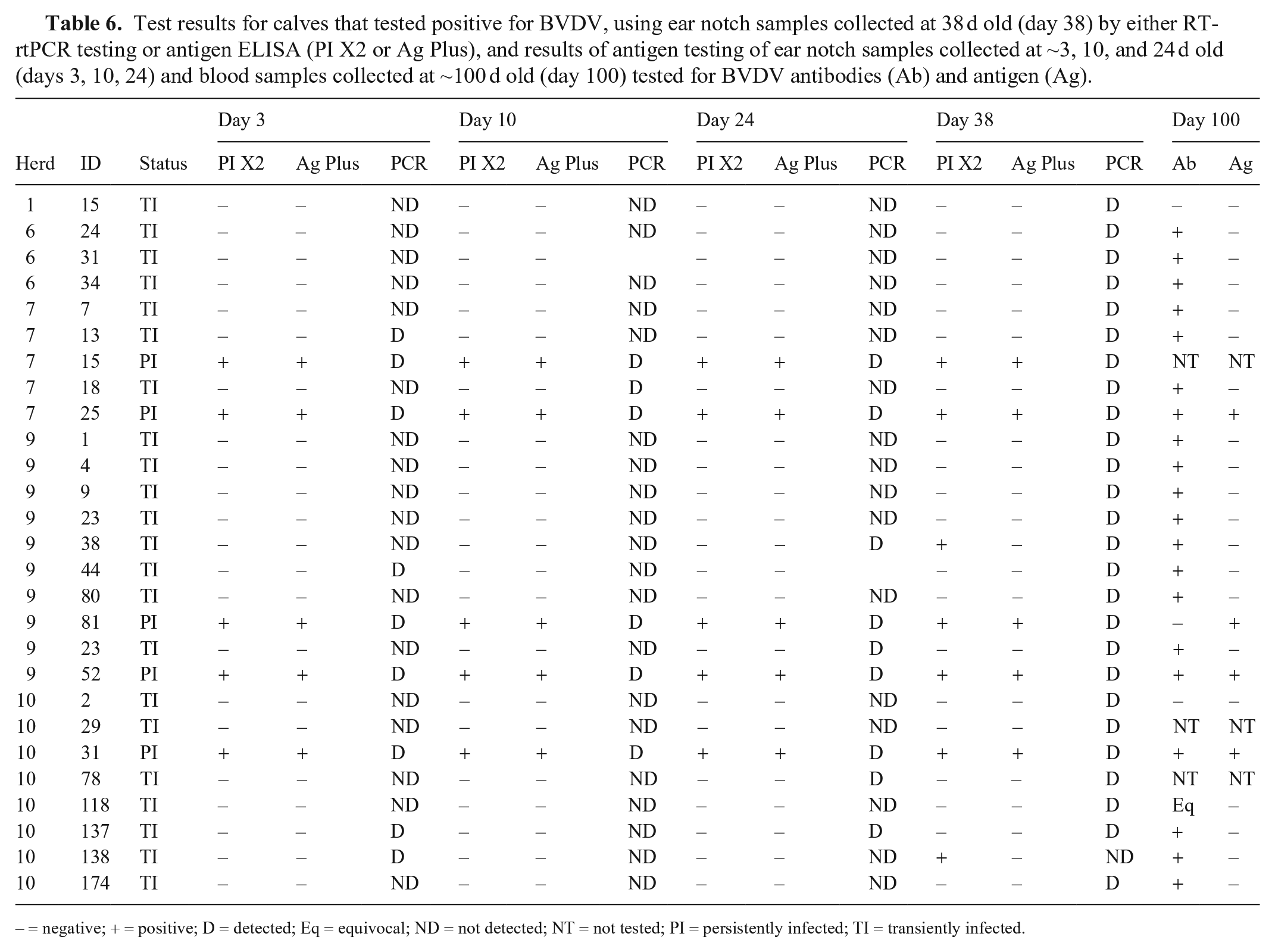
– = negative; + = positive; D = detected; Eq = equivocal; ND = not detected; NT = not tested; PI = persistently infected; TI = transiently infected.
Among the 5 animals defined as PI, all ear notch samples at all times tested positive on both the 2 antigen ELISAs and the RT-rtPCR assay. One calf was euthanized on-farm prior to final sampling with signs consistent with BVD. Among the other 4 PI calves, all tested positive for both serum antigen ELISAs on day 100, and 3 of the 4 also tested positive for antibody by both antibody ELISAs at this time.
Among the 22 animals defined as TI, none were antigen ELISA–positive at days 3, 10, or 24 on the ear notch samples, but 4, 1, and 4 were RT-rtPCR test–positive at days 3, 10, and 24, respectively. All were antigen-negative on serum ELISA at day 100, and 18 of 22 (82%) were positive or equivocal on antibody ELISA at day 100. Two were lost to follow-up, and 2 were antibody-negative at this time. The 2 TI calves that were both antibody- and antigen-negative at day 100 were RT-rtPCR test–positive only at day 38 and were negative at all other times.
PI X2 ELISA test characteristics
The S/P ratio for PI calves was higher than the TI and NI calves on days 3, 10, 24, and 38 for the PI X2 ELISA (Fig. 1A). Using a cut-point of S/P > 0.18, the PI X2 ELISA had a Se of 100% (95% CI: 83.2–100%) and Sp of 100% (95% CI: 99.7–100%; Fig. 1B). The laboratory-defined positive cut-point for this assay is S/P ≥ 0.15, which if used would have resulted in 2 false-positive results for the calves with an S/P of 0.17. The marginal mean S/P was lower for TI (−0.02 [SE 0.05]) calves than for PI (9.02 [SE 0.33]) calves (
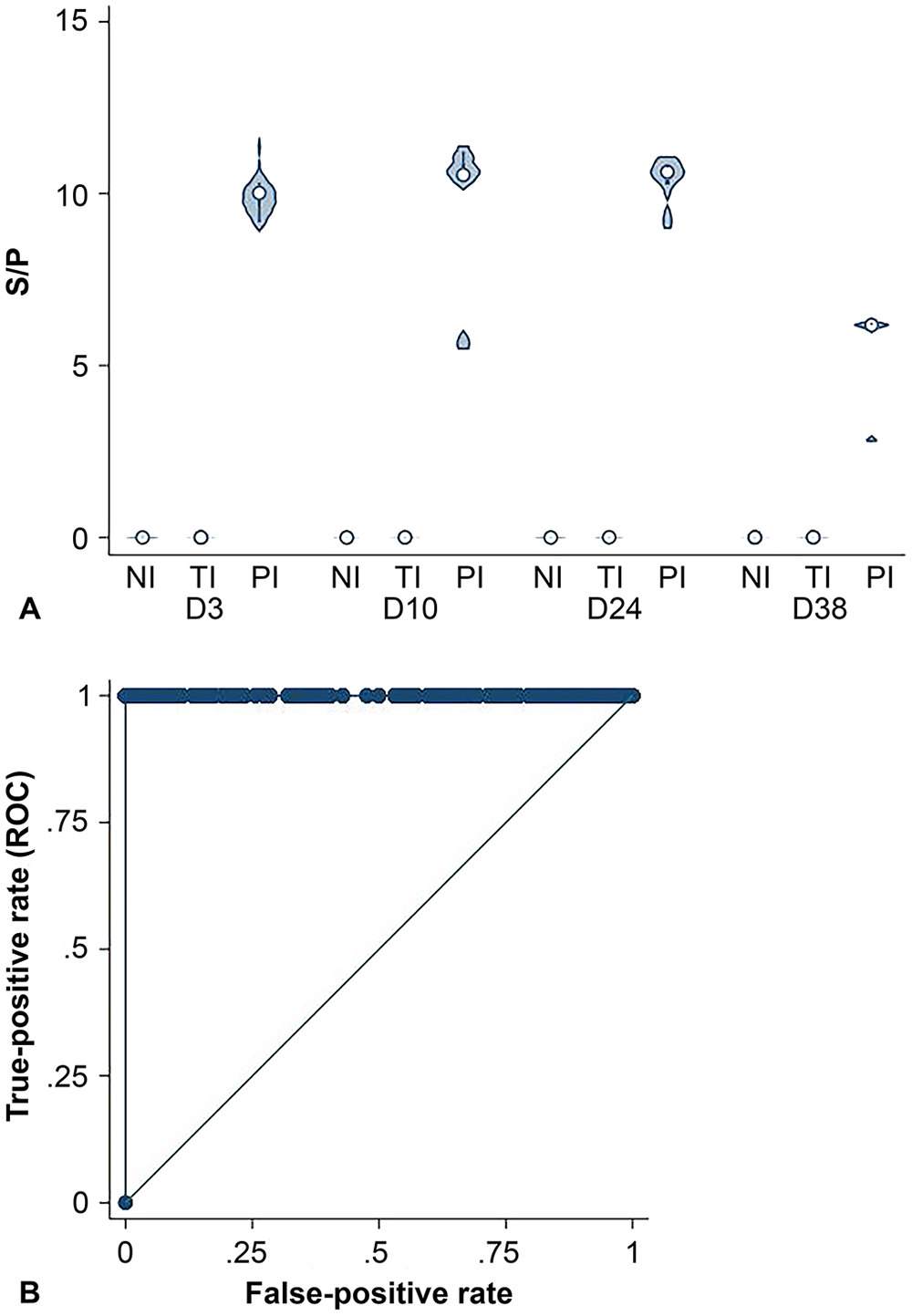
BVDV PI X2 ELISA.
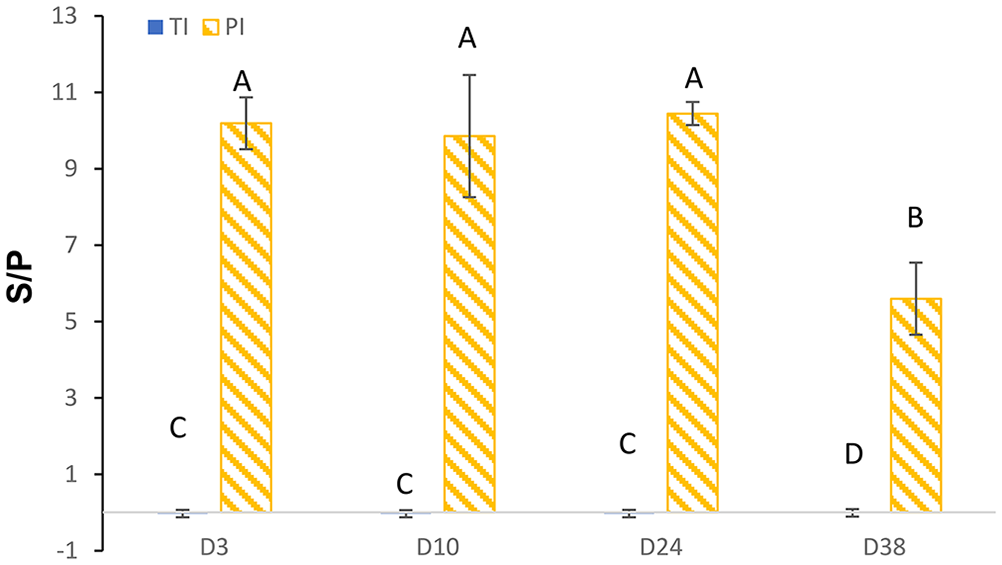
Estimated marginal mean (±95% confidence intervals) sample-to-positive control (S/P) ratio results for the BVDV PI X2 antigen ELISA for calves defined as transiently infected (TI;
Ag Plus ELISA characteristics
The S/N was higher for PI than TI and NI calves on days 3, 10, 24, and 38 for the Ag Plus ELISA (Fig. 3A). Using the laboratory-defined positive cut-point of S/N > 0.3, the Ag Plus ELISA had a Se of 100% (95% CI: 83.2–100%) and Sp of 100% (95% CI: 99.7–100%; Fig 3B). The marginal mean S/N was lower for TI (0.04 [SE = 0.02]) than PI (2.20 [SE = 0.17]) calves (
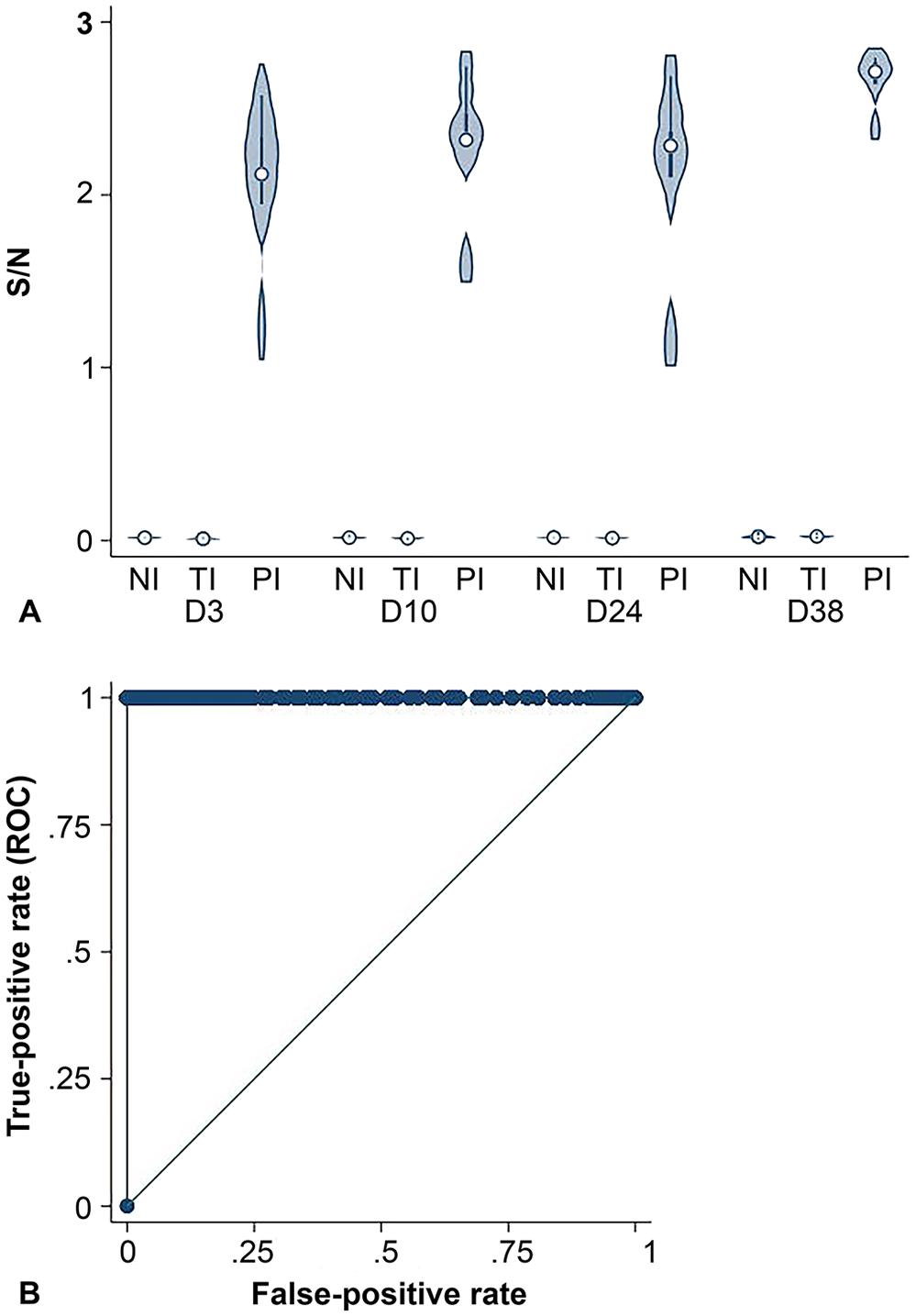
BVDV Ag/Serum Plus ELISA.
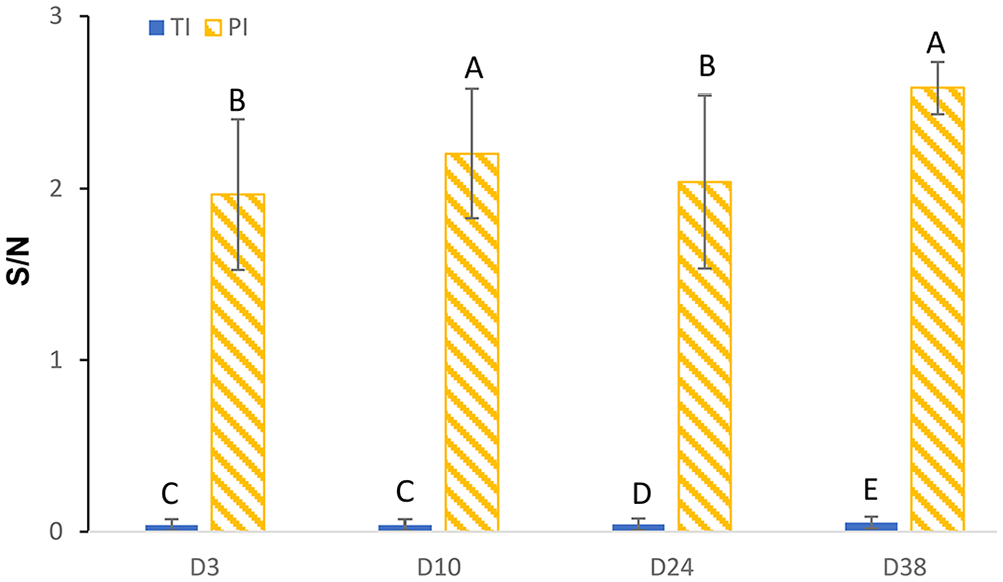
Estimated marginal mean (±95% confidence intervals) sample-to-negative control (S/N) ratio results for the BVDV Ag/Serum Plus ELISA for calves defined as transiently infected (TI;
RT-rtPCR assay characteristics
The Ct for PI calves was lower than for TI calves on days 3, 10, 24, and 38 for the RT-rtPCR assay (Fig. 5A). The area under the ROC curve was 0.999 (95% CI: 0.997–1.000). Using a positive cut-point of Ct ≤ 33.6, the RT-rtPCR assay had a Se of 100% (95% CI: 83.2–100%) and Sp of 99.9% (95% CI: 99.5–100.0%; Fig. 5B). When a cut-point of Ct ≤ 32.8 was used, the assay had a Se of 95% (95% CI: 75.1–99.9%) and Sp of 100% (95% CI: 99.7–100.0%). The Se and Sp were < 100% because one PI animal and one TI animal had Ct values of 32.8.
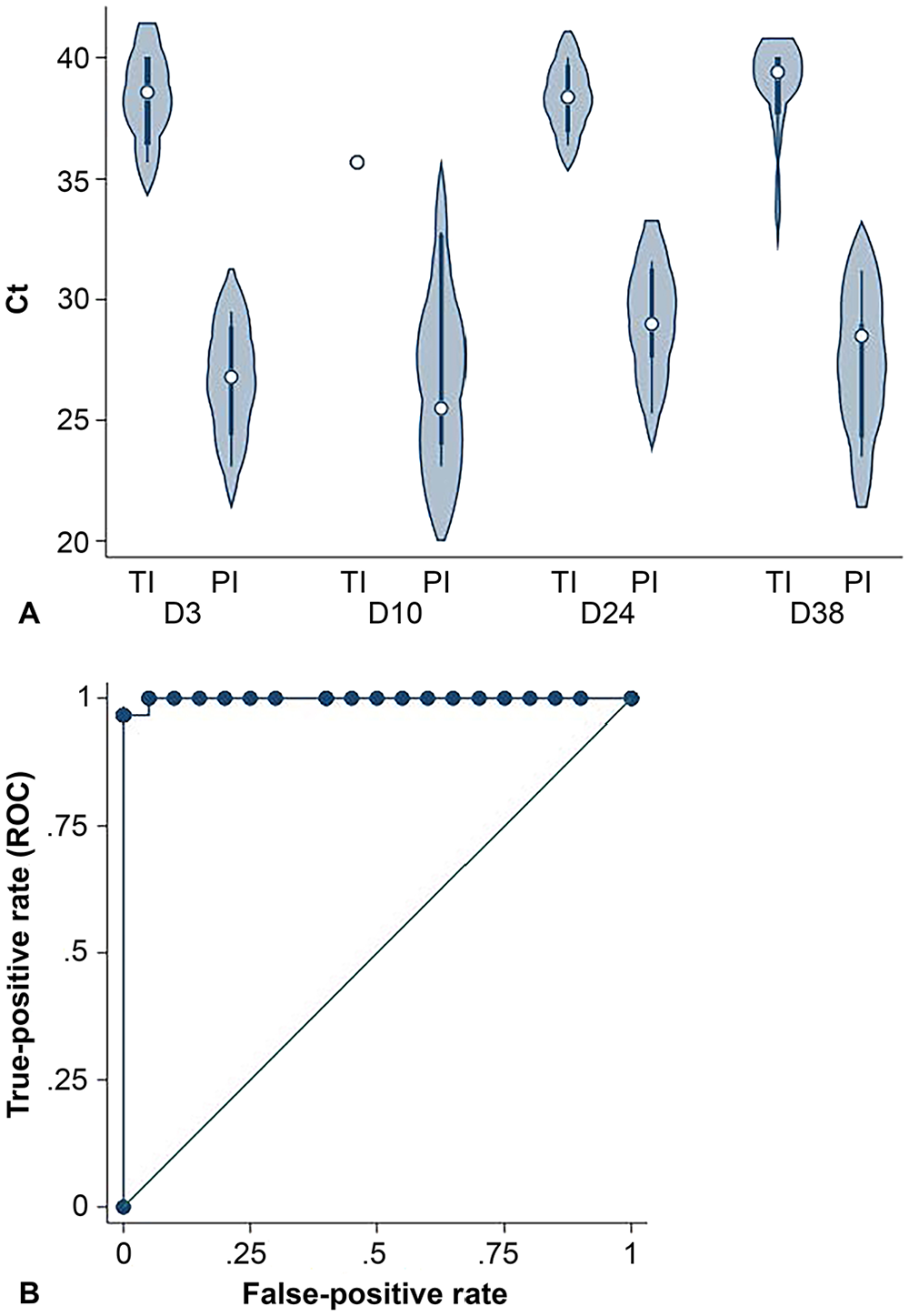
RealPCR BVDV (RT-rtPCR) assay.
The Ct values were lower for PI than TI animals (rank-sum = 210.5 vs. 1,064.5 for PI vs. TI Ct, respectively;
Antigen ELISAs and RT-rtPCR assay results from excluded calves
A total of 285 test results were available for analysis from 42 calves (from 8 herds) that were lost to follow-up because of incomplete initial sampling associated with lack of records (e.g., no birth date), illness, death, or failure to present the calves. All of these calves always tested negative with the Ag Plus and the PI X2 ELISAs. For one calf tested at 14 d old, the RT-rtPCR assay Ct value was 39.2. This calf tested negative to both the RT-rtPCR assay and antigen ELISA in 11 other tests (i.e., at all 4 sampling times). This animal likely had a false-positive RT-rtPCR test result or was TI. This animal was excluded given the lack of birth date records; no follow-up testing was undertaken.
Discussion
Calves were tested from dams in herds that likely had BVDV circulating during the breeding season when the calves were conceived. The prevalence of PI calves was relatively low (0.5%), and PI calves were only located in 3 of the 11 herds. Among the calves positive on either RT-rtPCR testing or antigen ELISA at day 38, > 80% were found to be antigen-negative and antibody-positive when tested at ~100 d old. This suggests that the majority of RT-rtPCR test–positive calves, when tested at 38 d old, were likely TI, rather than PI. Using a combination of the day 38 and day 100 test results to define animals as PI or TI, the Se and Sp of the antigen ELISA using ear notch samples were 100% irrespective of the age of the calves tested. The RT-rtPCR assay also had a Se of 100% and a Sp of 95% if a cut-point of Ct ≤ 32.8 was used. This suggests that the presence of maternal antibodies to BVDV did not interfere with the ability of the ELISA or PCR to successfully identify PI calves.
The current study has some limitations. The low prevalence of PI calves resulted in wide CIs around the Se and Sp estimates. Therefore, despite these estimates being close to 100% for all tests, the calculated 95% CIs reflect the small number of animals and hence samples available for the calculations. A second limitation is that, given the large number of calves to be sampled in a relatively short time, the study design involved weekly visits to farms. Hence, by chance, no test results are available for PI calves < 2 d old. Care should be taken extrapolating results to infer that the test characteristics would be the same for calves sampled on the day of birth or within 24 h of birth.
The low prevalence of PI calves detected in the current study, despite the presence of BVDV in herds during the breeding program, agrees with previous studies suggesting that only 1 or 2% of fetal infections result in PI animals.4,16 However, these estimates were most likely based on results from year-round calving herds in which only a small proportion of dams have fetuses that are between implantation and 120 d of gestation at any one time. In contrast, under New Zealand seasonal calving systems, most dams are at similar stages of gestation at any one time, and so potentially susceptible if BVDV is circulating during the breeding program. However, the proportion of susceptible dams may still be low, depending on the timing of the introduction of BVDV and the proportion of seropositive dams.
Sensitivity for detection of PI animals was consistently high in the current study, whatever the age of calves tested. Additionally, all 3 tests evaluated (RT-rtPCR assay, Ag Plus and PI X2 ELISAs) performed similarly. There was no evidence that age of the calf at testing affected the test results either for the antigen ELISAs or the RT-PCR assay. This contrasts with previous studies that have suggested that the presence of maternal antibodies may affect test results. One factor explaining these differences in interference is the target of the ELISA. In one study of 5 calves known to be PI, results of an ELISA targeted at the envelope glycoprotein Erns in skin biopsies were positive despite colostrum intake, whereas the NS3 antigen ELISA failed to detect antigen in biopsy samples, even prior to colostrum intake. 11 The authors hypothesized that the discrepancy among the antigen ELISAs could be the result of stability of the antigen, variation in Se of the test, or some effect of the target antigen. They report that storage of skin biopsy samples at temperatures higher than room temperature reduced the amount of detectable antigen and this was more noticeable for NS3 than the Erns antigen ELISA.
Concentrations of BVDV-specific antibodies in colostrum and absorption by calves may differ within and between studies. In the current study, 30% of calves sampled had FPT. This prevalence estimate is similar to that from a previous New Zealand study, which found 33% of calves sampled had FPT. 7 In that study and the current one, the prevalence of FPT varied widely between herds. In the current study, the within-herd prevalence of FPT was 13–53%. Hence the effect of maternal antibodies on test characteristics will likely vary both within herd, depending on whether individual calves have or have not received maternal antibodies against BVDV, and between herds, depending on the within-herd prevalence of FPT. However, within the small number of PI animals in the current study, there was no evidence of a consistent change in the S/P, S/N, or Ct values with increasing age of calves at sampling. It is likely that concentrations of maternal antibody would have declined across the time of testing. Thus, the impact of maternal antibodies on the test characteristics among the TI and PI calves appears to be minor. In previous studies, the effect of maternal antibodies on test results varied with age of calf. For example, antigen titers in skin biopsies were 1–2 logs lower at 1–5 d after birth relative to samples collected prior to intake of colostrum or those collected at a later age. 11 In the current study, the PI calves were first sampled at 2 (2 calves), 3, and 7 d old, and 1 animal had a missing birth date but was sampled at < 7 d old. Therefore, testing of animals with potentially higher intake of colostral BVDV antibodies, sampled within 24 h of colostrum intake, might result in a lower test Se than was observed in the current study.
More than 80% of calves that tested positive on RT-rtPCR testing at day 38 were subsequently defined as TI, following further testing. The majority of these animals consistently tested negative with the antigen ELISA at days 3, 10, 24, or 38. Thus, with the cut-point of Ct ≤ 40 used to screen calves at day 38 in the current study, RT-rtPCR is more sensitive to the presence of BVDV than is the antigen ELISA. There is a possibility that there were false-positive RT-rtPCR test results. Two of the TI animals were negative on serum ELISA antibody testing at day 100, and one had an equivocal result. All were antigen-negative in serum at this time, and all had tested negative using the antigen ELISA and RT-rtPCR assay at days 3, 10, and 24. The 2 antibody-negative calves were 90 and 107 d old at the time of testing, and the interval between the day 38 and day 100 test was 53 and 67 d, respectively. It seems surprising that antibodies had not developed over this time. The small number of negative and equivocal results does suggest that there is a possibility of false-positive RT-rtPCR test results when a cut-point of Ct < 40 is used, given that this may reflect a very low copy number.
Use of the antigen ELISA resulted in a higher Sp for identification of PI calves than the RT-rtPCR assay. However, Se for detection of TI calves was lower for the antigen ELISAs than for the RT-rtPCR assay given that most TI calves were not test-positive upon ELISA. Hence, if routine ear notch testing were undertaken using either of the antigen ELISAs alone, the great majority of the TI calves would not be identified. If the goal of calf testing is to identify PI, rather than TI, animals, then the antigen ELISAs may be better tests given that they achieved 100% Se and Sp for detection of PI, irrespective of the age of the calf tested. The effect of testing for and removing only PI animals compared with removing all TI and PI animals on the success of a BVD control program, and on animal productivity, remains to be determined.
Studies that have examined the transmission of BVDV among cohorts of calves in which only TI animals were present did not find any evidence of transmission of BVDV from TI animals to other calves.22,23,30 However, a case study suggested that there may have been persistence of BVDV in a herd over a 6-y period in the absence of identification of a PI animal. 21 Thus, although the risk of focusing on removal of PI animals only, and possibly leaving TI animals within the cohort, appears to be small, it may not be zero. Therefore, further modeling and observational studies assessing the impact of such a strategy need to be examined.
In determining the PI status of an animal, the positive predictive value of a single RT-rtPCR test at day 38 is relatively low because of the presence of TI animals. There may be a positive cost-benefit of an additional blood sample 3 or more wk after the initial ear notch sampling to enable retention of TI animals, on the assumption that they will be viremic for < 2 wk 8 and will consequently have minimal impact on the other calves. Conversely, the cost-benefit of removing all test-positive animals, whether PI or TI, remains to be calculated.
Three of the PI animals for which antibody tests at day 100 were available were positive on the antibody ELISA, despite also being antigen ELISA–positive. It is unclear whether this was a result of persistence of maternal antibodies, false-positive antibody ELISA results, or because of seroconversion to a heterologous BVDV strain. Maternal antibodies have been reported to persist for up to 30 wk after birth,5,36 making this the most plausible explanation for the presence of antibodies in PI animals at 100 d old. Interestingly, it has been reported that 5 PI calves tested prior to colostrum intake were test-positive for NS3-specific antibodies. 11 Those authors conjectured that there may be some cross-placental movement of antibodies. A study of German animal health laboratories did find some inconsistencies in antibody testing; however, most of the discrepancies were the result of false-negative rather than false-positive results. 34 Seroconversion of PI animals to heterologous BVDV strains has been reported. However New Zealand only has BVDV-1, with BVDV-1a and -1c reported.24,32,33 It is not clear whether seroconversion to a different type 1 virus can occur when an animal is PI with the other.
In no case was a loss to follow-up from the current study (i.e., incomplete sampling or lack of records) associated with a calf being PI. All the 42 calves lost to follow-up, for which at least some samples were available, always tested negative by all tests, other than one animal that had a weakly positive RT-rtPCR assay result at 10 d old. Thus, it appears unlikely that any loss to follow-up because of increased risk of morbidity or mortality associated with BVDV resulted in an underestimate of the prevalence of PI or TI calves in this study.
Footnotes
Acknowledgements
The cooperation of herd owners and staff for the multiple sampling time points of this study is greatly appreciated. The technician team of Cognosco (Elizabeth Blythe, Bev Brownlie, Cathy Yanez) did a great job of ensuring sampling was timely, samples were handled appropriately, and data were entered correctly and in a timely fashion. The laboratory team of Idexx Palmerston North is thanked for their attention to detail and timeliness of reporting.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by Idexx, New Zealand. Bayer New Zealand donated the topical anesthetic and antiseptic (Tri-solfen) applied after the ear notching. This study was not influenced in its design, interpretation, or reporting by either Idexx or Bayer.
