Abstract
A 2-year-old female, spur-thighed tortoise (Testudo graeca) was presented with poor body condition (1/5) and weakness. Fecal analysis revealed large numbers of oxyurid-like eggs, and radiographs were compatible with gastrointestinal obstruction. Despite supportive medical treatment, the animal died. At gross examination, an intestinal obstruction was confirmed. Histopathology revealed severe hyperplastic esophagitis and stomatitis with marked epithelial cytomegaly and enormous basophilic intranuclear inclusion bodies. Electron microscopy examination revealed a large number of 60–80 nm, nonenveloped, icosahedral virions arranged in crystalline arrays within nuclear inclusions of esophageal epithelial cells, morphologically compatible with adenovirus-like particles. PCR for virus identification was performed with DNA extracted from formalin-fixed, paraffin-embedded tissues. A nested, consensus pan-adenovirus PCR and sequencing analysis showed a novel adenovirus. According to phylogenetic calculations, it clustered to genus Atadenovirus in contrast with all other chelonian adenoviruses described to date. The present report details the pathologic findings associated with an adenovirus infection restricted to the upper digestive tract.
Infectious stomatitis is the most common clinical form of upper alimentary tract disease (UATD) in reptiles. 14 Various bacterial, fungal, and viral infections have been described that cause this condition in these animals.1,14,15 UATD may include gingivitis, glossitis, palatitis, cheilitis, and by extension, esophagitis, conjunctivitis, ophthalmitis, rhinitis, and pneumonia. In severe cases, UATD can cause septicemia and death, and often occurs secondary to other conditions, including improper environment husbandry, nutritional deficiencies, infestation with mites, oral trauma, and neoplasia. 14 In chelonians, infectious stomatitis is commonly related to herpesvirus, iridovirus, and bacterial infections such as Mycoplasma spp.1,14,15 However, clinical signs often overlap, making such infections grossly indistinguishable. 15 Adenoviral infections have been diagnosed in many species of reptiles, namely, crocodiles, snakes, lizards and, recently, in several species of chelonians.1,10,15 Adenoviruses have been described as opportunistic pathogens in many animal species, especially when additional factors (particularly concurrent infections) adversely affect the health of the host; however, some types of adenovirus can have a primary pathogenic role. 1
The adenoviruses found in snakes and lizards and studied by molecular techniques all belonged to the genus Atadenovirus.3,4,6,7,9 In chelonians, different types of adenoviral infections have been described. A systemic adenoviral infection that was associated with anorexia, lethargy, mucosal ulcerations, palatine erosions of the oral cavity, nasal and ocular discharge, and diarrhea was reported in Forsten’s tortoises (Indotestudo forstenii), and the virus was shown to belong to genus Siadenovirus.9,17 Interestingly, systemic adenoviral infection with the same siadenovirus was also reported in impressed tortoises (Manouria impressa) and a Burmese star tortoise (Geochelone platynota), with nonspecific or no clinical signs, which had been in contact with these Forsten’s tortoises. 4 A new type of adenovirus, differing from the rest of the adenovirus genera, was also isolated from several testudinoid turtles, including an ornate box turtle (syn. plains box turtle, Terrapene ornata ornata) with hepatic degeneration.5,8 In the present work, we describe a UATD in a spur-thighed tortoise (Testudo graeca) associated with an adenoviral infection.
A 2-year-old female, spur-thighed tortoise was presented with a 1-week history of anorexia and weakness. The animal belonged to a captive group of 10 spur-thighed tortoises from 2 to 4 years old. Two months before this clinical presentation, 4 new tortoises, 2 Mediterranean tortoises (Testudo hermanni), 1 spur-thighed tortoise, and 1 Russian tortoise (syn. Horsfield’s tortoise, Testudo horsfieldii), from another group, were added to the private collection. Two spur-thighed tortoises had died without premonitory signs during the 2 months following these introductions but no autopsies were performed.
On physical examination, the animal showed generalized weakness, weighed 76 g, was 7.4 cm long, and in poor body condition (1/5). The carapace was hard, but the animal could not stand up with its limbs. Oral mucosa was dry and pale, and 5% dehydration was estimated. A cloacal wash was performed to obtain a fecal sample, which showed large numbers of oxyurid-like eggs. Radiographic examination revealed severe gastric and duodenal gas dilation with intestinal foreign bodies, compatible with gastrointestinal obstruction.
The animal was hospitalized with the suspicion of a systemic infectious disease, and was placed in a terrarium, with a heat lamp to provide 28°C, ultraviolet B light, and water. Enrofloxacin a (5 mg/kg/24 h intramuscularly), vitamin complex, b and intracoelomic fluid c therapy (40 mL/kg/24 h Ringer lactate and glucose 5%) were administered but the animal died the following day.
The tortoise was submitted for autopsy. At gross examination, the gingiva and tongue were pale and covered with mucus. An intestinal obstruction composed of dehydrated feces and a few nematode worms was observed. Parasitologic identification of the intestinal worms was not performed. In addition, a few small calculi were detected within the urinary bladder.
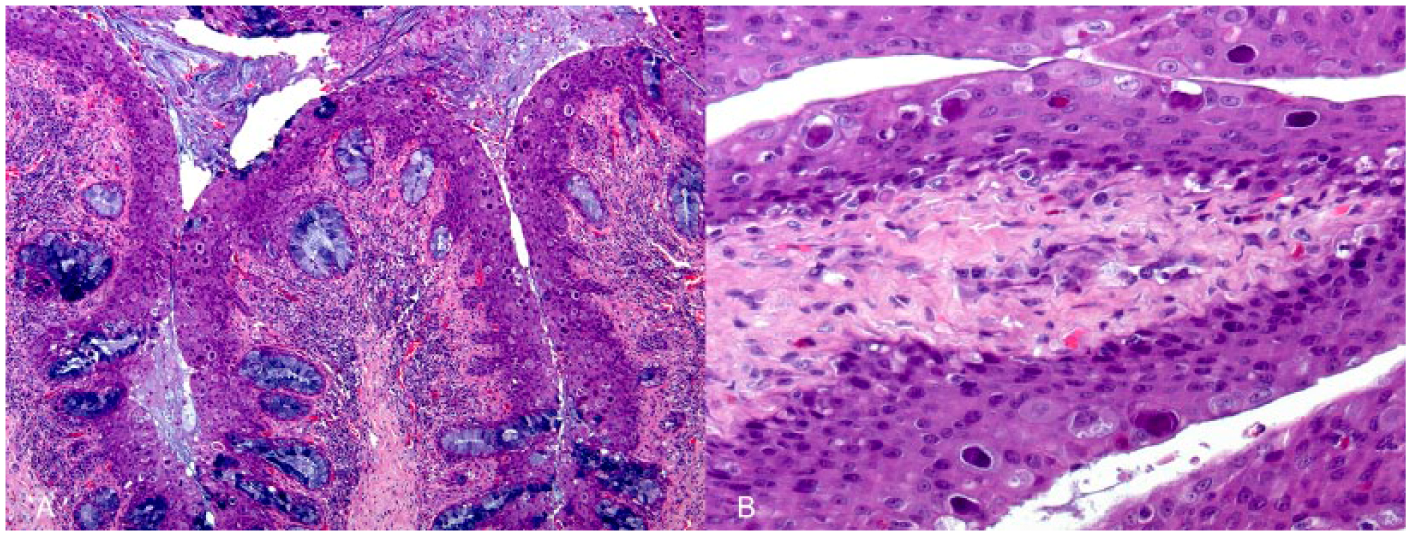
Tissues samples from tongue, esophagus, stomach, intestine, liver, kidney, heart, and lung were fixed by immersion in 10% neutral buffered formalin and were routinely embedded in paraffin wax, sectioned at 3 µm thickness, and stained with hematoxylin and eosin. Histologically, the epithelium of the tongue and the esophagus showed marked hyperplasia with mild to moderate lymphoplasmacytic inflammation in the superficial lamina propria (Fig. 1A). Numerous, round to oval, basophilic, 20–30 µm diameter, intranuclear inclusion bodies were observed within the nuclei of mucosal epithelial cells (Fig. 1B). Cell desquamation and marked hyperplasia of the mucous glands in the lamina propria were also observed in the esophagus. Multifocal, mild to moderate renal gout was also seen; it consisted of tubular dilation with abundant intraluminal acicular and unstained urate crystals, surrounded by macrophage infiltration. No other lesions were observed.
Tortoise (Testudo graeca).
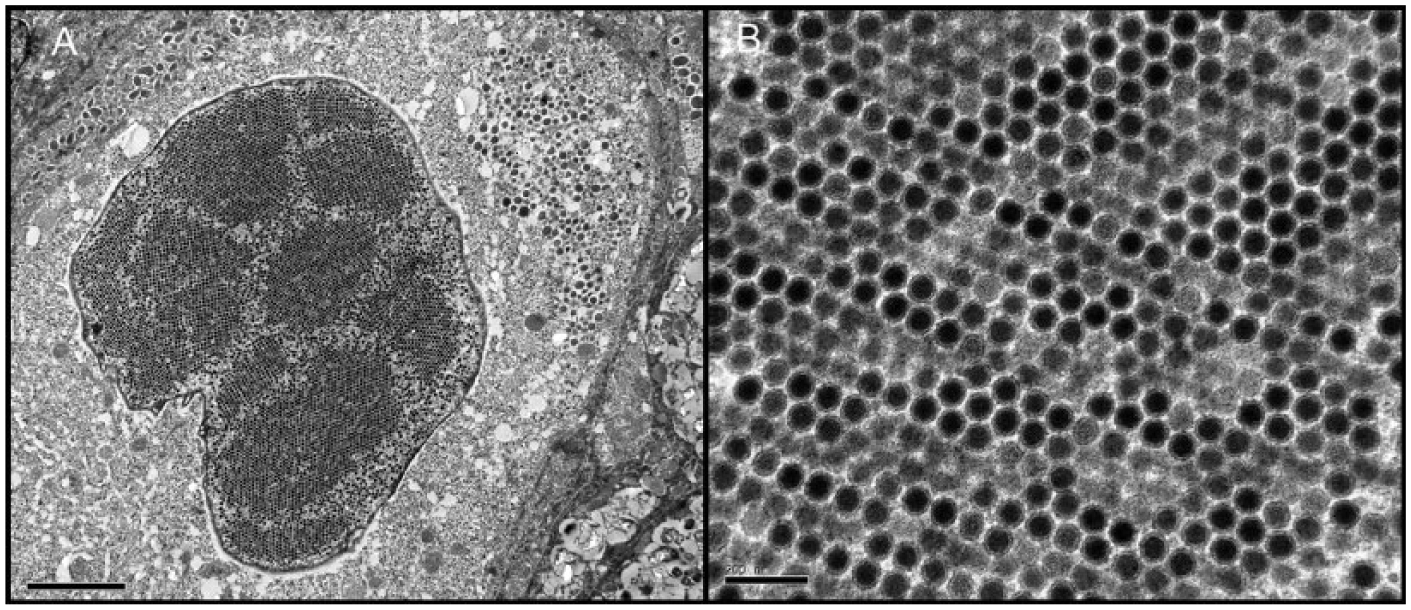
For ultrastructural evaluation using transmission electron microscopy (TEM), a sample of paraffin-embedded tongue was pretreated with xylene for deparaffinization and transferred to 100% acetone, embedded in commercial resin, d and polymerized at 60°C for 48 h. Semithin sections (1 µm thick) were obtained with an ultra-cut microtome, stained with 1% (w/v) aqueous toluidine blue solution, and examined with a light microscope to identify areas with intranuclear inclusion bodies for further analysis. Ultra-thin sections (70 nm thick) were cut with a diamond knife, f placed on noncoated 200-mesh copper grids, and contrasted with conventional uranyl acetate (30 min) and Reynolds lead citrate (5 min) solutions. Sections were observed with an electron microscope. g TEM revealed the presence of a large accumulation of 60–80 nm diameter, nonenveloped, icosahedral viral particles in the nuclei of esophageal epithelial cells (Fig. 2A). Viral particles were arranged in crystalline arrays morphologically compatible with adenovirus-like particles (Fig. 2B). Epithelial cells also showed chromatin margination and abnormal nuclear shapes and sizes.
Tortoise (Testudo graeca).
After histologic and TEM observations, DNA was extracted from the paraffin blocks. A DNA extraction protocol with no additional heating was applied. Samples were incubated first in 100% then in 75% xylene, and the same was repeated with ethanol at the same percentages. After the extraction of all paraffin remains, the tissue was digested in Tris-EDTA buffer using proteinase K. The procedure was terminated with the precipitation of DNA from the mixture. The success of DNA isolation was verified with the universal primers L14841 and H15149 targeting the cytochrome b gene, 11 and the extracted DNA was then submitted to nested pan-adenovirus PCR by consensus primers as described. 18
PCR fragments were sequenced from both directions using a cycle sequencing kit e and sent for electrophoresis at a commercial service, using a genetic analyzer. h Assembly was performed by Staden package version 4.10 (http://staden.sourceforge.net/). Phylogenetic inference was based on the derived amino acid sequence of the PCR fragment. Furthermore, sequences of the homologous protein fragment of at least 4 distinct adenoviruses of each genus (with the exception of the monotypic genus Ichtadenovirus) were collected from GenBank utilizing the BLASTP algorithm (http://goo.gl/kH3mfw). Despite the rather short size of these partial polymerase fragments, the fragments have proven to be adequate for reliable classification of adenoviruses at the genus level.2,12,18 Sequences were aligned with the insertion of only 2 gaps using the Clustal X BLOSUM62 algorithm (http://www.clustal.org/clustal2/). A guide tree, using PHYLIP version 3.695 (http://goo.gl/yqnFOM) was constructed based on the protein sequence of the fragment amplified, using the protdist (JTT) and Fitch (Fitch–Margoliash with global rearrangements) algorithms. Model selection and maximum likelihood tree estimation were performed by ProtTest version 2.4 (http://goo.gl/arf6ui). Models were ranked by their Akaike information criteria, and the tree was constructed based on the LG + I + G model with α = 0.58 and ρ = 0.18. The phylogenetic tree was visualized using FigTree version 1.3.1 (http://goo.gl/DCrvfN).
In the present case, the histologic lesions and TEM findings were highly indicative of an adenoviral infection, which was confirmed by the results of the nested pan-adenovirus PCR. This technique yielded a 275-bp long fragment corresponding to a portion of the DNA polymerase gene of a novel adenovirus displaying the highest (70%) sequence identity with lizard adenovirus 2.12,13 The G + C content was 56.12%. According to phylogenetic calculations, the novel spur-thighed tortoise adenovirus is a new member of genus Atadenovirus (Fig. 3). The sequence obtained has been deposited to GenBank under accession KT310086.
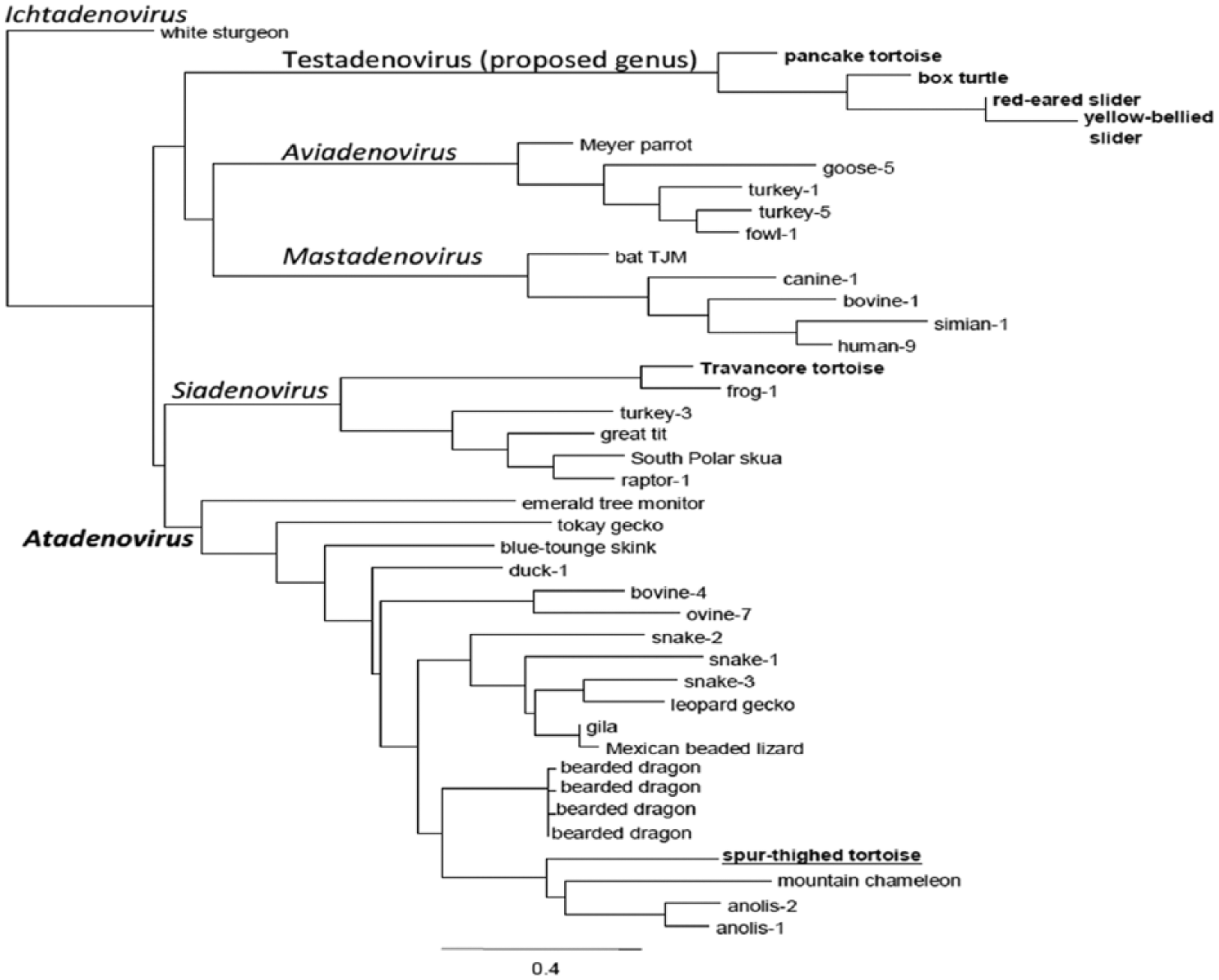
Result of the phylogenetic tree reconstruction (maximum likelihood with LG + I + G, α = 0.58, ρ = 0.18). The novel spur-thighed tortoise adenovirus is underlined and in bold, whereas bold fonts indicate all previously detected and partially sequenced chelonian adenoviruses. The spur-thighed tortoise adenovirus clusters to Atadenovirus as the only chelonian member of this genus.
Adenoviral infections in reptiles can cause respiratory infections that can be accompanied by lethargy, neurologic disorders, esophagitis, hepatitis, splenitis, or gastroenteritis.1,15 However, the pathogenicity of reptilian adenoviruses may vary depending on the type of virus and the host.1,15 Some authors described adeno-like viral particles associated with proliferative esophageal and tracheal mucosal epithelium in a Jackson’s chameleon (Chamaeleo jacksonii), 10 mimicking some of the changes seen in the present case. In chelonians, a systemic infection caused by a novel siadenovirus has been reported in several species of tortoises: Forsten’s tortoises, impressed tortoises, and a Burmese star tortoise.16,17 This virus was identified as Sulawesi tortoise adenovirus 1 (STAdV-1) and caused inflammatory lesions in multiple organ systems including bone marrow. Hence, a possible immunosuppressive role was suggested. 16 Adenovirus was also detected in an ornate box turtle with hepatic vacuolar degeneration and intranuclear inclusion bodies. 8 In other species of chelonians, namely, pancake tortoise (Malacochersus tornieri), eastern box turtles (Terrapene carolina carolina), red-eared sliders (Trachemys scripta elegans), and yellow-bellied sliders (T. scripta scripta), a new adenovirus lineage was isolated and is being proposed as a candidate for a new genus. 5 This lineage did not show a clear pathogenic role and was therefore speculated to have coevolved with testudinoid turtles. 5
In the present case, we reported an adenoviral-like infection restricted to the upper digestive tract. Regrettably, it was not possible to establish whether this animal suffered from a primary pathogenic adenoviral infection, or if it was a consequence of a stressful state produced by the intestinal parasitosis or other unrelated factors. Additionally, as no bacterial cultures were performed, mycoplasmal or other bacterial co-infections could not be ruled out. Nevertheless, the combination of this viral infection and the parasitic obstruction most likely contributed to the death of the animal.
Although the source of this viral infection could not be elucidated, based on the appearance of clinical signs 2 months after incorporation of 4 new tortoises representing 3 different species into the group, it is possible that one of them was carrying this virus. Some authors suggest that some adenoviruses (e.g., STAdV-1) can be transmitted to and infect different tortoise species. 17 Current taxonomy of the Adenoviridae family indicates a co-evolutionary lineage of the viruses with their hosts, including additional host switches (e.g., members of genus Atadenovirus from squamata to birds and ruminants, or more often between closer species). Because of a lack of time to adapt to the new species, host switching seems to result in a more pathogenic outcome. 4 Hence, in the present case, a hypothetical transmission of this adenovirus between different species could not be ruled out.
Footnotes
Acknowledgements
We thank Blanca Pérez and Aida Neira for their technical assistance with histologic processing; the personnel of Servei de Microscopia Electrònica from Universitat Autònoma de Barcelona, particularly Alejandro Sánchez-Chardi, for their excellent technical support, and Dr. Enric Vidal for critical review of the manuscript. We also thank the Translations and Proofreading Service of the Universitat Autònoma de Barcelona for English correction and editing. We also thank Dr. Norman Cheville for his help in TEM interpretation.
Authors’ contributions
B Garcia-Morante, JJ Pénzes, and T Costa contributed to design of the study and contributed to analysis and interpretation of data. J Martorell contributed to conception of the study and contributed to acquisition, analysis, and interpretation of data. J Martínez contributed to conception and design of the study and contributed to acquisition, analysis, and interpretation of data. All authors drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Baytril 2.5%, Bayer plc, Newbury, Berkshire, United Kingdom.
b.
Duphafral Multi, Zoetis, Vall de Bianya (Girona), Spain.
c.
B. Braun Medical Inc., Rubí (Barcelona), Spain.
d.
Eponate 12 resin, Ted Pella Inc., Redding, CA.
e.
BigDye Terminator v3.1, Leica Microsystems GmbH, Wetzlar, Germany.
f.
45°, Diatome AG, Biel, Switzerland.
g.
Jeol 1400, Jeol Ltd., Tokyo, Japan.
h.
ABI PRISM 3100, Life Technologies Corp., Carlsbad, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support came from Servei de Diagnòstic de Patologia Veterinaria (Universitat Autònoma Barcelona) and from the Hungarian Scientific Research Fund (OTKA K100163).
