Abstract
Forty-five Horsfield’s tortoises (Testudo horsfieldii; syn. Agrionemys horfieldii, Russian tortoise) belonging to different owners had decreased appetite and respiratory issues. Twenty-nine tortoises had epiphora, dyspnea, and white necrotic diphtheroid oral plaques (group G1). Ten of the remaining 16 tortoises had serious dehydration, appetite disorder, and depression (G2). The last 6 tortoises had only decreased appetite and moderate conjunctival discharge (G3). During the physical examination of all 45 tortoises, a cytologic sample and an oral swab for herpesvirus and Mycoplasma agassizii PCR testing were taken. In 20 of 29 specimens from G1, in 8 of 16 from G2, and 0 of 6 from G3, the cytologic exam revealed intranuclear acidophilic inclusion bodies, multinucleate cellular syncytia, and further abnormalities caused by herpesviral infection. Moreover, all 45 tested subjects were found to be positive for testudinid herpesvirus 1; 2 were positive for M. agassizii. This prospective study suggests that Horsfield’s tortoises with such signs would benefit from this screening procedure, given that it was effective in a significant proportion of infected and symptomatic animals, and no negative effects were seen.
Herpesviruses are nonenveloped DNA viruses in the family Herpesviridae. The current taxonomy is divided into 3 subgroups: Alphaherpesvirinae, generally considered pathogenic in avian and reptilian species; Betaherpesvirinae; and Gammaherpesvirinae. 5 Herpesviruses can remain dormant for a variable period of time depending on the immune system of the host and the species specificity of the virus. Species specificity as well as whether the species co-evolved with the virus are critical to the virulence of the viral strains. 5 DNA replication takes place inside the nucleus of the host cell, and during this replication it is possible to visualize the typical acidophilic intranuclear inclusion bodies (INIBs) in both cytologic and histologic samples. The presence of INIBs and their evolution are related to various factors, which can include immunity, secondary concomitant bacterial infections, and the virulence of the viral strain. 10 The virus is transmitted horizontally from infected to healthy subjects by direct contact with both secreted and excreted material.5,13 Vertical transmission has not yet been demonstrated. The excretion of virus may not coincide with clinical signs, given that seemingly healthy individuals can shed virus. The occurrence of clinical lesions appears to be more common in animals during springtime in conjunction with the end of the hibernation period. 15
Interest in keeping exotic animals as pets is growing and, consequently, veterinarians are increasingly involved in clinical activities related to exotic animals, using increasingly advanced testing techniques.2,14 In view of the existence of reptile-breeding farms with high concentrations of animals, it is important to study new methods for clinical and reproductive monitoring as well as for deepening the knowledge on their microbiologic characteristics, and for rapid diagnosis of reproductive and infectious diseases in order to prevent the loss of animals.3,16,17 Numerous cases of herpesviral infection have been reported in snakes, 7 saurians, 10 and chelonians. 6 Histologic enteric lesions (such as glossitis, stomatitis, and enteritis), central nervous system lesions (meningoencephalitis), and respiratory lesions (tracheitis and pneumonia) have been identified in infected tortoises. 11 Clinical signs could include anorexia, lethargy, vomiting, and oculonasal discharge. Mediterranean spur-thighed tortoises (Testudo graeca; syn. Greek tortoise) are frequently observed to have stomatitis, encephalitis, and rhinitis. 18 In the most serious cases of glossitis and stomatitis, one can often find white necrotic diphtheroid patches with underlying ulcerations. Secondary opportunistic bacterial and fungal infections are common and can result in septicemia and death. 7
There are numerous serologic techniques for the detection and typing of the virus or the direct highlighting of the acidophilic INIBs in the tissues where the virus replicates.5,8,18 Nevertheless, early identification of eosinophilic INIBs in the keratinocytes of the oropharynx would be very useful. First, the finding of acidophilic INIBs in the cells of the oropharyngeal epithelium is likely related to testudinid herpesvirus (TeHV) infection. 8 Another organism responsible for mild-to-severe respiratory tract diseases in chelonians is Mycoplasma spp., which are well-investigated pathogens that have been detected regularly in tortoises and turtles in Europe and the United States. 9 Upper respiratory tract disease (URTD) has been observed in a number of tortoises infected by Mycoplasma spp.4,9 Clinical signs of URTD in gopher tortoises are similar to those in desert tortoises and include serous, mucoid, or purulent discharge from the nares, excessive tearing to purulent ocular discharge, conjunctivitis, and edema of the eyelids and ocular glands. These signs are often comparable to herpesviral infection, and for this reason, clinical differentiation is not possible without serology or molecular techniques.
The protocol of our study was approved by the Ethics Committee for Animal Experimentation of the University of Parma (PROT.N. 14/CE/2019). We brushed the oral mucosa of 45 Horsfield’s tortoises (Testudo horsfieldii, syn. Agrionemys horfieldii, Russian tortoise), 3–7 y old, from April 2015 to April 2018. All subjects belonged to different owners. The tortoises were housed outside with the intent of allowing natural hibernation to occur. The tortoises were examined because of a lack of appetite, lethargy, and ocular and nasal discharge. Of the 45 subjects, 29 had epiphora, dyspnea, and white necrotic diphtheroid plaques in the oral cavity (Fig. 1A; group G1). When removed, mucosal ulcers were found under the plaques. Ten of the remaining 16 tortoises had only severe dehydration, inappetence, and depression (G2). The last 6 tortoises were alert to stimuli and had inappetence and mild conjunctival discharge (G3). During examination at the time of patient admission, 2 samples were taken: a cytologic sample and an oral swab without transport media that were both sent to a laboratory (Laboklin) for PCR testing for herpesvirus and Mycoplasma spp. None of the animals were rechecked.
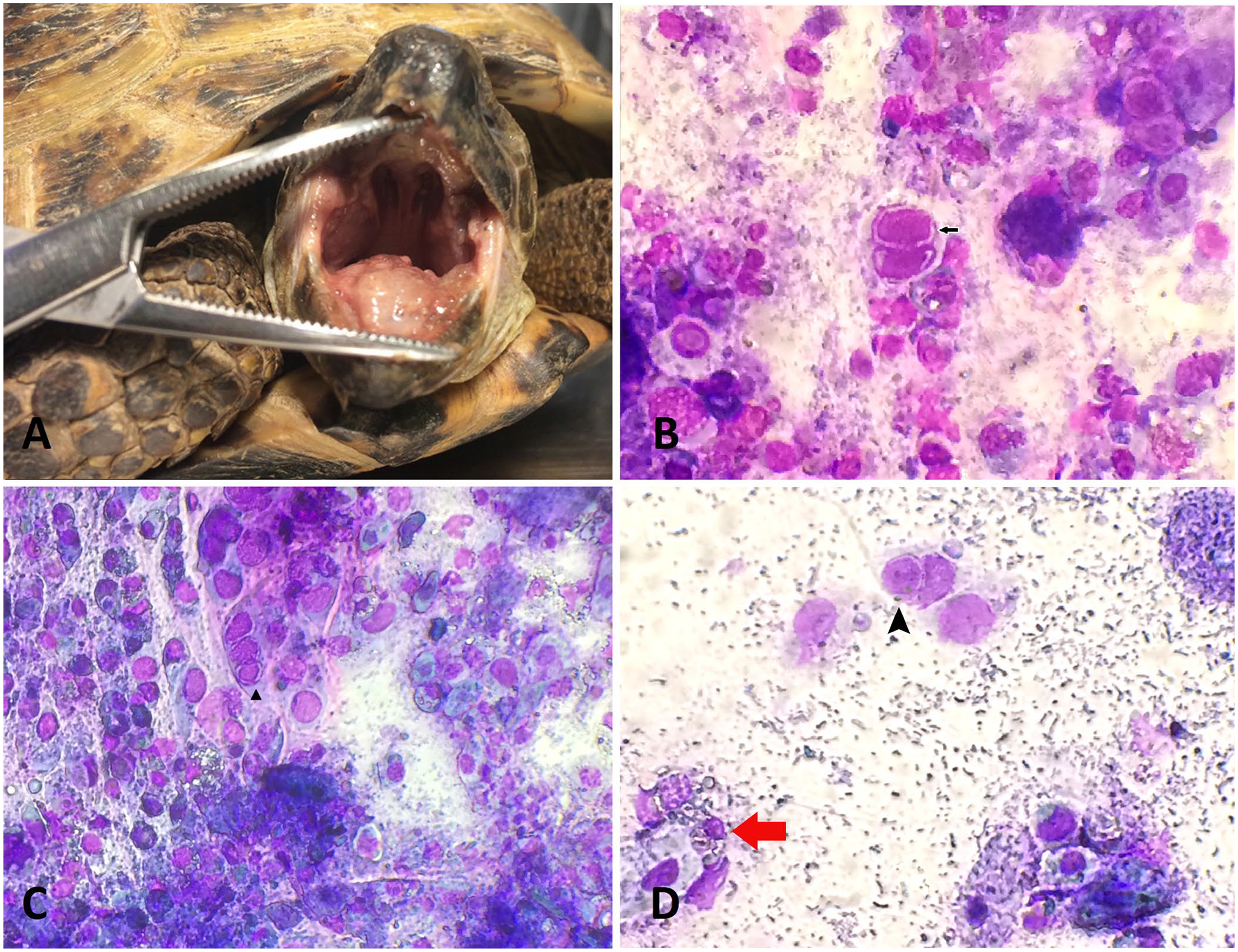
Mucosal diphtheroid plaques and mucosal brushing specimens from the oral cavity of symptomatic Horsfield’s tortoises.
The G1 and G2 animals were hospitalized separately in glass terraria with 12 h/d 5.0 spectrum UVB light for 1 mo. Temperature and humidity were selected according to species husbandry parameters. 4 All G3 animals were hospitalized until they ate spontaneously. Prepared DNA and RNA were stored at 4°C for 1 wk and then at −18°C. DNA and RNA were then extracted from samples (MagNA Pure 96 DNA and viral NA small volume kit; Roche). PCR tests were carried out for the detection of Mycoplasma spp. and herpesviruses. For the detection of herpesviruses, 3 different PCR tests were performed (Table 1). Consensus PCR detected a small region of the DNA polymerase gene of herpesviruses and was able to detect testudinid herpesviruses types 1 and 3 as well as many other members of the family Herpesviridae. 9 The second PCR also amplified a part of the DNA polymerase gene but was able to detect only TeHV-1.9,12 The third PCR detected a part of the UL5 gene and was specific for TeHV-3.9,18 Cytology was performed (Cytobrush Plus GT; Alcyon) by gentle scarification of the lingual surface and the oral vestibule. With the obtained material, several slides (SuperFrost; Thermo Fisher Scientific) were made and left to air-dry for several minutes. These samples were then stained (MGG quick stain; Bio Optica Milano) and observed under a microscope at 200× and 400×.
Primers used and references for the detection of different pathogens by PCR.

Cytologic evaluation showed the presence of acidophilic INIBs in the epithelial acantholytic cells of the oral cavity in 20 of 29 G1 tortoises and in 8 of 16 G2 tortoises. These INIBs were not found in specimens from any G3 tortoises. In some samples, there were clusters of epithelial cells with moderate-to-severe karyolysis and necrosis, numerous clusters of necrotic debris, and viral INIBs (Fig. 1B). Some of the infected cells were in multinucleate syncytia with chromatin margination (Fig. 1C). All of the samples had in common the numerous bacteria that originated from the oral cavity associated with heterophilic inflammation (Fig. 1D). The PCR results confirmed a TeHV-1 infection in all subjects of all 3 groups. Samples were also positive for Mycoplasma agassizii in 6 G1 tortoises and in 2 tortoises in each of G2 and G3. One month after testing, all 29 G1 tortoises had died, as had 10 of 16 G2 tortoises. Five of the 6 G3 tortoises had begun to eat regularly, without showing any physical problems; the sixth tortoise was found dead by the owner for unknown reasons, and the owner did not authorize an autopsy.
The Fisher exact test was used to evaluate whether the causality in the data distribution was an effect of the scarification results, and the results were statistically significant (p < 0.0025). A 2 × 3 contingency table was used. Fisher exact test was also used as a post hoc test. In the 2 × 2 contingency table, a Bonferroni corrected p < 0.05/3 or 0.02 for multiple correlation was used as the threshold of significance. 1 The post hoc results showed that there was no statistically significant difference between G1 and G2, but there were significant differences between G1 and G3 (p < 0.003) and between G2 and G3 (p < 0.004). Both tests were performed with statistical calculators (VassarStats, http://vassarstats.net/).
With gentle scarification, we demonstrated that it is possible to determine whether there are INIBs in tortoises. It is difficult to determine the exact moment when the virus replicates in the oropharyngeal epithelium and therefore would be positive for INIBs. We believe that the cytologic samples in our study were obtained in the period when the disease in these animals was in its acute or replication phase. The results suggest that mucosal brushing could be a useful early detection tool to identify herpesvirus-related INIBs, while awaiting PCR test results in animals affected by mild or severe nonspecific signs, such as dyspnea, ocular and nasal discharge, and oral necrotic diphtheroid plaques, given that it detected a significant proportion of infected and symptomatic animals, and no negative effects were seen. However, our results also suggest that it is not recommended to use mucosal scarification in search of INIBs in subjects with only nonspecific signs, such as decreased appetite and moderate ocular conjunctival discharge because the results could be inconclusive. Molecular techniques, such as PCR, and serology require several hours to days to obtain results. The identification of the INIBs through a rapid staining technique is immediate, inexpensive, and would support early changes to animal management by owners.
Footnotes
Acknowledgements
We thank Laboklin, Francesco Origgi of Universität Bern, Rachel E. Marschang of Laboklin, Massimo Millefanti and Ferlini Elena of Ambulatorio Veterinario Gaggiano, and Caterina Spiezio of Parco Natura Viva.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
