Abstract
Lawsonia intracellularis is an obligate intracellular bacterium associated with enteric disease in pigs. Clinical signs include weight loss, diarrhea, and, in some cases, sudden death. The hallmark lesion is the thickening of the intestinal mucosa caused by increased epithelial cell replication, known as proliferative enteropathy. The immune response to L. intracellularis is not well defined, and detection of the infection, especially in the early stages, is still a significant challenge. We review here the main approaches used to identify this important but poorly understood pathogen. Detection of L. intracellularis infection as the cause of clinical disease is confounded by the high prevalence of the pathogen in many countries and that several other pathogens can produce similar clinical signs. A single L. intracellularis–specific ELISA and several amplification assays are available commercially to aid detection and surveillance, although histopathology remains the primary way to reach a conclusive diagnosis. There are major gaps in our understanding of L. intracellularis pathogenesis, especially how the host responds to infection and the factors that drive infection toward different clinical outcomes. Knowledge of pathogenesis will increase the predictive value of antemortem tests to guide appropriate interventions, including identification and treatment of subclinically affected pigs in the early stages of disease, given that this important manifestation reduces pig productivity and contributes to the economic burden of L. intracellularis worldwide.
The obligate intracellular bacterium Lawsonia intracellularis has been associated with enteric disease in various animal species, but it is of greatest importance in pigs.17,18,40,42,72,77 In 1931, a clinical presentation in pigs, characterized by diarrhea, weight loss, and, in some cases, sudden death, was described for the first time. 7 In 1953, the disease was given the name proliferative enteropathy (PE) to reflect the main pathologic changes (i.e., proliferation of crypt enterocytes). 24 Since then, PE has emerged as a disease of economic significance to the pig industry, causing clinical disease in a wide age range of pigs depending on specific manifestations. Economic impact assessments vary depending on the country of origin and clinical subtype but, overall, they indicate a potential for significant financial impact if L. intracellularis infection is highly prevalent.17,40,72,77
Detection of L. intracellularis infection remains a challenge for swine practitioners and producers. Infection can be subclinical but, even when clinical signs are apparent, diagnosis of PE may be complicated because several pathogens can have similar clinical presentations (e.g., Brachyspira hyodysenteriae, Salmonella enterica serovar Typhimurium, and porcine circovirus 2 [PCV-2]).3,9,100 The scarcity of suitably validated detection tools, together with a lack of pathognomonic clinical signs and intermittent shedding of bacteria, can hinder confident and meaningful diagnosis.
Our review focuses on recent advances in the diagnosis of PE. We also compare the value, advantages, and drawbacks of detection tools available to the pig production industry, to date.
Disease overview
Clinical signs of L. intracellularis infection in pigs have been well documented. The disease generally manifests in 1 of 2 ways clinically, acute PE and chronic PE, or the infection may remain subclinical.21,65,107
In the most fulminant (and less frequent) acute form of PE, L. intracellularis infection can be associated with sudden death of pigs > 4 mo old as a result of severe acute hemorrhage into the intestinal tract, variably known as proliferative hemorrhagic enteropathy or acute PE.60,93 In these severe cases, pigs may exsanguinate prior to the development of diarrhea, and the only other clinical sign may be pallor. However, in more prolonged cases, melena or hematochezia are appreciable. 107 The mortality rate for acute PE is 12–50% and is more typical in replacement breeder animals.31,55,74,106
The chronic form of PE causes non-hemorrhagic diarrhea (i.e., gray-green, semi-solid to liquid feces), weight loss, or reduced rates of weight gain in younger pigs, but clinical signs are often subtle, and diarrhea may not be present. 78 Affected pigs are 6–16 wk old and are often in suboptimal-to-poor body condition, with a normal-to-dull demeanor. Although widely known as chronic PE, this subtype is also termed porcine intestinal adenomatosis, proliferative ileitis, or just ileitis.24,93,107 Chronic PE is generally not fatal, and recovery within several weeks is the norm.92,107 The mortality rate is ~2%, and death is generally the result of secondary infections. However, fecal shedding of L. intracellularis increases the likelihood of pig-to-pig transmission, negatively impacting morbidity, feed conversion rates, treatment cost, and time to slaughter.2,17,29,74
Subclinical infection is usually characterized by reduced growth rates and increased feed conversion rates, in the absence of other clinical signs.21,24,43,84,92,98,106 Although subclinically infected pigs do not usually have diarrhea, they still generally have histologic lesions, shed the bacterium, and may remain a source of infection for other pigs. 11 Studies to account for the dichotomous outcome of L. intracellularis infection (i.e., subclinical infection vs. overt clinical signs [including acute or chronic PE]), are lacking to date.
Lesions
Macroscopic lesions associated with L. intracellularis infection can occur at all levels of the intestinal tract. Lesions are typically localized in the terminal ileum from where they may extend distally to the colon and cecum, or proximally to the distal jejunum.37,45,48 The intestinal mucosa may become thickened by raised islands of the mucosa (originally described as polyps). 7 In more severe or advanced cases, the islands coalesce, resulting in mucosa that has a thickened, corrugated, or cerebriform appearance (Fig. 1A). In some cases, there is focal necrosis, with superimposed yellow or gray deposits of fibrin and cellular debris (Fig. 1B). 57 Secondary infection may lead to necrotic enteritis, characterized by coagulative necrosis and superimposed fibrin. If the pig survives, the necrotic tissue is replaced by granulation tissue. This lesion is known as regional ileitis or, more colloquially, “hosepipe gut.” 94 Mucosal thickening is common to both the acute and chronic forms of PE, but only in acute PE does the intestinal content consist of blood, including blood clots, free blood, and fibrin, the exact origin of which remains unclear. Although highly suggestive of PE, these lesions are not solely indicative of L. intracellularis. For example, the gross features of PCV-2 enteritis can be very similar to the necrotizing form of PE. 46 Pigs may also be infected in the absence of macroscopically appreciable lesions, or lesions may be small and focal, and thus are easily overlooked postmortem. 36 Diagnosis may also be hampered if PE is masked or complicated by other diseases, such as salmonellosis or swine dysentery.78,100
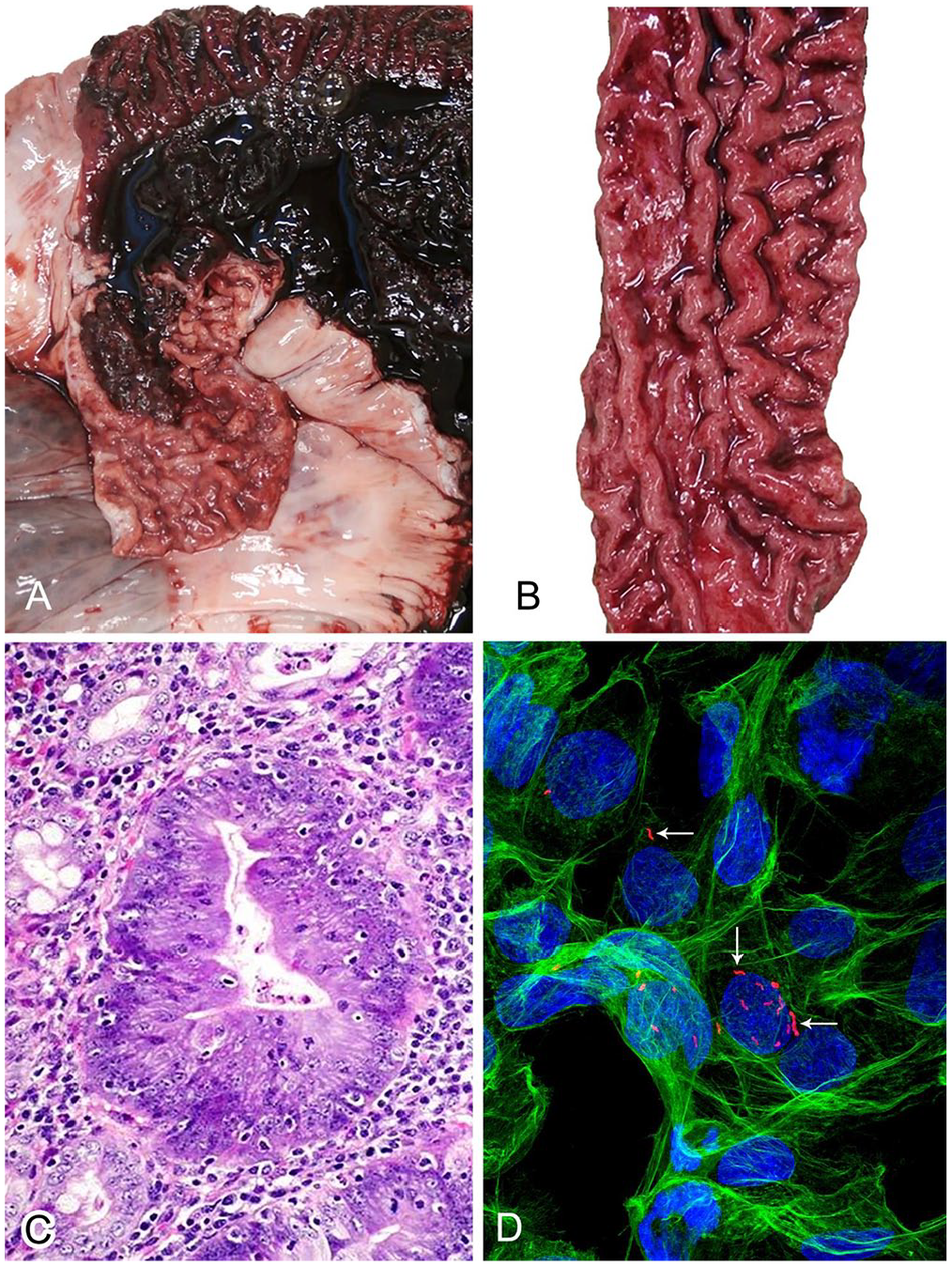
Lawsonia intracellularis infection and associated lesions.
The microscopic lesions of PE are very characteristic and generally pathognomonic, unless mild, infrequent, or complicated by secondary or concomitant infections. The lesions have been well characterized and, in short, consist of hyperplasia of crypt enterocytes with the formation of elongated, dilated, and branching crypts. The hyperplasia is accompanied by increased mitoses and a reduction in, or complete loss of, goblet cells (Fig. 1C; i.e., loss of differentiation of the mucosal epithelial population).6,57 Crypt hyperplasia is not specific, however, and can occur as a nonspecific reactive or regenerative change in the face of mucosal injury or ulceration.14,86
Immunology
Understanding the host immune response against a specific pathogen can help in the development of specific laboratory tests. A review summarizing the current knowledge of the porcine immune response to L. intracellularis and the potential applications of this response for diagnostic purposes was published in 2020. 83 We aim to put the timing of these responses into the context of their value for detection of the infection in relation to clinical signs and direct detection of the pathogen (Fig. 2).
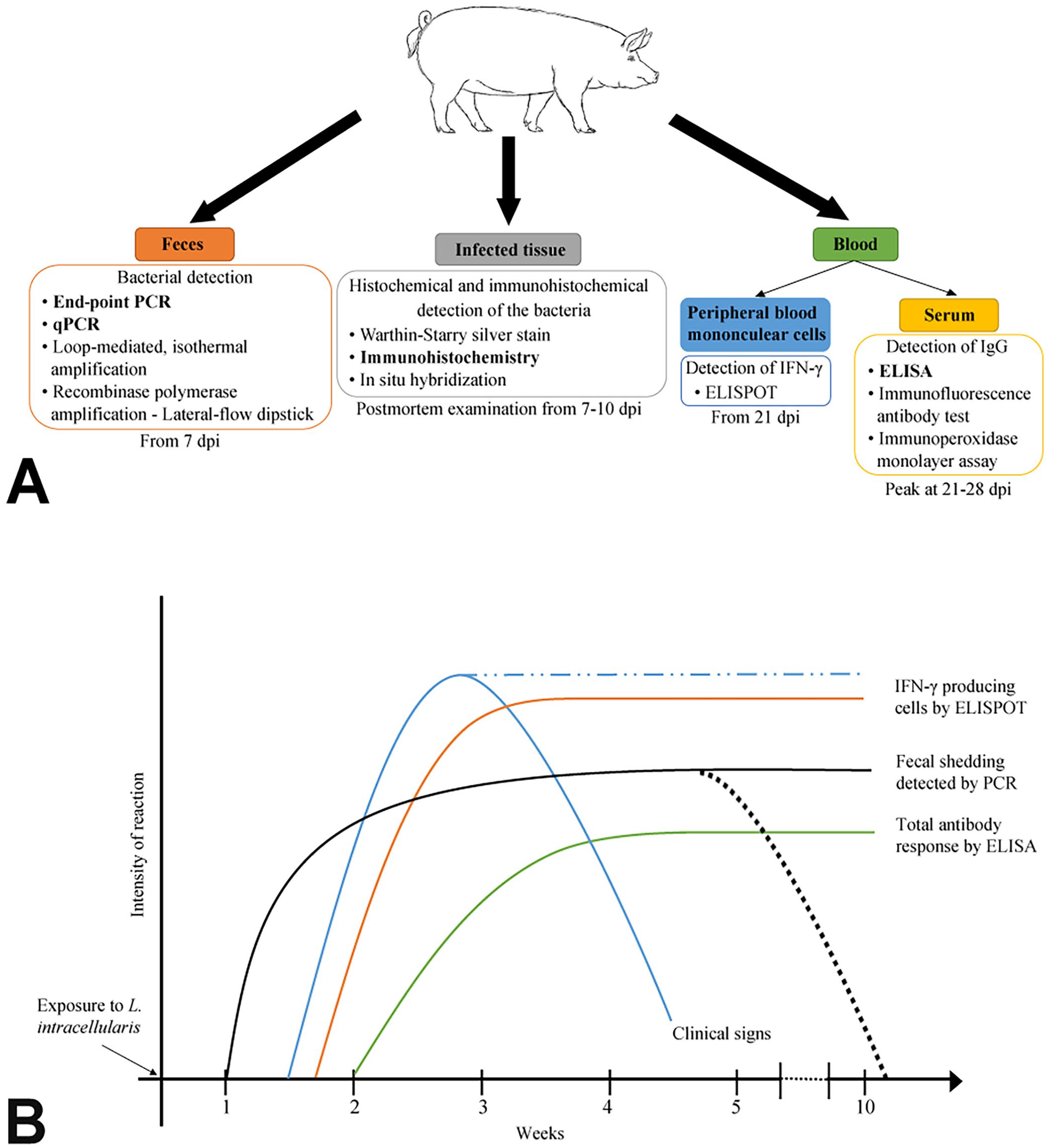
Detection of Lawsonia intracellularis infection.
Innate immunity is the first line of defense and, in pigs, the first signs of a general inflammatory response are expected 2–3 d post-infection (dpi). 68 In chronic PE, the absence of an inflammatory response including the relative dearth of immune cells in affected areas could be a consequence of bacterial inhibition.75,91 In contrast, during acute PE, some authors have described an immune response characterized by increased numbers of macrophages in the mucosa, as well as high levels of proinflammatory cytokines.65,66 The reason for these differences is unknown.19,30,65,82,91,105
The cell-mediated immune response to L. intracellularis is poorly defined. However, type 1 cytokine levels are often altered; for example, increased levels of interferon-gamma (IFN-γ), IL-6, IL-10, transforming growth factor–beta (TGF-β), and tumor necrosis factor–alpha (TNF-α) have been reported.82,112 Insights into the L. intracellularis–specific IFN-γ–producing cell responses have been published using the enzyme-linked immunospot (ELISpot) assay for IFN-γ detection, as well as the use of IFN-γ receptor knockout mice.28,30,83,91,95,112 These studies have concluded that IFN-γ is elevated in infected pigs from 10 dpi, peaking at 20 dpi, earlier than other cytokines (Fig. 2B). They have also demonstrated that IFN-γ plays a role in the control of L. intracellularis infection, given that mice lacking IFN-γ receptors are unable to control infection.28,95 These cytokine changes may be of use diagnostically in the future if faster and more inexpensive tools to detect them become available, but defining L. intracellularis infection specifically with such readouts alone would be impossible unless unique signatures are demonstrated. 25
With respect to humoral immunity, specific antibodies have been detected in the sera, intestinal mucosa, and feces of pigs experimentally infected with L. intracellularis. 30 Antibodies were also detected in oral fluids in pigs naturally infected with L. intracellularis.4,39,103 Circulating and/or fecal antibodies may have some potential for diagnostic purposes, as they may indicate previous exposure to L. intracellularis. Furthermore, in the case of vaccinated and field infections, the main antibody isotypes are IgG, IgM, and IgA and, hence, based on antibody types or levels, differentiation between vaccinated and naturally infected pigs is not possible. 82 Lawsonia-specific IgG and IgM responses can be detected in serum 2 wk after infection (Fig. 2B). They peak at 3–4 wk and remain detectable for up to 13 wk post-infection.31,82 IgG responses may result from active infection, vaccination, or colostrum intake.31,33,82,90 On the other hand, although IgA has been observed in feces, the concentration of IgA bears no relationship to the concentration of serum IgG.19,30,82 The mechanism by which IgA is involved in bacterial clearance is unknown.30,82
In vitro culture of L. intracellularis
L. intracellularis is an obligate intracellular, straight or curved, gram-negative, non–spore-forming bacillus in the Desulfovibrionaceae family. 26 There is a strong correlation between the presence of the organism in the apical cytoplasm of crypt enterocytes and the development of lesions.57,76,92,93 The diagnosis of many bacterial infections involves bacterial culture, 27 but culture has never been an option for L. intracellularis. Even when unadulterated by fecal material or competing gut flora, in vitro propagation of L. intracellularis is extremely difficult and remains a primary research hurdle. Only a few laboratories have the required in vitro culture systems available to satisfy the specific growth requirements of this organism. Briefly, the culture method, optimized from the first descriptions, comprises an anaerobic tri-gas mixture (80% nitrogen, 10% hydrogen, 10% carbon dioxide) that is used to generate strict atmospheric requirements. 58 Further optimization resulted in an alternative method using Original Space Bags (Storage Packs) inflated with the gas mixture rather than the use of a conventional anaerobic jar. 108 L. intracellularis can be cultured in PK15 cells (porcine kidney epithelial cells) and imaged by immunofluorescence (Fig. 1D).
Common tools for detection of L. intracellularis
Histochemical, immunohistochemical, and in situ hybridization techniques
There are several methods for confirming the presence of L. intracellularis bacteria, antigens, or nucleic acids in formalin-fixed or frozen tissue sections (Fig. 2A). Post-infection, up to 5 d of intracellular proliferation may be required for bacteria to reach levels that are detectable using these methods, although antigen can be detected until at least 14 dpi.10,37,106
Warthin–Starry silver stain
In 1920, a silver nitrate–based stain was first developed for improved detection of spirochetes. 110 The Warthin–Starry (WS) histologic stain works on the ability of spirochetal organisms to absorb silver from solution, given their distinctive double-layered envelope (diderm). 12 By confirming the localization of the bacterium within the intestinal crypt cells, the WS stain is a cost-effective, often frontline, method for confirming L. intracellularis infection. The WS stain is readily available in most veterinary diagnostic laboratories.12,41 However, the disadvantages of the WS stain include low specificity (e.g., L. intracellularis cannot be distinguished from other curved bacilli, such as Campylobacter spp.), reproducibility, and repeatability. 41
Immunohistochemistry and immunofluorescence
Immunohistochemistry (IHC) and immunofluorescence (IF) are similar tests based on the same principle but using different labeling methods. IHC has been well-reviewed in the general veterinary literature.15,88,89 IHC detection of L. intracellularis antigen is an established research and diagnostic tool.53,101 The test result can be visualized using a chromogen, most commonly 3,3′-diaminobenzidine, which serves as a substrate for the catalyst horseradish peroxidase. In practice, IHC applies mainly to formalin-fixed, paraffin-embedded tissue. The IF test, which uses a fluorescent label, is less applicable to routine use given the requirement for fluorescence microscopy.22,88
IHC was first described for use in PE in 1987, several years before the organism was classified and accepted as a novel genus and species. 73 Specifically, L. intracellularis bacteria (“Campylobacter-like organisms”) were detected in the intestinal mucosa and fecal smears from infected pigs and hamsters using a mouse monoclonal antibody (mAb) generated by immunizing mice with L. intracellularis that had originated from pigs with acute PE. This approach has subsequently formed the basis of what is now a commonly used technique for PE diagnosis; over the years, other groups have generated their own mAbs (Table 1).32,73 These antibodies can be used to detect organisms in fecal smears, and in formalin-fixed and fresh-frozen tissues, with the major benefit that they facilitate co-location of causative organisms and lesions in tissues. In 2005, a new mAb (Law-1-DK) was developed against a 21-kDa molecule that was resistant to proteinase K digestion, suggesting a non-protein target. 8 This antibody has since been used to detect L. intracellularis in feces and infected tissues, and there is also a commercial mAb (Moab a-Lawsonia intracellularis; Bio-X Diagnostics) that improves the accessibility of the technique for diagnostic laboratories.8,45
Histochemical and immune-based tests used in the diagnosis of proliferative enteropathy.

Ag = antigen; IF = immunofluorescence (IF) assay; WS = Warthin–Starry.
IHC and IF methods have proven to be the most reliable tests for the postmortem diagnosis of PE.13,41,45,49,56 One study using ileal samples from slaughterhouse pigs reported IF sensitivity of 89% and specificity of 97%, in addition to high agreement compared to PCR. 36 Similarly, another study comparing IHC with Warthin–Starry silver and routine hematoxylin and eosin staining concluded that IHC was superior to both and an excellent test overall, with a sensitivity of almost 87% when compared to PCR, the gold standard in that study. 36 Disadvantages of these antibody-based techniques are that they require specialized equipment and appropriately trained staff to analyze and interpret, contributing further to costs. However, the main advantage of both IF and IHC over PCR is that they can confirm the presence of Lawsonia within histologic lesions.
In situ hybridization
In situ hybridization (ISH) using L. intracellularis–specific oligonucleotide probes allows a comparable, if more expensive, approach to IHC, 45 and can be used on fixed tissue samples from infected pigs, similar to IHC. However, one study also found that fluorescent ISH (FISH) was more vulnerable to the effects of autolysis compared to IF when testing formalin-fixed porcine intestinal tissue samples, effectively reducing assay sensitivity. 45 Similarly to IHC, ISH requires specialized equipment and technical knowledge. Nonetheless, both IHC and ISH techniques are considered gold standards in the diagnosis of PE. 45
Detection of antibodies against L. intracellularis
As noted previously, IgG and IgM antibodies against L. intracellularis can be detected from ~14 dpi (Fig. 2B), and IgG has been shown to persist for at least 13 wk post-infection. 31 Demonstration of antibodies against L. intracellularis can be interpreted in 3 ways: 1) pigs are actively infected and have developed a humoral immune response; 2) pigs have received antibodies passively through colostrum; and 3) pigs were vaccinated against L. intracellularis. Any combination of these 3 scenarios is also possible, so meaningful interpretation of serology results can be difficult unless samples are compared over time or are collected in a cross-sectional manner from different age groups. To date, serum is the most commonly used sample type, and the only commercial kit (Svanovir L. intracellularis/Ileitis-Ab; Boehringer Ingelheim Svanova) that detects total immunoglobulins without differentiation of IgA, IgM, or IgG.
ELISA
Several ELISAs have been developed for the detection of L. intracellularis antibodies in serum.16,44,54,61 Most are competitive ELISAs, the major advantage of which is their ability to selectively detect the antibody recognized by the plated antigen, even in crude or impure samples. To date, only one commercial kit is available (Svanovir L. intracellularis/Ileitis-Ab; Table 1).44,109 This test is unable to quantify the response against L. intracellularis, and sensitivity may vary depending on the value of percent inhibition (PI) selected; at PI = 35, the test sensitivity is 72% and the specificity reaches 93%. 44 Lawsonia-specific serum antibody responses, IgG and IgM, can be detected in serum 2 wk after infection (Fig. 2B); they peak at 3–4 wk and remain detectable for up to 13 wk post-infection.31,82
Indirect fluorescent antibody test
The indirect fluorescent antibody test (IFAT) was first described for the detection of IgG antibodies in serum of pigs exposed to L. intracellularis.32,59 Briefly, serum samples are applied to a plate to which L. intracellularis–infected McCoy cells have been fixed. Following incubation and a series of washes, a detection antibody labeled with a fluorophore is added to the plate. Serial dilutions of serum samples are used to determine the concentration of antibodies directed against L. intracellularis. One disadvantage of IFAT is the need for a specialized fluorescence microscope. Furthermore, the generation of infected culture monolayers in slides for the test can require 5–7 d. 58 The principal advantage of IFAT is that results are measured as titers, which can be considered semiquantitative.
Immunoperoxidase monolayer assay
The immunoperoxidase monolayer assay (IPMA) is similar to the IFAT except that a colorimetric change is produced by the breakdown of a chemical substrate by a peroxidase conjugated to the secondary antibody. The major advantage of IPMA is that the resulting brown stain, unlike a fluorescent label, can be evaluated using a more readily available light microscope. Another advantage is that the brown stain is usually more stable and lasts longer than fluorescent stains, which fade over time and must be protected from light. The principal disadvantages of both IFAT and IMPA are nonspecific staining (IFAT more so than IPMA), low repeatability and reproducibility, and the highly subjective interpretation of results.34,36,61 Although IPMA and IFAT are considered the most specific techniques for detection of L. intracellularis seroconversion compared to the commercial and other research ELISAs, IPMA and IFAT both require a source of infected cells.20,35,67 Thus, there is the need to culture L. intracellularis in vitro, which remains one of the persistent challenges in PE research.20,35,56,67 Another drawback is that these tests can only detect infection >21 dpi, long after the appearance of clinical signs and establishment of L. intracellularis shedding (Fig. 2B). 31
Molecular detection
Amplification techniques, such as standard PCR, real-time PCR, and loop-mediated isothermal amplification (LAMP), offer potentially rapid and sensitive ways to demonstrate the presence of L. intracellularis DNA or RNA in samples (Fig. 2A). The assays are commonly applied to fecal samples, rectal swabs, or intestinal samples (Table 2, Fig. 2).50,62,64,81,97,99,111 Pigs experimentally infected with L. intracellularis are expected to show L. intracellularis shedding in feces by 7 dpi and may sustain shedding of the bacterium for up to 10 wk after infection (Fig. 2B).31,96 The gene target commonly used is the 16S ribosomal DNA gene (16S rDNA), 64 although aspA 99 and dnaA 111 have also been used as markers for L. intracellularis.42,64,111 Various primer pairs have been used for detection of L. intracellularis (Table 2). “In situ” amplification in tissue samples has also been reported and could potentially be a good candidate to substitute for tissue IHC. 80
Reported PCR techniques used to detect Lawsonia intracellularis DNA.
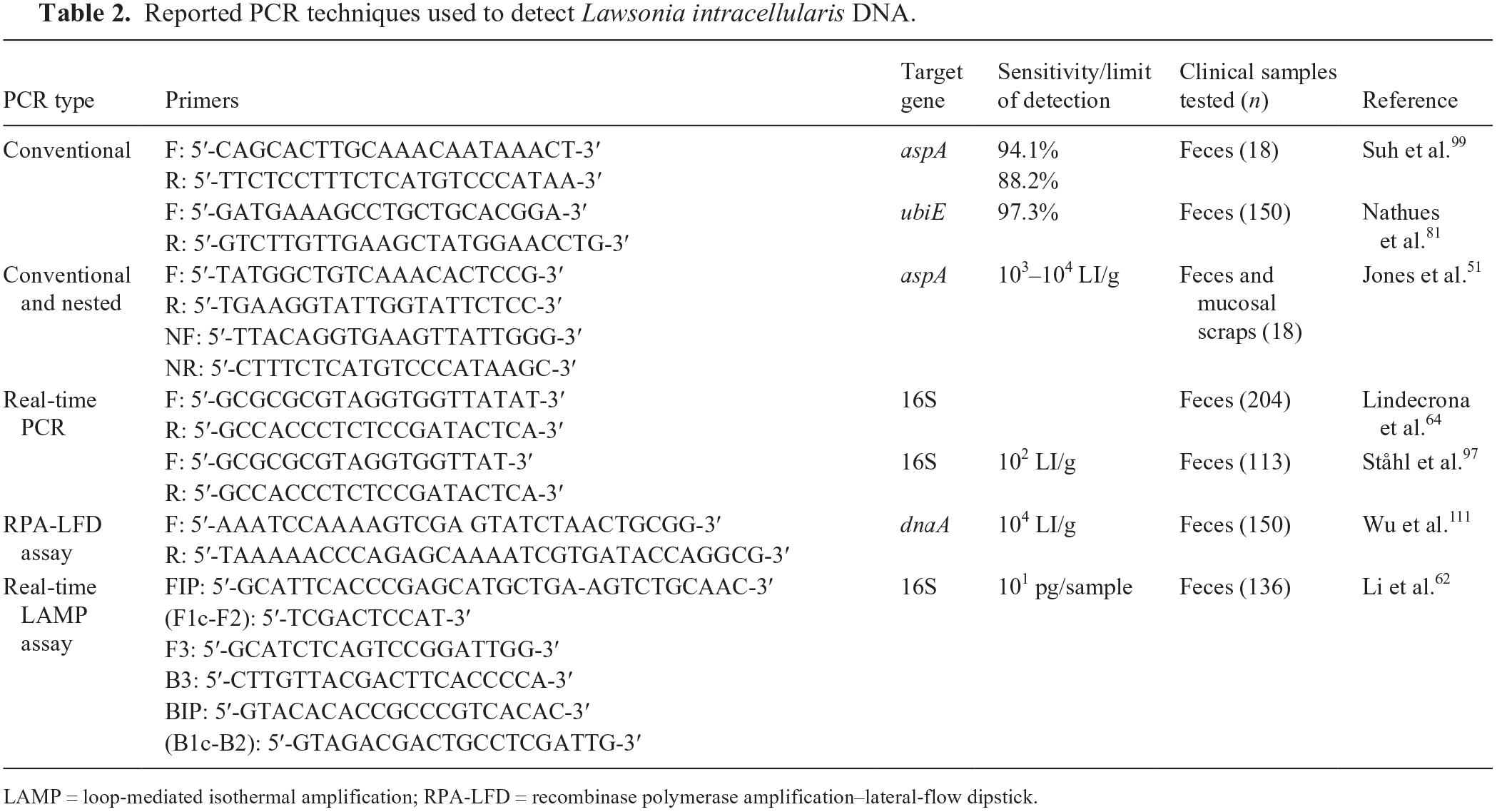
LAMP = loop-mediated isothermal amplification; RPA-LFD = recombinase polymerase amplification–lateral-flow dipstick.
Molecular methods are capable of more rapid and accurate antemortem detection of L. intracellularis during the first stages of infection, but their reported sensitivities and specificities span a wide range.85,87 Two innovative amplification assays have been developed for the detection of L. intracellularis. The first is a real-time LAMP assay that allows quantification of bacteria in a single 60-min reaction, targeting a conserved region of the 16S rDNA gene. 62 The second assay, initially described in early 2019, is a detection assay that combines an isothermal recombinase polymerase amplification of the L. intracellularis dnaA gene with the visual observation of the results using a lateral-flow dipstick containing streptavidin-coated colloidal gold. 111 These new approaches allow more rapid detection and could potentially be developed as point-of-care tests for use on-farm. Furthermore, a multiplex PCR has been used to simultaneously detect specific combinations of L. intracellularis, B. hyodysenteriae, Mycoplasma spp., and Salmonella serovars.23,78
Although some complex samples, such as feces, can contain contaminants that can inhibit amplification,52,79 this can be overcome with careful selection of extraction methods. Indeed, nucleic acid extraction is a critical step to allow the advantages of amplification techniques to be brought to “farm-side” use. New opportunities are emerging and driving the development of innovative techniques for better on-site testing. One example is the use of a cellulose-based filter paper that retains nucleic acids.69,113 This allows quick and clean on-site nucleic acid purification for amplification methods, such as the ones mentioned above. The next step for a truly on-farm amplification process will be the use of portable amplification systems. During 2020, the improvement in biosensor technology has made this type of system emerge and improve significantly.38,47,63
Although these devices will become useful for field veterinarians, there is still a need to improve the systems by better dealing with the various environmental conditions that swine veterinarians encounter in the field, such as highly variable temperatures, moisture, dust, biosecurity issues, availability of suitable facilities, sample processing, or the overall processing time from sample collection to result reading. Aside from the well-recognized and previously mentioned disadvantages of PCR, another issue is the inability to differentiate pathogenic strains of L. intracellularis from the live attenuated vaccine strain (Enterisol Ileitis; Boehringer Ingelheim Vetmedica). No such test is yet available, but comparative genomic studies may help define robust differences that can be exploited. Laboratory methods that can differentiate infected from vaccinated animals (DIVA) have been developed for other swine pathogens, such as pseudorabies virus (Suid alphaherpesvirus 1), classical swine fever virus (Pestivirus C), foot-and-mouth disease virus, and Salmonella.5,102,104 Some of these methods target specific markers that are added to vaccine formulations at the time of manufacture, ensuring a route for distinguishing vaccinated from naturally infected animals by serology.1,71 To date, there are no markers in commercial L. intracellularis vaccines, but the design of an amplification method based on the genomic differences between the Boehringer Ingelheim Vetmedica vaccine and wild-type strains would be an interesting approach for the development of a L. intracellularis DIVA method.
Summary
L. intracellularis remains an important bacterial pathogen in pigs, and PE remains a challenge to diagnose. At a practical level, diagnosis is tightly associated with the possible interventions available for the infection, with antimicrobial treatments used and a commercial vaccine available. Nevertheless, given that the impact of L. intracellularis infection varies widely in countries and between farms, it would not necessarily be beneficial to target L. intracellularis by blanket use of vaccines. In addition, the global drive toward reducing antimicrobial use will also deter prophylactic use of antibiotics for this infection. Hence, accurate diagnosis of PE remains important to rule out other infections with similar gross presentations and to enable appropriate advice on vaccination and antimicrobial treatment to be given, especially when more serious outbreaks occur.
Histochemical, IHC, and ISH techniques are invaluable for confirming L. intracellularis infections, but these require tissue samples at postmortem, extended time to process the samples, and require specialist skills to interpret the results. In turn, this makes diagnosis exclusive to a limited number of veterinary practices and diagnostic and research centers. By comparison, both serologic and pathogen detection assays can indicate past or current presence of the bacteria, respectively. However, they cannot be used unambiguously to define the etiology of the intestinal disease. Serologic assays for L. intracellularis are therefore applied mainly to measure the impact of vaccination treatments, determine when infection takes place, and examine prevalence.39,103
Although our review demonstrates that good progress is being made on ways to identify L. intracellularis infection antemortem, with the potential for the development of pen-side tests, we still need to understand the interactions that lead to the different clinical presentations in pigs so that diagnosis can quickly predict the likelihood and severity of an outbreak and appropriate action can be taken swiftly. Thus, we highlight the need to develop tools to improve the diagnosis of L. intracellularis disease, in relation to pathogen load and the immune response of the pig, as well as improve measurement of the efficacy of disease control actions on farms.
Footnotes
Acknowledgements
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding was provided by Biological Sciences Research Council (BBSRC)–Boehringer Ingelheim Training grant, Industrial CASE CVS (BB/P505006/1). Additional support was received through the BBSRC Institute Strategic Programme Grant awarded to the Roslin Institute (BB/P013740/1; BBS/E/D/20002174).
