Abstract
A 4-year-old donkey was evaluated for progressive neurological abnormalities consisting of depression, stupor, weakness, and recumbency. Diagnostic evaluation for viral involvement identified an asinine herpesvirus in DNA extracted from deep pharyngeal swabs. Specific primers were designed based on comparison with equine herpesviral DNA polymerase sequences and yielded an 875-base pair product from the donkey. This sequence had complete identity with short sequences of asinine herpesvirus previously identified in donkeys with interstitial pneumonia. Amino acid analysis of the entire sequence indicated high similarity with
A 4-year-old jenny standard donkey was referred to the Veterinary Teaching Hospital, Ontario Veterinary College (University of Guelph, Guelph, Ontario, Canada), for evaluation of depression and neurological signs. The patient had become progressively lame on the left hind limb on the day before referral and then gradually progressed to generalized weakness, head pressing, and eventual stupor and recumbency. The donkey had been treated by the referring veterinarian with intravenous trimethoprim-sulfadoxine a and intramuscular flunixin meglumine b and isoflupredone acetate c injections.
The donkey shared the pasture with another jenny donkey of the same age and 2 goats. All animals had been purchased 6 months previously. Two dogs and 1 cat were present on the premises and had been in regular contact with the affected animal. Feeding consisted of pasture access and free choice hay. Both donkeys had been vaccinated for rabies, tetanus, equine influenza, and
Upon presentation at the teaching hospital, the donkey was in lateral recumbency, mildly hypothermic (36.5°C), and near nonresponsive to stimuli. Physical exam revealed an increased heart rate and a normal respiratory rate. The mucous membranes were tacky, and dehydration was estimated to be 7%. Blood gases, plasma electrolytes, a complete blood cell count, and a serum biochemistry profile were assessed. Hematological abnormalities were mild neutrophilia (10.9 × 10 9 /l) and lymphopenia (1.5 × 10 9 /l), which were attributed to stress and treatment with corticosteroids. Mild hyperglycemia and hyperproteinemia identified on biochemical analysis were likely due to stress and dehydration, respectively. Fecal parasite examination identified a large number of strongyle eggs.
The donkey was placed in an Anderson sling. In the sling, the patient was able to eat and drink while supporting her body weight through standing for short periods of time. Intravenous fluid therapy with lactated Ringer solution was instituted to correct dehydration. The donkey was stimulated frequently to prevent her from returning to a stuporous state. Such arousal was applied every 2 hr to move the animal and relieve pressure from the sling support and to assure water and feed intake. Therapy with intravenous trimethoprim-sulfadoxine a was continued, and intravenous dexamethasone d was administered.
Neurological examination identified bilaterally absent menace reflexes with normal palpebral reflexes. The nasal septum stimulation response was decreased, and mild paresis and ataxia were observed. These signs were consistent with a lesion in the thalamocortex and possibly involving the spinal cord. Differential diagnoses included a thalamocortical encephalopathy of metabolic origin, herpesvirus-associated neurological disease,
A deep pharyngeal swab was submitted for EHV-1 and consensus herpesviral polymerase chain reaction (PCR) testing. DNA was extracted
e
and amplified. Primers for EHV-1 were f-

Multiple sequence alignment of 55 amino acids in the viral DNA polymerase gene from asinine herpesviruses (indicated by the prefix AsHV),
Twenty-four hours after hospitalization, the donkey was able to stand and the Anderson sling was removed. The animal continued to improve neurologically, but developed mild laminitis, which was treated with appropriate therapy. The donkey was discharged, and 3 weeks later no clinical abnormalities were present.
Herpesviruses are well recognized as causes of neurological disease in horses and other species. 1 In horses, disease is mostly characterized by myeloencephalopathy manifesting with clinical signs referable to spinal cord dysfunction. Microscopically, the lesions consist of vasculitis with hemorrhage and thrombosis and secondary ischemic degeneration. 13 Neurological disease is most frequently attributed to reactivation of latent EHV-1, an alphaherpesvirus, although EHV-2, a gammaherpesvirus, has also been identified in the central nervous system of horses and might act as a transactivating factor. 10 To the authors' knowledge, neurological disease in donkeys associated with a herpesvirus has not previously been described.
The donkey in the current report had neurological disease consistent with lesions in the thalamocortex and possibly spinal cord. Lack of inflammation in the CSF rendered bacterial or parasitic encephalomyelopathy unlikely, and recovery of the animal ruled out rabies. A herpesvirus was identified in oronasal mucosa obtained with deep pharyngeal swabs by PCR using first nonspecific and then specific primers. Although the positive PCR product did not prove causation of the neurological disease by this herpesvirus, similarity of the clinical features with EHV-1-associated neurological disease in horses
1
and the clinical course with full recovery, suggested that the asinine herpesvirus designated AsHV-OVC was the cause of disease in this animal. Whether disease resulted from primary or reactivated latent infection could not be determined. The viral sequence recovered was distinct from EHV-2 and −7, but closely related and of sufficient length to confidently assign this virus to the gammaherpesviruses.
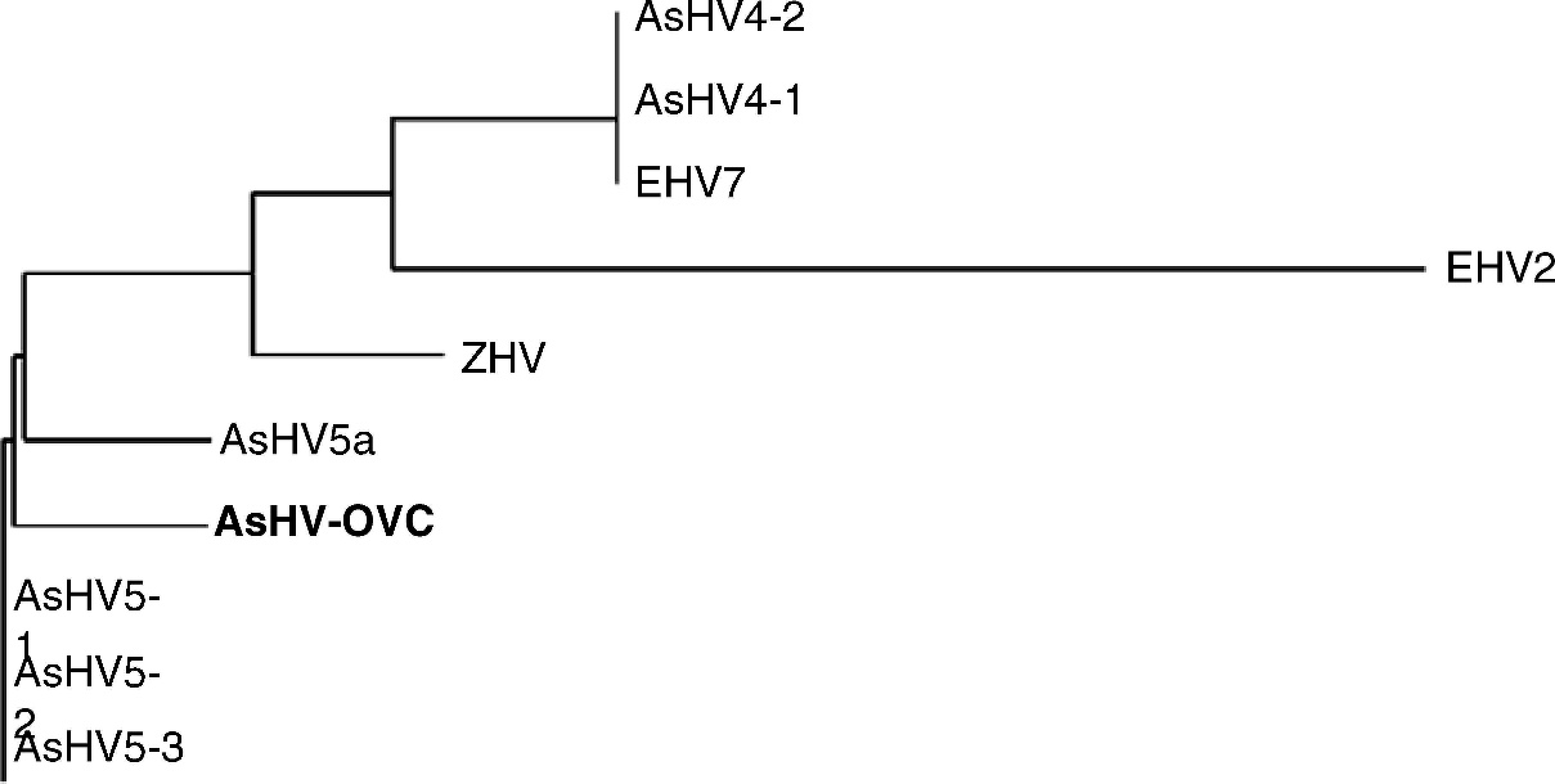
Phylogenetic tree of a 55-amino acid sequence from multiple equine gammaherpesvims sequences. AsHV indicates asinine herpesviruses, EHV7 indicates
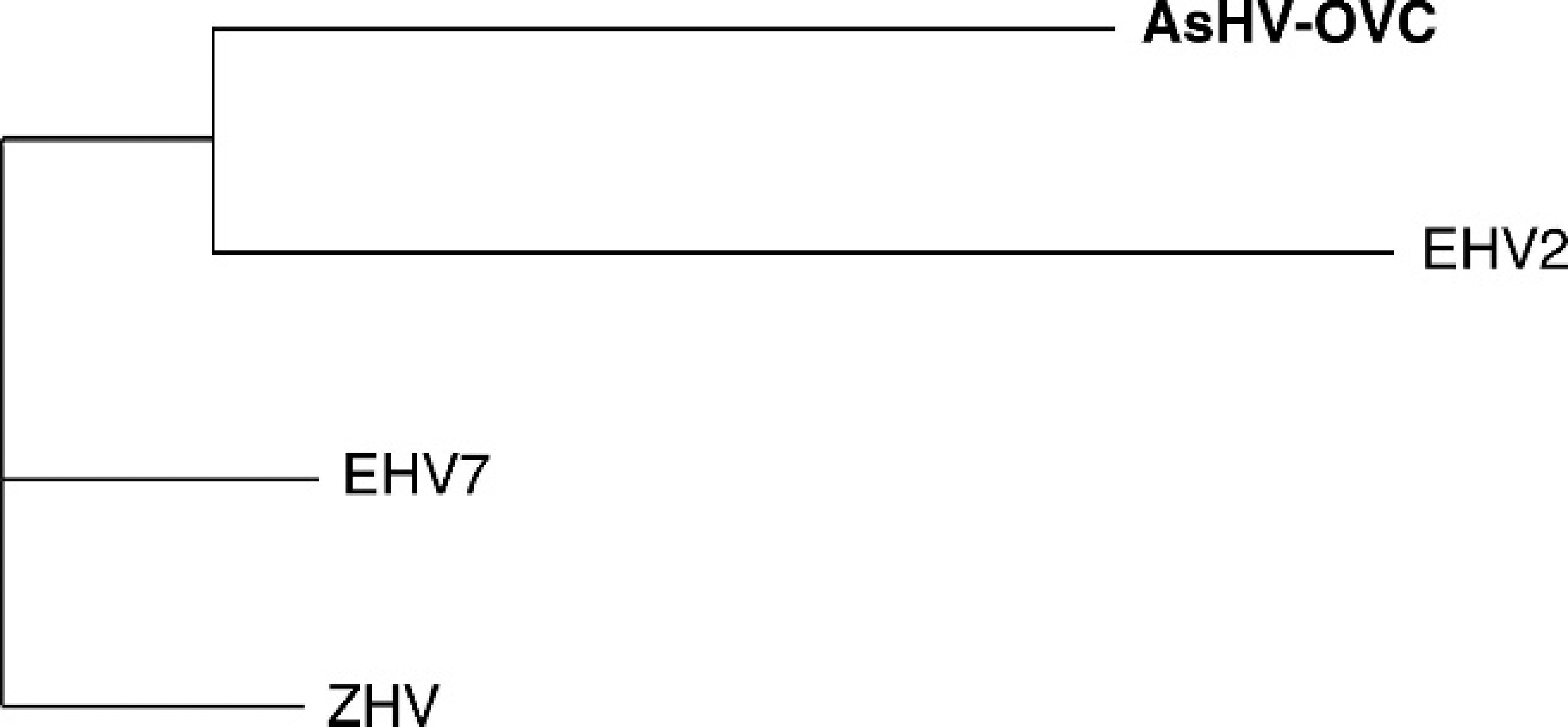
Phylogenetic tree of 291 amino acids located in the viral DNA polymerase gene of the novel asinine herpesvirus (indicated by AsHV-OVC) and
Asinine herpesviruses previously identified include AsHV-1 associated with coital exanthema, AsHV-2 isolated from leukocytes of healthy animals, AsHV-3 obtained from the nasal cavity of immunosuppressed animals, 2 and AsHV-4, −5, and −6 from animals with interstitial pneumonia. 7,8 Asinine herpesvirus 1 was classified as an alphaherpesvirus, but had little similarity with EHV-3 by restriction endonuclease and Southern blot analysis. 2 Asinine herpesvirus 2 had characteristics of betaherpesviruses, but little similarity with EHV-2 and −5 as determined with the same molecular methods. 2 Asinine herpesvirus 3 is an alphaherpesvirus based on sequence analysis of glycoprotein G and serological cross-reactivity of AsHV-3 with antibodies to EHV-1 and −4 glycoproteins. 3,6,9 Asinine herpesviruses 4, 5, and 6 were designated as novel gammaherpesviruses based on sequences obtained from donkeys with interstitial pneumonia. 7,8 The latter viruses were highly similar over ∼165 bp in the DNA polymerase gene and ∼380 bp in the terminase gene and appeared to represent closely related gammaherpesviruses. Overall, paucity of sequence information for AsHV-1–3, and limited sequence availability for AsHV-4–6, constrains comparison and classification of equine herpesviruses. Hence, although the longer sequence obtained for the virus in the current report allowed convincing classification as a gammaherpesvirus and assignment of close relation with EHV-2 and −7, it was not possible to determine with confidence whether this virus is identical to AsHV-5, or whether AsHV-4, −5, −6 and this novel virus represent a very closely related group of asinine gammaherpesviruses. However, identification of this particular herpesvirus with an assay capable of amplifying a range of herpesviruses in an animal with neurological disease adds to the spectrum of diseases potentially attributable to herpesviral infection of donkeys.
Footnotes
a.
Borgal®, Schering Canada Inc., Pointe Claire, Quebec, Canada.
b.
Banamine Solution Injectable, Schering Canada Inc., Pointe Claire, Quebec, Canada.
c.
Predef®, Pfizer Animal Health, Pfizer Canada Inc., Kirkland, Quebec, Canada.
d.
Dexamethasone, Vétoquinol North America Inc., Lavaltrie, Quebec, Canada.
e.
QIAamp® DNA Mini Kit, Qiagen Inc., Mississauga, Ontario, Canada.
f.
Vector NTI®, Invitrogen Canada Inc., Burlington, Ontario, Canada.
g.
Buzone Concentrate Powder, Vétoquinol North America Inc., Lavaltrie, Quebec, Canada.
