Abstract
Thromboelastography (TEG) is a point-of-care whole blood test of hemostasis. While TEG is becoming more widely used in veterinary medicine, few studies describe the use of TEG in cats. The objectives of the current study were to: 1) document the range of TEG variables produced in healthy cats using 3 sample types (citrated native, kaolin-activated, and tissue factor–activated), and 2) determine if there was a significant difference between 2 separate samples obtained from individual healthy cats on the same day. Jugular venipuncture was performed in 20 cats, and citrated blood collected for TEG. TEG analysis was performed on citrated native, kaolin-activated, and tissue factor–activated blood for each sample. Two hours later, the procedure was repeated from the opposite jugular vein, yielding a total of 120 analyses. Reaction time (
Introduction
Thromboelastography (TEG) is a point-of-care measure of hemostasis, and a kinetic determination of thrombus development, allowing assessment of rate and strength of clot formation, as well as subsequent clot lysis. 3 Thromboelastography evaluates the entire hemostatic process whereas traditional tests of hemostasis such as prothrombin time (PT), activated partial thromboplastin time (aPTT), activated clotting time (ACT), and platelet aggregation only measure isolated portions of the hemostatic pathway. 13 Another advantage of TEG is its ability to detect both hyper- and hypocoagulable states. 3 Thromboelastography has been used to identify hypercoagulability in dogs with parvoviral infections, disseminated intravascular coagulation, neoplasia, and immune-mediated hemolytic anemia,7,10,11,15 and was a better predictor of bleeding in dogs compared to PT and aPTT. 17
Several variables describing clot formation are recorded during TEG analysis. The reaction time (
Various sample types have been used for TEG analysis in veterinary and human medicine. The addition of an activator, such as tissue factor or kaolin, to whole blood has been reported to minimize variability and produce more rapid results in horses and human beings.4,5 Additionally, activated samples produced lower analytical variability compared to unactivated samples in a group of healthy cats. 9
While TEG is extensively used to detect hemostatic abnormalities in dogs,3,7,11,17few studies have reported the use of TEG in cats.1,9 The goals of the current study were to compare 3 TEG sample types in healthy cats and to determine the intraindividual variability of TEG measured at 2 different time points in individual cats.
Materials and methods
Twenty healthy cats, owned by students and staff at the Ontario Veterinary College, University of Guelph (Guelph, Ontario, Canada), were recruited for the study. Cats were not on any medications except parasite prophylaxis, and were determined to be healthy on the basis of a normal physical examination, complete blood cell count, serum biochemical profile, and coagulation profile. The study was designed in accordance with the standards of the Canadian Council on Animal Care and the Ontario Animals for Research Act and was approved by the University of Guelph Animal Care Committee.
A 5-ml blood sample was obtained from a single, atraumatic jugular venipuncture using a 22-gauge needle and a 6-ml syringe. Blood for TEG analysis was transferred into a 1.8-ml 3.2% sodium citrate tube, ensuring a 9:1 blood to citrate ratio. Tubes were gently inverted 5 times to allow mixing of the blood and citrate. Samples were allowed to equilibrate at room temperature for 30 min prior to TEG analysis. The procedure was repeated from the opposite jugular vein, approximately 2 hr after the first sample collection.
Thromboelastography analysis
Thromboelastography was performed using 2 thromboelastography analyzers
a
to allow for all 3 samples to be analyzed simultaneously. For every sample, 20 µl of calcium citrate was added to the pre-warmed (37°C) cup.
b
For the citrated native samples, 340 μl of citrated whole blood was then added to the cup. For kaolin-activated samples, 1 ml of citrated whole blood was poured into a kaolin-coated vial,
c
and 340 μl of this activated sample was then added to the cup. For tissue factor–activated samples, 20 μl of recombinant human tissue factor (1:50,000 dilution) stabilized in a HEPES (N-2-hydroxyethylpiperazone-N-2-ethanesulfonic acid) buffer with 2% bovine serum albumin
d
and 320 μl of citrated whole blood were added to the cup. Each sample was run for 75 min;
Statistical analysis
Paired
Results
The mean age of cats in the current study was 3.8 years (range: 1–10 years). Of the 20 cats, 19 were Domestic Short or Longhair cats and 1 was a Norwegian Forest Cat. Eleven were neutered males; 9 were spayed females.
There were no significant differences between first and second samples for each TEG variable (
Using a Levene test, there was significantly more variation for
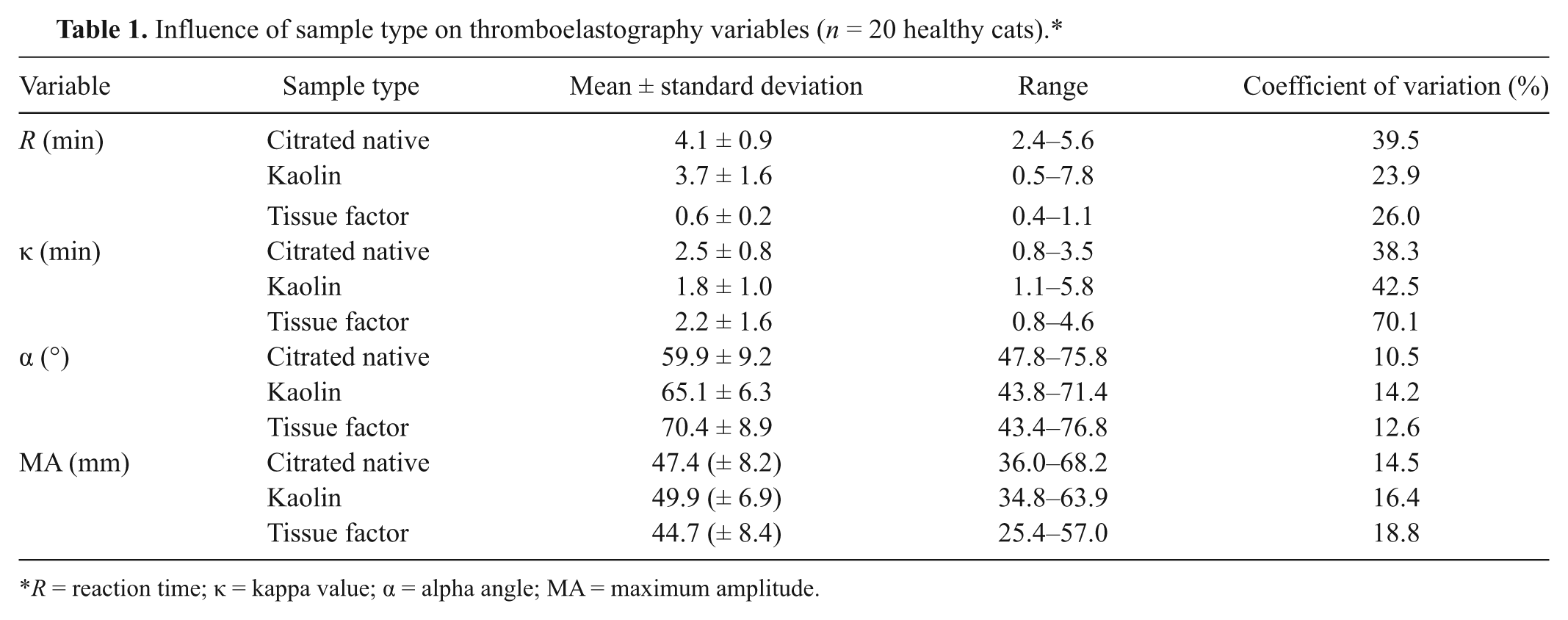
Influence of sample type on thromboelastography variables (
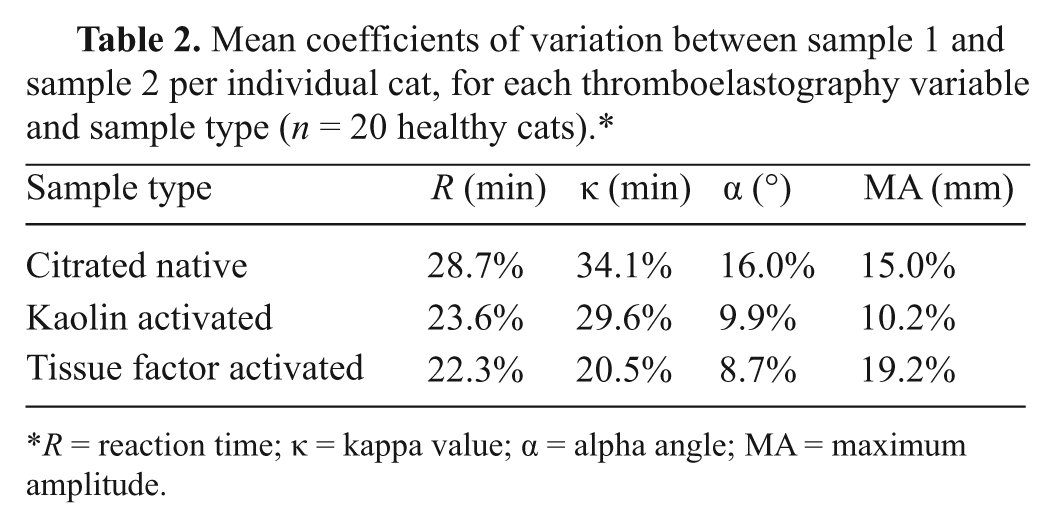
Mean coefficients of variation between sample 1 and sample 2 per individual cat, for each thromboelastography variable and sample type (
Discussion
Three different sample types for TEG analysis (citrated native, kaolin-activated, and tissue factor–activated) have been described previously in dogs and cats.1,2,9,10,16 In the current study, the 3 TEG sample types were performed twice on each cat using 2 separate samples acquired and analyzed approximately 2 hr apart. There were no significant differences between first and second samples for each TEG parameter for any of the 3 sample types, suggesting there is minimal biological variability for TEG variables in cats. Additionally, within-cat variability between 2 samples obtained at separate times ranged from 8.7% to 34.1% for each TEG variable.
Activators such as kaolin and tissue factor are used in other species to reduce the variability in TEG tracing as well as accelerating initiation of the coagulation process, providing more uniform and faster TEG results.4,5 Kaolin (hydrated aluminum silicate) initiates coagulation via the contact activator pathway. Ready-to-use kaolin vials make the use of this activator convenient and consistent.2,5 Tissue factor is a stimulus of in vivo coagulation, making it the preferred activator in some laboratories. However, recombinant tissue factor must be diluted from stock solution, potentially introducing a source of variability in TEG analysis.4,5,16 The
Kaolin activation produced the most variability with respect to measurement of
One limitation of the present study was the small number of cats used (20). It is recommended that samples from at least 40 individuals are obtained when generating a reference interval. 12 The current study used lower than optimal numbers for generation of a reference interval, but may still provide a useful interval for interpretation of clinical data. Most cats included in the study were Domestic Short and Longhair, and therefore may not be universally applicable to other breed populations. It has been shown that Greyhound dogs have significantly different TEG values from other breeds, 14 and the effects of breed on TEG should be investigated in cats.
No statistically significant differences were found for TEG variables of individual cats measured over 2 separate samples. Further research evaluating day-to-day variability of TEG, or repeatability between 3 or more samples should be performed to evaluate the consistency of TEG measurements in cats. The current study established TEG variables for
Footnotes
Acknowledgements
The authors thank Gabrielle Monteith for her help with statistical analysis.
a.
TEG® 5000 Thrombelastograph Hemostasis Analyzer, Haemonetics Corp., Braintree, MA.
b.
TEG® Hemostasis System Cups and Pins, Haemonetics Corp., Braintree, MA.
c.
TEG® Hemostasis System Kaolin, Haemonetics Corp., Braintree, MA.
d.
Innovin®, Dade-Behring, Newark, DE.
e.
SAS version 9.1, SAS Inc., Cary, NC.
f.
GraphPad Prism version 5.0, GraphPad Software Inc., San Diego, CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This work was supported by the Ontario Veterinary College Pet Trust Fund (grant no. 49853).
