Abstract
We identified a novel herpesvirus in a captive juvenile white stork (Ciconia ciconia) that experienced progressive weight loss followed by death. Histologic findings included severe splenic and hepatic necrosis, and intranuclear inclusion bodies in hepatocytes and unidentified splenic cells. The virus was amplified, sequenced, and subsequently accessioned as Ciconiid alphaherpesvirus 1. Phylogenetic analysis was performed and revealed that this virus is more closely related to mammalian herpesviruses than those within the genus Mardivirus. Additional sequence of viruses in this area may elucidate the ancestral virus that jumped from reptilian to mammalian hosts.
Keywords
Herpesviridae is a family of viruses that causes disease in diverse animal hosts, including avian species. Viruses within this family are large, linear, double-stranded DNA viruses that range from 125 to 290 kbp. 3 Herpesviruses display strong host specificity and are thought to evolve with the hosts they infect. Clinical signs of infected animals are extremely variable, and often nonspecific, but tend to be mild in adult endemic hosts and fatal in young and aberrant hosts.9,15 Although disease manifestations vary, infections with avian herpesviruses are often microscopically characterized by multi-organ necrosis with intranuclear inclusion bodies. 1
The white stork (Ciconia ciconia) is a migratory bird within the family Ciconiidae that is found in parts of Europe and Africa. In the wild, global population size is estimated to be 700,000–704,000 individuals (Wetlands International, http://wpe.wetlands.org/search?form[species]=ciconiaciconia&form[population]=&form[publication]=5). This species was listed as “least concern” by the IUCN Red List in 2016, although population threats, such as habitat alteration, a shortage of nesting sites, collisions with powerlines, and hunting, continue to exist (http://dx.doi.org/10.2305/IUCN.UK.2016-3.RLTS.T22697691A86248677.en).
Herpesviruses in the white stork have been reported in European populations. In one study, 12 herpesvirus isolates were obtained via cell culture from storks maintained in open aviaries in northern Germany. 11 No evidence of clinical disease attributable to herpesvirus infection was identified in these cases. Another study evaluated lesions associated with herpesvirus infection in 6 juvenile white storks in Europe. Gross and histologic lesions included hemorrhagic enteritis, hepatic and splenic necrosis, and intranuclear inclusions within multiple tissue sections. 6 Additionally, herpesvirus isolates were obtained from 83 of 253 white storks in a study conducted from 1983 to 2001 at 4 rehabilitation centers within northern Germany. 10 No herpesvirus-related lesions were identified in these cases, and no sequence data are available for any of these viruses. To date, no studies have isolated a herpesvirus in captive white storks within North America, to our knowledge.
We describe herein a fatal herpesvirus infection that manifested as severe hepatic and splenic necrosis in a captive juvenile white stork in the United States. A 5-mo-old, captive-hatched, white stork was observed to be lethargic, breathing heavily, and did not react upon approach by keepers. Over a 3-wk interval, the stork had lost 0.4 kg, 14% of its body weight, and it died upon arrival at the treatment facility. Postmortem examination was performed a few hours after death. Liver, spleen, intestine, lung, brain, kidney, skeletal muscle, esophagus, proventriculus, ventriculus, myenteric ganglia, trachea, heart, great vessels, adipose tissue, and eye were fixed in 10% neutral-buffered formalin, processed routinely, and sections were stained with hematoxylin and eosin (H&E). Sections of lung and intestine were additionally stained with Gram stain.
Gross postmortem findings included luminal thick, gray, material in the proventriculus and ventriculus consistent with ingested mud. The liver was speckled green and brown, and the spleen was tan to dark brown with purple speckling (Figs. 1, 3). No additional abnormalities were noted at postmortem examination. Histologic evaluation revealed coalescing periportal-to-random foci of acute hepatocellular lytic necrosis. These regions were associated with intranuclear eosinophilic inclusions within hepatocytes (Fig. 2). Near-complete necrosis of the spleen was present, with fibrin deposition associated with numerous unidentified cells that contained large intranuclear inclusions similar to those described in the liver (Fig. 4). One segment of the small intestine had erosion and pseudomembrane formation associated with bacterial overgrowth. Herpesviral inclusions were not seen in mucosal epithelial cells. Parabronchi of the lung were suffused with extravasated blood, and a bacterial embolus was present within an interstitial capillary. The bacteria in the enteritic lesion were large gram-positive bacilli with morphology and tinctorial properties consistent with Clostridium spp. Bacteria within the pulmonary embolus were small bacilli on standard H&E staining, additionally consistent with Clostridium spp.; however, absence of this embolus on subsequent Gram staining precluded thorough assessment of the bacterial population at this location. A small focus of perivascular hemorrhage and necrosis was present within the cerebrum. A few small foci of mineralization were present within myofibers of skeletal muscle. Within the kidney, a few tubules were dilated and contained protein casts; low numbers of tubules were lined by necrotic epithelium. The esophagus, proventriculus, ventriculus, ganglia, trachea, heart, great vessels, adipose tissue, and eye were histologically within normal limits.
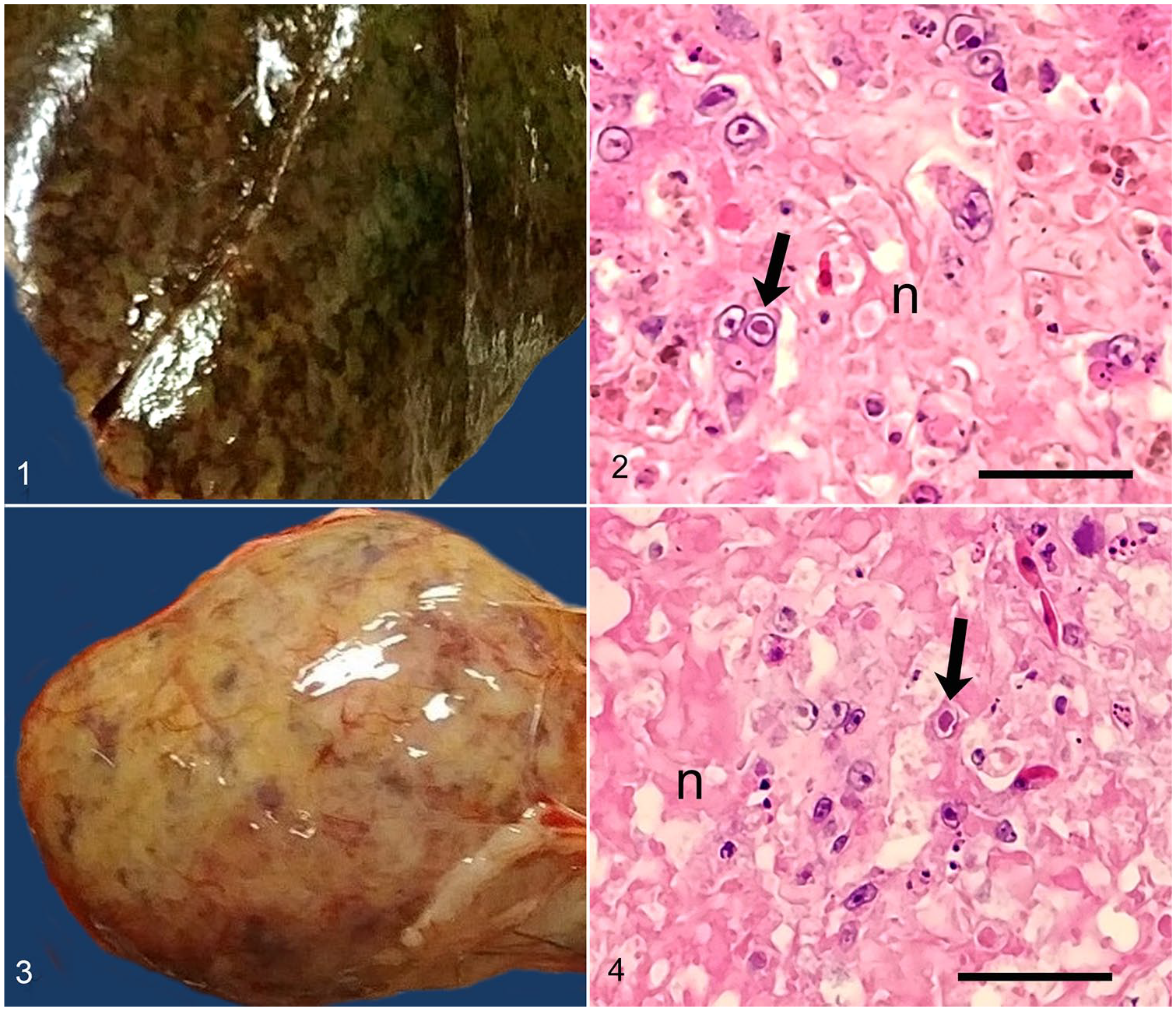
Ciconiid alphaherpesvirus 1 infection in a juvenile white stork (Ciconia ciconia).
DNA was extracted from pooled liver and spleen samples using a commercial kit, and blank extractions were used as negative controls (DNeasy tissue kit; Qiagen, Valencia, CA). A nested PCR amplification was performed utilizing a previously described technique for the herpesvirus DNA–dependent DNA polymerase gene. 19 The second round of amplification was modified to use primers DFA and IYG. An additional clean 3′-sequence was obtained using CicHV1F: (5′-GATAGCACCGAGGAAGATGC-3′) as the forward primer, designed from the initial sequence obtained. The PCR amplicon was resolved in 1% agarose gels, excised, and purified (QIAquick gel extraction kit; Qiagen). The PCR amplicon was sequenced through the Sanger method (Big-Dye terminator kit; Applied Biosystems, Foster City, CA) and analyzed with an automated DNA sequencer. Amplicons were sequenced twice in each direction. The partial viral sequence of the DNA-dependent DNA polymerase gene obtained had a total length of 480 bp excluding primers. This sequence was submitted to GenBank under accession MN017363. Following standards set by the International Committee on Taxonomy of Viruses, the herpesvirus associated with this sequence is henceforth referred to as Ciconiid alphaherpesvirus 1 (CicAHV-1). 13
The obtained viral sequence was compared with sequences within GenBank (National Center for Biotechnology Information, Bethesda, MD), EMBL (Cambridge, UK), and the Data Bank of Japan (Mishima, Shizuoka, Japan) using BLASTx. 2 The predicted homologous 58–167 amino acid sequences of 71 herpesvirus DNA–dependent DNA polymerases were aligned using MAFFT. 7 Partial sequences were included within this alignment, with ambiguities added for unknown amino acids. Elephantid betaherpesvirus 1 (GenBank accession AF322977) was selected as the outgroup. Bayesian analysis of each alignment was conducted utilizing Mr. Bayes v.3.2.6 on the CIPRES server with mixed amino acid substitution models, gamma distributed rate variation, and a proportion of invariable sites. 16 Four chains were run, and statistical convergence was assessed with the average standard deviation of split frequencies and potential scale reduction factors of parameters. The initial 25% of 2,000,000 iterations were discarded as burn-in.
Maximum likelihood (ML) analyses of each alignment was performed using RAxML-HPC2 v.8.2.10 on the CIPRES server, with a gamma-distributed rate variation and proportion of invariable sites. Elephantid betaherpesvirus 1 was again selected as the outgroup. Bootstrap analysis was used to test the strength of the tree topology, and 1,000 re-samplings were performed.
Bayesian model jumping found the WAG model of amino acid substitution most probable, with a posterior probability of 100%. CicAHV-1 was found to cluster in the subfamily Alphaherpesvirinae, within a clade containing Varicellovirus, Simplexvirus, and Mardivirus. The Bayesian analysis found a 100% posterior probability for the grouping of this clade; the ML analysis found 66.2% bootstrap support (Fig. 5).
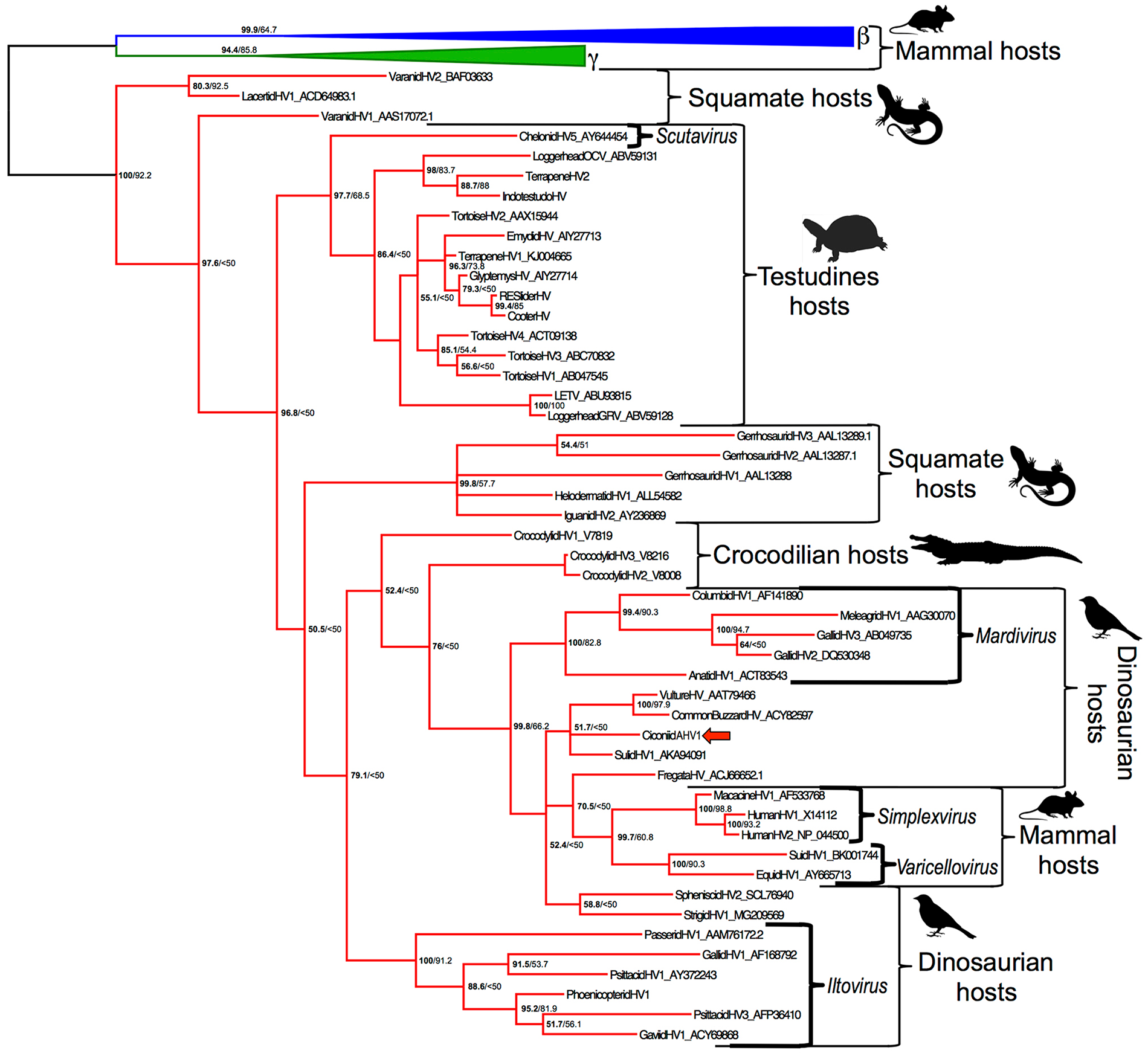
Phylogenetic tree demonstrating the relationship of the viruses within family Herpesviridae affecting vertebrate species. The first number listed at each node represents the posterior probability; the second number is the maximum likelihood bootstrap value. Alphaherpesvirinae are in red, Gammaherpesvirinae are in green, and Betaherpesvirinae are in blue. Brackets indicate select genera. Ciconiid alphaherpesvirus 1 is indicated with a red arrow.
Weight loss in this juvenile white stork predated the development of herpetic lesions, which were likely only a few days in duration. There was no evidence of inflammation or tissue repair in areas of necrosis, and the severity of the necrotic lesions would be incompatible with life. Immunosuppression associated with the weight loss may have predisposed the bird to developing fatal herpesvirus-associated lesions. In addition, this animal was septic at the time of death, the septic process likely originating in the focus of pseudomembranous enteritis. This gut lesion was likely secondary to enteric stasis or dysbiosis. No inclusions were identified in this location.
Given this animal’s young age, it appears that this white stork was likely an endemic host that succumbed to CicAHV-1. 8 It is less probable that the white stork is an aberrant host species, given that no adult animals were affected and there was no known close exposure to other species. 14 Herpesviruses are often characterized by host coevolution, and latency is a key feature in endemic hosts, given the few reports of reactivation of herpesviruses in aberrant hosts.4,8,17
The source of CicAHV-1 in this juvenile white stork is not definitively known, and, to our knowledge, other animals were not affected within the collection. The bursa of Fabricius was not examined histologically in our case and may have provided insight into the immunologic status of this animal prior to infection. 18
Mammalian alphaherpesviruses originate within lineages infecting reptiles. 12 Although the herpesvirus that initiated the jump from reptiles to mammals is probably long extinct, CicAHV-1 clusters within a clade containing the genera Varicellovirus, Simplexvirus, and Mardivirus, with strong support in the Bayesian analysis (posterior probability 100%, ML bootstrap 66%). The named genera in this clade all have strong support as monophyletic groups (100% posterior probability, 82.8–93.2% ML bootstrap values). CicAHV-1 did not cluster within any of these previously established genera but was weakly found to be more closely related to mammalian alphaherpesviruses than the genus Mardivirus (posterior probability 52%). A similar and statistically supported relationship has been reported for strigid herpesvirus 1, a novel herpesvirus that was found to also cluster with CicAHV-1 in our study. 5 In addition, ML bootstrap values are likely artificially low given the use of several partial amino acid sequences. There are now several avian alphaherpesviruses that diverge near the root of the mammalian alphaherpesviruses; additional sequencing of these viruses and taxa in this area is expected to provide insight into the ancestral virus that jumped into mammals. It remains to be seen whether these viruses represent one or more novel genera.
Herpesviruses have been isolated previously in European populations of white storks, although, to date, these isolates have not been sequenced or accessioned. Six white storks in Europe were 3–4 mo old, similar in age to the stork in our study, and histologic findings paralleled those identified with CicAHV-1. 6 Further studies to characterize and compare the herpesviruses affecting European white stork populations could provide insight to their relationship with CicAHV-1.
Footnotes
Acknowledgements
We thank Cathy Minogue of Northwest ZooPath for data retrieval, and Christie Buie of Northwest ZooPath for image layout.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
