Abstract
Neospora caninum is a protozoan parasite that causes abortion and important economic losses in cattle worldwide. There are no treatments or vaccines available; disease control is based on diagnosis and herd management strategies. We developed, validated, and evaluated under field conditions a competitive inhibition ELISA based on the truncated SAG1 protein (tSAG1), expressed in Escherichia coli, and the RafNeo5 monoclonal antibody (ciELISAtSAG1). A criterion based on the 3-y sequential serologic analysis of 230 dairy cows by IFAT was used as the gold standard. The assay was validated using 860 serum samples from cows that were consistently positive or negative by IFAT throughout the study period. ciELISAtSAG1 was then used to evaluate the prevalence of neosporosis in 16 beef cow herds (22 samples per herd, 352 total samples). The results were compared with those from IFAT and a commercial cELISA (cELISAVMRD). The ciELISAtSAG1 cutoff was ≥ 29%I, with a diagnostic sensitivity of 98.7% (95% CI = 96.8–99.7%) and a diagnostic specificity of 97.9% (95% CI = 96.4–99.0%). Concordance among IFAT, cELISAVMRD, and ciELISAtSAG1 was 90.3%. The agreement (κ) between ciELISAtSAG1 and the other 2 tests was ≥ 0.81. The overall prevalence of neosporosis in the 16 beef herds was 30% (range: 5–60%). The ciELISAtSAG1 could be useful for large-scale detection of anti–N. caninum antibodies in cattle and seroepidemiologic investigations, given its appropriate sensitivity and specificity, and the simplicity of production.
Introduction
Bovine neosporosis is a parasitic disease caused by the apicomplexan protozoan Neospora caninum (Eucoccidiorida: Sarcocystidae) and characterized by producing abortions in cows and neuromuscular lesions in calves.4,14,16 The disease is distributed globally and causes severe economic losses to the cattle industry. 27 Cattle, an intermediate host, become infected through the ingestion of N. caninum oocysts shed in the feces of canids, the definitive hosts, or transplacentally from the infected dam to the fetus. 17 The parasites remain quiescent in tissue cysts, avoiding the host immune response and hence being undetectable by serologic tests. 20 To date, there is no effective chemotherapeutic treatment or vaccine for prevention of neosporosis, with partial control being achieved through management strategies based on efficient detection tests. 28
The immunofluorescent antibody test (IFAT) is still used extensively to detect anti–N. caninum antibodies in cattle, although it is a laborious technique and interpretation of results is subjective. Various ELISAs are available and are replacing IFAT for large-scale seroepidemiologic studies. Most commercial ELISAs are based on tachyzoite lysates. 2 Given that an ELISA based on native tachyzoite antigens may detect antibodies induced by other members of the Sarcocystidae family, 19 recombinant antigens from dense granules 23 or tachyzoite surface antigens11,13,30 have been used to develop various indirect ELISAs (iELISAs). Some surface antigens of apicomplexans are efficiently recognized by the host immune system and are strong inducers of antibodies. Therefore, the recombinant forms of surface antigens are optimal candidates to develop more specific assays. Furthermore, the use of recombinant antigens instead of native tachyzoite antigens facilitates production and improves technique standardization.
A competitive ELISA (cELISA) generates less background than other ELISAs because, with the exception of the anti-mouse antibody, no species-specific conjugates are needed. Therefore, this test can detect antibodies to different species. 9 There is a commercial cELISA based on the immunodominant 65-kDa native surface protein from N. caninum tachyzoites and the monoclonal antibody (mAb) 4A4-2. 6 A cELISA based on the soluble extract of purified tachyzoites and the RafNeo5 mAb (cELISAtach), which recognizes the major surface antigen (SAG1) from N. caninum tachyzoites, has been validated (Valentini BS, et al. Bovine neosporosis: infection status defined by IFAT and cELISA in serum samples obtained periodically from dairy cows. Proc XXI Scientific-Tech Meeting Arg Assoc Vet Diagn Lab; Oct 2016; San Salvador de Jujuy; Argentina). SAG1 protein is immunodominant and conserved among isolates; 21 therefore, it is a solid candidate antigen for ELISA development.
One of the drawbacks of the expression of eukaryotic proteins in prokaryotic cells is the formation of protein aggregates commonly referred to as inclusion bodies. 3 In previous work, 31 SAG1 protein was expressed as inclusion bodies in Escherichia coli and refolded to develop an applied printing immunoassay. Another research group 13 expressed a variant of recombinant SAG1 protein lacking the signal peptide and the hydrophobic c-terminal portion in E. coli as a glutathione-S-transferase (GST) fusion protein, improving its solubility.
We describe herein the development, validation, and field evaluation of a competitive inhibition ELISA based on a soluble truncated variant of SAG1 (tSAG1) and the RafNeo5 mAb (ciELISAtSAG1) for the detection of anti–N. caninum antibodies in cattle.
Materials and methods
Parasites
Tachyzoites of the NC-1 strain of N. caninum 15 were maintained in vitro by continuous passage in Vero (African green monkey kidney) cell culture. The monolayer was scraped from the flask when 80% of the cells were infected. 5 Tachyzoites were released by passing the parasitized cells through a 25-gauge needle twice 7 and purified from detritus on a gradient (Percoll; MilliporeSigma, Darmstadt, Germany). 29
Cattle sera
To validate the ciELISAtSAG1, 871 sera from 230 cows sampled from a dairy herd over a 3-y period (average re-sampling interval of 7 mo) were selected from the serum bank of the Laboratory of Veterinary Immunology and Parasitology (LIPVet) of INTA Rafaela, Santa Fe, Argentina (Table 1). The selected herd located in Córdoba province had periodic abortions caused by N. caninum and a low frequency of cow replacement, which allowed us to obtain periodic serum samples. A criterion based on the 3-y sequential serologic analysis by IFAT of samples from dairy cows was used as the gold standard. Cows were considered infected if they were positive in 2 or more samplings, and uninfected if they were negative in all samplings; those with a single positive sample were excluded from the validation study.
Bovine neosporosis: serum samples obtained from dairy cows over a 3-y period.

For the field evaluation of the ciELISAtSAG1, serum samples of 352 beef cows, from 16 herds of the littoral area of Santa Fe province, were obtained and stored at −20°C until analysis. Given that the level of neosporosis in this region was unknown, the number of samples (22 per herd) was used for an expected herd prevalence of 90% and adult cattle prevalence of 15%, with a 95% confidence interval (CI) and a 15% error (WinEpiscope, http://www.winepi.net/).
The negative control serum (C–) was obtained from a steer considered uninfected after 3 serum samplings taken at 30-d intervals, analyzed by IFAT and iELISA. 12 The same steer was then inoculated intravenously with 1 dose of 108 live tachyzoites of N. caninum. The weak positive control (C+) and the strong positive control (C++) sera were obtained after bleeding the steer 21 and 42 d after inoculation, respectively; sera were stored in aliquots at −20°C until use.
Sera from animals infected with Toxoplasma gondii and Sarcocystis cruzi, closely related apicomplexan parasites, were analyzed to study cross-reactivity with tSAG1 protein. We used sera from a cow experimentally infected with 107 tachyzoites of T. gondii, obtained 35 d after experimental inoculation. T. gondii infection was confirmed by IFAT. Sera from a cow with natural S. cruzi infection, kindly provided by Dr. Lucía Campero and Dr. Gaston Moré from the Immunoparasitology Laboratory of the Veterinary Sciences Faculty, National University of La Plata, Argentina, was also analyzed. Sarcocystis infection was detected using the visualization of tissue cysts in the heart via histologic examination. 2 These samples tested negative to N. caninum antibodies by IFAT.
All animals used in our study were handled in strict accordance with good animal practice and the conditions defined by the Animal Ethics Committee of the Veterinary Science Faculty of the Littoral National University, Esperanza, Santa Fe, Argentina (license 226/15, file 20,390).
Monoclonal antibody
RafNeo5 mAb reactive against SAG1 was selected from a group of mAbs obtained previously at the LIPVet by immunization of mice with a soluble fraction of sonicated tachyzoites (NcSf; https://inta.gob.ar/documentos/neosporosis-bovina-estatus-de-infeccion-definido-por-ifi-y-celisa-en-muestras-de-suero-obtenidas-periodicamente-de-vacas-lecheras).
Cloning of truncated SAG1 gene from N. caninum
Genomic DNA (gDNA) was extracted from tachyzoites using a standard phenol–chloroform–isoamyl alcohol method. DNA encoding amino acid residues 22–303 of the SAG1 gene was amplified by PCR using the primers tSAG1-F (5′-CATATGGCGTTCTTTGACTGTG-3′) and tSAG1-R (5′-GGATCCTCAGTGATGGTGATGGTGATGCCGGCCTTCAAT-3′), which contain introduced NdeI and BamHI sites to facilitate cloning. The tSAG1-R primer contains an introduced poly-histidine sequence to facilitate protein purification. Subsequently, tSAG1 was cloned into pGEM-T Easy (Promega, Madison, WI). The tSAG1 gene fragment was excised with NdeI and BamHI and subcloned into pET-9b (Novagen; MilliporeSigma) to be expressed as a polyhistidine-tagged fusion protein in E. coli. The identity of the DNA construct was confirmed by sequencing (Institute of Biotechnology, INTA CICVyA, Buenos Aires, Argentina).
Protein expression and purification
E. coli BL21 RIL (DE3) pLysS competent cells (Novagen; MilliporeSigma) transformed with pET9b/tSAG1 plasmid were cultured at 37°C in 500 mL of lysogeny broth medium supplemented with 50 μg/mL kanamycin and 34 μg/mL chloramphenicol to an optical density at 660 nm (OD600 nm) = 1. Protein expression was induced with 1% lactose. After 3 h of incubation at 37°C, bacteria were harvested by centrifugation and suspended in 10 mL of lysis buffer (50 mM NaH2PO4, 300 mM NaCl, 10 mM imidazole, pH 8) containing 1:1,000 of protease inhibitor cocktail set III (Calbiochem; MilliporeSigma). The cell suspension was lysed by 2 passes through a cell disruptor at 20,000 psi (Emulsiflex B15; Avestin, Ottawa, Ontario, Canada) and centrifuged (12,000 × g, 30 min, 4°C). The soluble fraction was separated and added to 2 mL of Ni-NTA agarose (Qiagen, Hilden, Germany) previously equilibrated with lysis buffer. After incubation at 4°C for 1 h, the suspension was poured into a 1.5 × 5.0 cm column and washed with 5 volumes of lysis buffer containing 30 mM imidazole. Bound tSAG1 was eluted with 5 volumes of 200 mM imidazole lysis buffer. Finally, the buffer was exchanged into phosphate-buffered saline (PBS; 10 mM Na2HPO4, 1.8 mM KH2PO4, 137 mM NaCl, 2.7 mM KCl, pH 7.4) by dialysis. Molar concentration in pure samples was calculated by absorbance at 280 nm using a molar extinction coefficient (ε280 nm) equal to 27,220/M/cm.
Western blot
An uninduced and an induced transformed E. coli culture, purified tSAG1, NcSf, and a T. gondii tachyzoite lysate were electrophoresed in a sodium dodecyl sulfate (SDS)–12% polyacrylamide gel electrophoresis (PAGE) and transferred to an adsorbent nitrocellulose membrane (Trans-Blot transfer medium; Bio-Rad Laboratories, Hercules, CA). The membrane was incubated with Tris-buffered saline (TBS; 50 mM Tris-HCl, 150 mM NaCl, pH 7.6) containing 5% w/v of skimmed milk (TBS-M; Svelty; Nestlé, Buenos Aires, Argentina). After 3 washes with TBS containing 0.05% v/v of Tween-20 (TBS-T), the membrane was incubated with RafNeo5 mAb diluted 1:100 in TBS-M for 1 h. Then, the membrane was washed as described previously; bound RafNeo5 mAb was detected with peroxidase-conjugated goat anti-mouse immunoglobulin (Jackson InmunoResearch Laboratories, West Grove, PA) diluted 1:1,000 in TBS-M. After incubation and 3 washes, the antigen–antibody reaction was revealed with the colorimetric substrate 3,3’-diaminobenzidine tetrahydrochloride (DAB; MilliporeSigma) and 0.1% v/v of hydrogen peroxide (H2O2).
A western blot (WB) was performed to study cross-reactivity of antibodies against other apicomplexan parasites to SAG1. Purified tSAG1 was used as antigen under non-reducing conditions, separated by electrophoresis in a SDS-PAGE, and transferred to an adsorbent nitrocellulose membrane (Trans-Blot transfer medium; Bio-Rad). Serum samples from T. gondii, S. cruzi, and N. caninum infected cattle and a negative control serum were diluted 1:100. A conjugate anti-bovine IgG peroxidase (Jackson ImmunoResearch) diluted 1:2,000 in TBS-M was used. The reaction was revealed as described above.
Indirect fluorescent antibody test
Purified whole tachyzoites were used for IFAT according to the technique described previously, with modifications. 25 Briefly, purified tachyzoites were washed 3 times with PBS and fixed with 0.025% v/v 37% formalin for 16 h. A suspension of tachyzoites in PBS (5 μL) was distributed in 12-well heavy-Teflon–coated slides (Redilab, Humberto Primo, Santa Fe, Argentina). Slides were dried at 37°C and stored at −20°C until use. Test sera dilution 1:200 was considered as the cutoff, 6 and the goat anti-bovine IgG fluorescein isothiocyanate-conjugate (MilliporeSigma) was diluted 1:800 in PBS. Slides were observed under an epifluorescence microscope (Eclipse 80i; Nikon, Melville, NY). The fluorescence over the whole tachyzoite surface was considered positive, and nonspecific (negative) when it was apical. 9
Commercial competitive ELISA
A commercial cELISA (cELISAVMRD; VMRD, Pullman, WA) was used following the manufacturer’s instructions. The antigen of cELISAVMRD is a N. caninum surface protein (GP65) captured from a lysate of tachyzoites by a mAb. The suggested ≥ 30% inhibition cutoff was used.
Competitive inhibition ELISA based on tSAG1 and RafNeo5 mAb (ciELISAtSAG1)
The 96-well ELISA plates (Nunc; Thermo Fisher Scientific, Waltham, MA) were coated with 1 μg/well of tSAG1 diluted in PBS to a concentration of 0.02 μg/μL at 4°C for 16 h. The wells were washed twice with PBS containing 0.05% Tween 20 (PBS-T) and blocked with PBS containing 10% skimmed milk (PBS-M). The test sera, diluted 1:2 in PBS containing 0.05% Tween 20 and 10% skimmed milk (PBS-T-M), were added to duplicate wells. After incubation, the RafNeo5 mAb diluted 1:2,000 in PBS-T-M was added. Finally, horseradish peroxidase–conjugated goat anti-mouse antibody (Jackson ImmunoResearch) diluted 1:4,000 in PBS-T-M was added. All incubations were carried out for 40 min at 37°C. Plates were washed 3 times with PBS-T between steps. The antigen–antibody reaction was revealed with 100 μL of 1 mM 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS) chromogenic substrate (MilliporeSigma) in 0.05 M sodium citrate (pH 4.5) and 0.03% v/v of H2O2, and the plates were incubated in the dark for 10 min. Each plate included controls of known positive strong and weak bovine sera (C++ and C+, respectively), known negative bovine sera (C–), and RafNeo5 mAb without serum (Cc) to determine its maximal OD405 nm. The color of the reaction was measured at 405 nm in an ELISA plate reader (Labsystems Multiskan FC; Microlat, Buenos Aires, Argentina). Results were expressed as percent inhibition (%I) according to the formula %I = 100 – [(sample OD/Cc OD) × 100].
Data analysis
A receiver operating characteristic analysis (MedCalc v.13.0; MedCalc Software, Ostend, Belgium) was performed to determine the optimal cutoff value, diagnostic sensitivity (DSe), and diagnostic specificity (DSp), with 95% CI for ciELISAtSAG1.
A retrospective analysis of neosporosis infection status in dairy herd throughout the 3-y study was performed by comparing IFAT and ciELISAtSAG1.
The prevalence of neosporosis in beef herds and cattle was analyzed. Concordance (%) among IFAT, cELISAVMRD, and ciELISAtSAG1 and kappa values (κ) between pairs of tests, with 95% CI, were evaluated using MedCalc software. Strength of agreement was considered poor (κ ≤ 0.20), fair (κ = 0.21–0.40), moderate (κ = 0.41–0.60), good (κ = 0.61–0.80), or very good (κ = 0.81–1.00). 1
Results
Expression and purification of tSAG1 recombinant protein
tSAG1 recombinant protein was expressed with high efficiency in E. coli; ~ 30% of the recombinant protein was soluble in the bacterial cytoplasm. The amount of purified protein was ~ 5 mg/L of culture. The c-terminal his-tag added to tSAG1 allowed us to purify large amounts of protein in a single step with a final purity > 95%. After electrophoretic separation, purified tSAG1 migrated as a thick band between 25 and 37 kDa markers and a weak band between 50 and 75 kDa markers. The molecular weights obtained for the bands correspond to the expected monomeric and dimeric forms of tSAG1 (Fig. 1A).
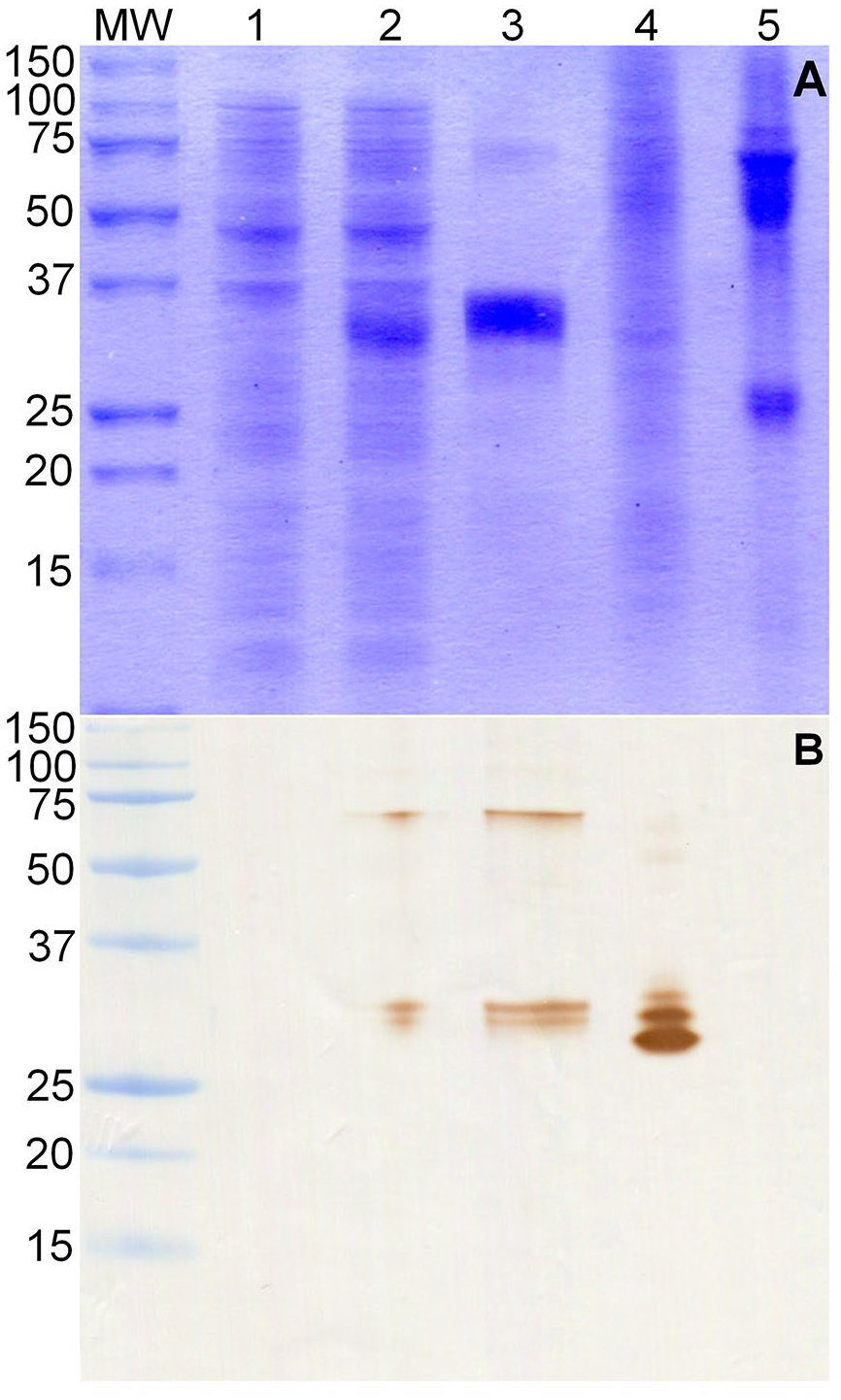
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and western blot (WB) analysis of recombinant tSAG1.
The WB assay revealed RafNeo5 mAb binding to tSAG1 recombinant protein in its monomeric and dimeric forms. This assay also showed that the thick band observed at SDS-PAGE is composed of 2 protein bands. The RafNeo5 mAb bound to sonicated N. caninum tachyzoites and revealed monomer and dimer forms of native SAG1 protein. In this case, the monomer is composed of 3 nearby protein bands. The RafNeo5 mAb did not bind to sonicated T. gondii tachyzoites (Fig. 1B).
Cross-reactions
No antigen–antibody reactions were detected in the WB performed with tSAG1 as antigen and sera from animals infected with T. gondii and S. cruzi (Fig. 2).
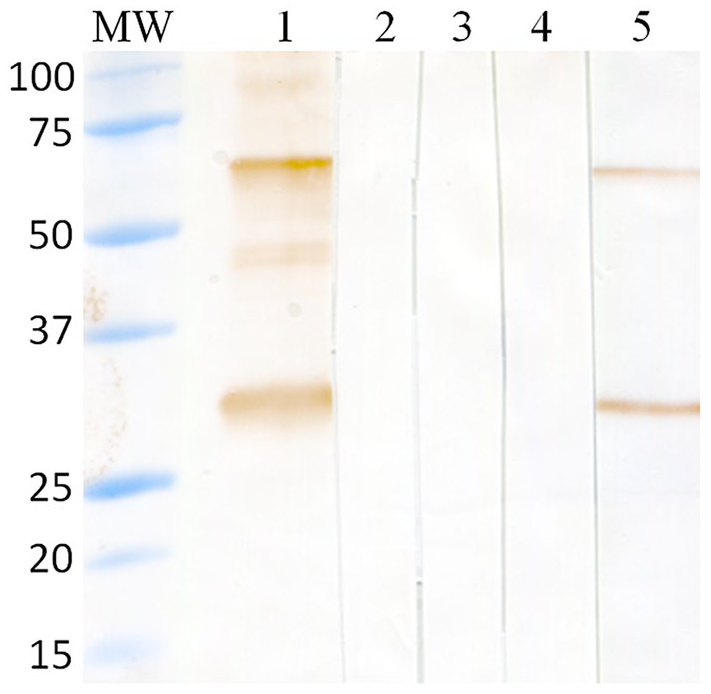
Western blot analysis of the recognition of tSAG1 antigen using sera from animals infected with Neospora caninum and closely related apicomplexan parasites. MW = molecular weight marker (Precision Plus protein all blue pre-stained protein standards; Bio-Rad); lane 1 = sera from a cow experimentally infected with N. caninum; lane 2 = sera from a steer experimentally infected with Toxoplasma gondii; lane 3 = sera from a cow naturally infected with Sarcocystis cruzi; lane 4 = sera from an uninfected steer; and lane 5 = RafNeo5 monoclonal antibody.
Validation of ciELISAtSAG1
Of the 871 serum samples from 230 selected dairy cows, 860 serum samples from 227 cows were used for test validation. Eighty-five cows were classified as infected (314 sera) and 142 as uninfected (546 sera). Three cows with only one IFAT-positive sample were excluded from validation; 1 of them (cow 509) was ciELISAtSAG1 positive in 3 samples and was categorized as infected. The status of neosporosis infection for the other 2 cows (533 and 6,842) remained undefined (Table 2).
Identification of dairy cows in which antibody fluctuation was detected in 2–5 samplings over a 3-y period, analyzed by IFAT and ciELISAtSAG1. Cutoffs were the serum dilution of ≥ 1:200 for IFAT and ≥ 29%I for ciELISAtSAG1.
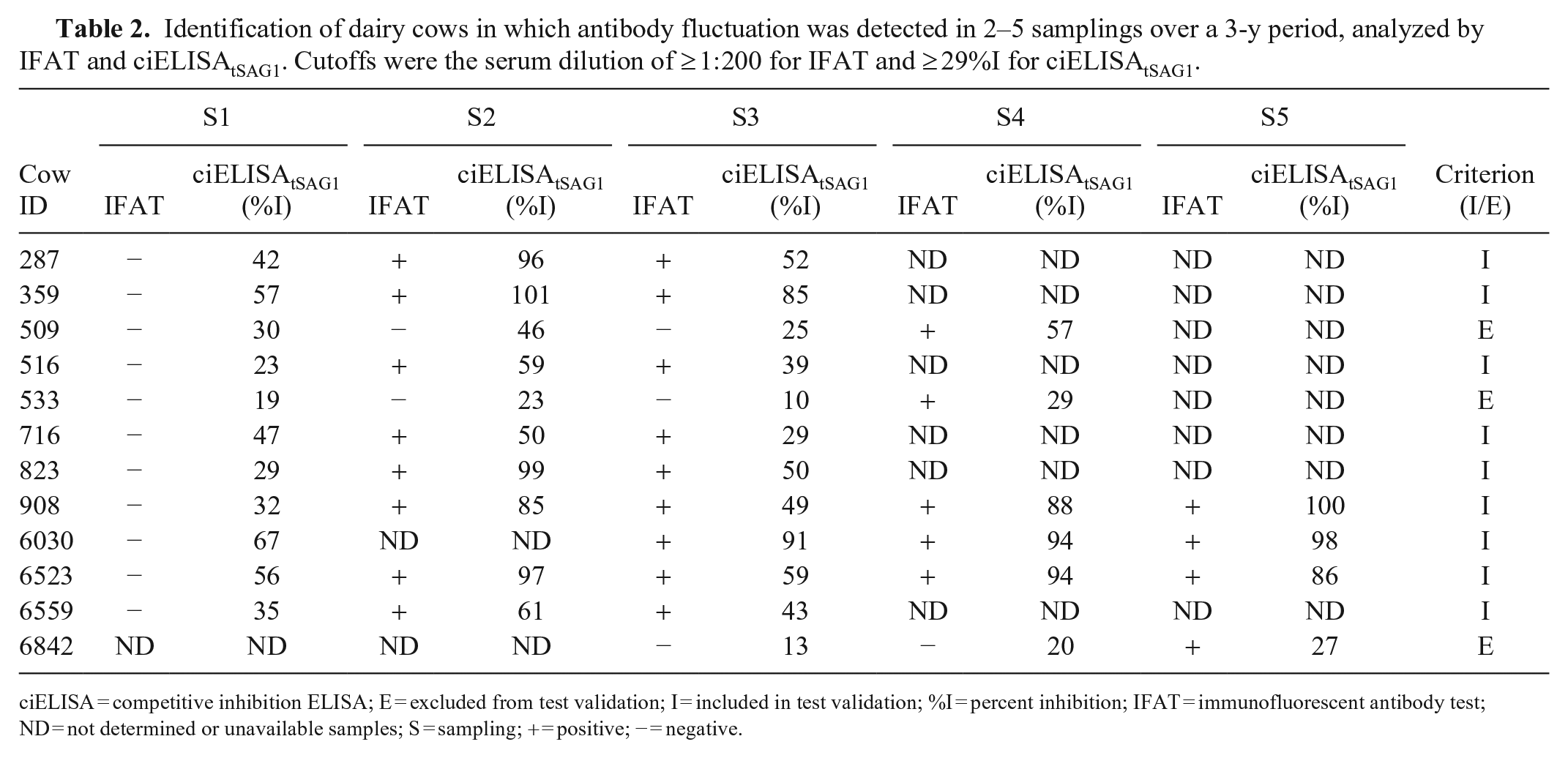
ciELISA = competitive inhibition ELISA; E = excluded from test validation; I = included in test validation; %I = percent inhibition; IFAT = immunofluorescent antibody test; ND = not determined or unavailable samples; S = sampling; + = positive; − = negative.
The ciELISAtSAG1 cutoff was ≥ 29%I, with a DSe of 98.7% (95% CI = 96.8–99.7%) and a DSp of 97.9 (95% CI = 96.4–99.0%). The mean %I was 83% (± 19%) for the infected and 13% (± 9%) for uninfected cows (Fig. 3).
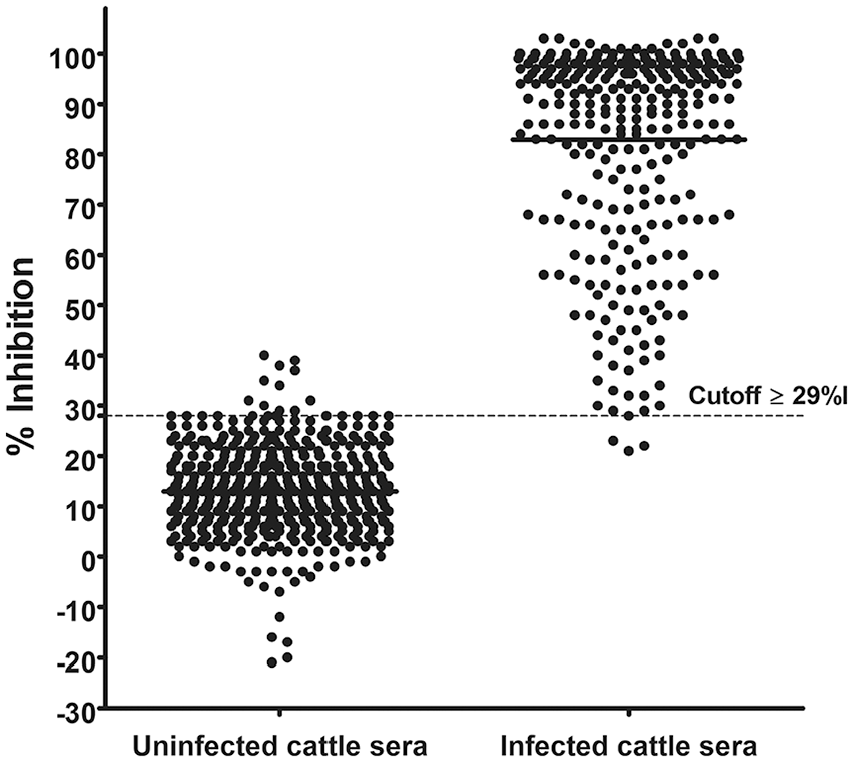
Receiver operating characteristic (ROC) analyses of the competition inhibition ELISAtSAG1 for diagnosis of neosporosis in cattle. Determination of the cutoff using 546 serum samples from 142 uninfected cows and 314 serum samples from 85 infected cows. Results are expressed as percent inhibition (%I).
The retrospective analysis of the status of neosporosis infection over 3 y showed that 9 of the 227 cows had variable results among samplings (Table 2). Of 227 cows present in the first sampling, 61 remained in the herd throughout the study period and maintained their original neosporosis infection status. Concordance between IFAT and ciELISAtSAG1 throughout the study was 98.1% with κ = 0.96 (95% CI = 0.94–0.98).
Field evaluation
Anti–N. caninum antibodies were detected by IFAT, cELISAVMRD, and ciELISAtSAG1 in all 16 analyzed herds. The overall prevalence of neosporosis was 30% by IFAT and ciELISAtSAG1, and 36% by cELISAVMRD, with a range of 5–60% among herds. In 10 of the 16 analyzed herds, the prevalence established by IFAT and ciELISAtSAG1 was identical. Compared with IFAT and ciELISAtSAG1, the prevalence detected by cELISAVMRD was higher in 14 herds and identical in 2 herds (Fig. 4).
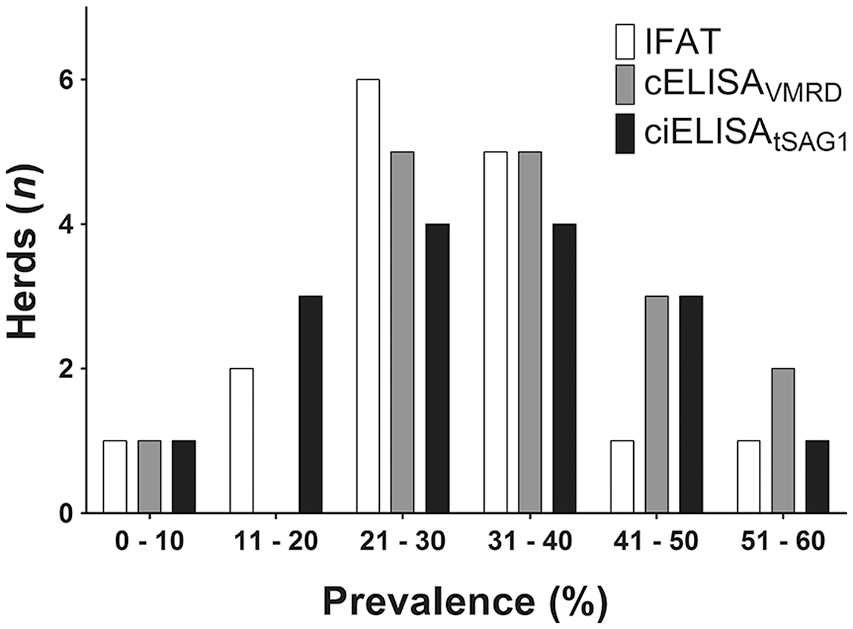
Frequency distribution of neosporosis prevalence in 16 beef herds analyzed using immunofluorescent antibody test (IFAT), competitive (c)ELISAVMRD, and competitive inhibition (ci)ELISAtSAG1.
Analysis of the 352 serum samples from beef cattle revealed 89.8% concordance among IFAT, cELISAVMRD, and ciELISAtSAG1 (Fig. 5). The highest and lowest agreement between pairs of tests was observed between IFAT and ciELISAtSAG1, and between IFAT and cELISAVMRD, respectively (Table 3).
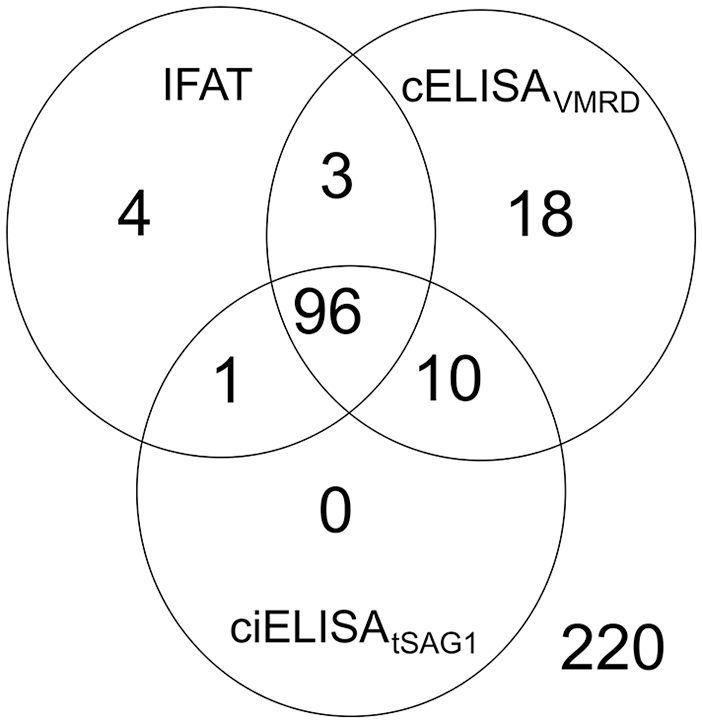
Integrated results of antibodies against Neospora caninum in 352 beef cattle sera obtained by immunofluorescent antibody test (IFAT), competitive (c)ELISAVMRD, and competitive inhibition (ci)ELISAtSAG1.
Concordance and agreement (κ) between pairs of tests used to analyze 352 beef cattle sera.

CI = confidence interval; cELISA, ciELISA = competitive and competitive inhibition ELISA, respectively; IFAT = immunofluorescent antibody test.
Discussion
Our ciELISAtSAG1 was able to differentiate N. caninum–infected cattle from uninfected ones, even under the demanding conditions required by the gold standard, based on a 3-y sequential analysis of samples using IFAT. The performance of ciELISAtSAG1, with a DSe of 98.7% and a DSp of 97.9%, was similar to that of other commercial serologic assays. 2
IFAT has been used frequently as a reference test to determine the relative sensitivity and specificity of ELISA.6,8,26,32 This comparison criterion adds a cumulative error to the sensitivity and specificity of the new assay and underestimates its value. 22 The gold standard that we used allowed us to include infected cows with negative IFAT results. The main reason to use this strategy was to identify infected cattle in which parasites remain hidden inside tissue cysts. With this mechanism, the parasite avoids the immune response and remains undetectable by serologic tests. 20
The ciELISAtSAG1 detected 8 of 9 infected cows in which antibody fluctuation was detected by IFAT; that finding may indicate that the sensitivity of the cELISAtSAG1 is greater than that of the IFAT in chronically infected cattle. In addition, of 3 cows with positive IFAT in 1 sample (excluded from the validation), our ciELISAtSAG1 identified 3 of 4 samples of cow 509 as positive, highlighting the greater sensitivity of our test. It is expected that the ciELISAtSAG1 expresses a better DSp, given that we did not detect cross-reactions of tSAG1 with sera from T. gondii– and S. cruzi–infected cattle.
Most serologic assays, such as IFAT and ELISA variants, use whole or lysed tachyzoites of N. caninum.2,12 Although these assays allow the differentiation of infected from uninfected cattle, all of them have some degree of inherent limitation in sensitivity and specificity. The cytoplasmic antigens of the parasite included in the lysates can lead to cross-reactions with other related parasites (Sarcocystidae family); 19 therefore, greater dilution (1:100 or 1:200) of the serum is required. 9 The use of a N. caninum recombinant antigen for ciELISAtSAG1 development improved the specificity of this test, given that tSAG1 showed no cross-reactivity with sera from animals infected with closely related apicomplexan parasites. Furthermore, recombinant antigens have the benefit that their production does not depend on the maintenance of cell culture, given that they can be produced easily in large quantities and the produced antigen is homogeneous in different production batches. This improves the standardization of antigen production for test kit development. Sera for use in ciELISAtSAG1 can be diluted 1:2, which favors the sensitivity of the test. The lower IFAT sensitivity detected could be a result of the cutoff used (1:200) based on a previous validation. 10 A lower dilution might increase the sensitivity but would reduce the specificity. 9 Our retrospective analysis of this dairy herd suggests that the infection by neosporosis was mainly transplacental, given that 61 cows that remained in the herd throughout the study period maintained their original infected or non-infected status with absence of seroconversion.
The overall prevalence (30%) detected by ciELISAtSAG1 in beef cattle was higher than values estimated in other regions of Argentina. A seroepidemiologic study using IFAT in the southeast (37°50′47″S) of the humid Pampas of Argentina 24 found an average prevalence of 11.2% (4.7–20.3%). Another study 18 using an iELISA found a 9.6% (± 10.5%) prevalence in La Pampa province (36°31′00″S), Argentina. In these areas, British-origin cattle predominate. The higher prevalence in beef cattle of Santa Fe province could be associated with the breeds (Bos indicus hybrids), climate (higher temperatures), geographic conditions (waterlogged areas), neosporosis infection status of dogs present on the farms, and management system (confinement and supplemental feeding during floods of the littoral zone). 17
cELISAVMRD results differed from IFAT and ciELISAtSAG1 and showed the highest neosporosis prevalence value in beef cattle (36%). This difference might be a consequence of the cutoff used (manufacturer’s protocol), given that no revalidation was performed for local epidemiologic conditions as recommended by the OIE for validated tests used in new regions. 33 A comparative study of commercial ELISAs 2 had a κ value < 0.67 for the cELISAVMRD when compared with a gold standard of “majority of tests” prior to cutoff adjustment.
The polarized distribution of the positive and negative results in ciELISAtSAG1 allows an increase or decrease of the cutoff under different epidemiologic situations, without significantly affecting the DSe and DSp. 34 The ciELISAtSAG1 is useful for a large-scale survey of neosporosis in cattle and seroepidemiologic investigations, given its appropriate sensitivity and specificity, and the simplicity of production. Furthermore, given that no species-specific conjugated antibodies are needed, except for the secondary anti-mouse antibody, this test can likely detect antibodies against N. caninum in different hosts once the required validation is performed. 9
Footnotes
Acknowledgements
We thank Paula Ramirez, Estefania Ugarte, and Nerina Aguirre from LIPVet for their technical assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was supported by INTA, Asociación Cooperadora INTA Rafaela, and Agencia Nacional de Promoción Científica y Tecnológica (PICT0369).
