Abstract
An 18-month-old captive black howler monkey (Alouatta caraya) died after a 3-day history of neurologic signs. Gross findings at autopsy were limited to bloody, yellow, and foul-smelling intestinal contents. Histologically, there was extensive necrotizing meningoencephalitis predominantly in both cerebral hemispheres, and lymphohistiocytic, neutrophilic infiltrate expanded the subarachnoid and Virchow–Robbin space. In the most severely affected regions, neurons contained eosinophilic intranuclear inclusion bodies surrounded by a clear halo and margination of the chromatin. Electron microscopy of the affected cells revealed numerous intranuclear viral particles characteristic of herpesvirus. Immunohistochemically, neurons and glial cells in the affected regions were labeled with a monoclonal antibody against Human herpesvirus 1, and was confirmed by polymerase chain reaction.
Nonhuman primates are the primary hosts to numerous herpesviruses, including Macacine herpesvirus 1 (cercopithecine herpesvirus 1, B virus). In contrast, humans are the primary host to Human herpesvirus 1 (HSV-1). While infection with alphaherpesviruses, such as B virus or HSV-1, causes mild or unapparent clinical signs within the primary host, disease can be severe to fatal when the virus is transmitted to other species.3,6 B virus and HSV-1 are biologically similar, resulting in neurovirulent disease in the unnatural host. Interestingly, HSV-1 infection in Old World primates is typically characterized by mild localized mucocutaneous lesions, while New World primates, such as the black howler monkey (Alouatta caraya), may develop HSV-1–induced encephalitis.3,8 Our article reports on a spontaneous infection with HSV-1 in a captive black howler monkey housed in a zoo.
An 18-month old howler monkey was presented for postmortem examination with a 3-day history of inappetence, progressive weakness, lethargy, and horizontal nystagmus. Autopsy revealed a dark red, thin-walled jejunum containing bloody, yellow, and foul mucoid material. All other organs were grossly unremarkable.
On microscopic examination, there was necrotizing meningoencephalitis affecting predominantly the gray matter in both cerebral hemispheres. Lesions were most severe in the temporal and frontal lobes, but were also observed in the hippocampus, periventricular region, diencephalon, and mesencephalon. There were no lesions in the metencephalon. The primary lesion was severe lymphohistiocytic neutrophilic meningoencephalitis with marked expansion of the subarachnoid spaces by large numbers of lymphocytes and histiocytes and fewer plasma cells and neutrophils. Virchow–Robbins spaces in affected areas were expanded by similar inflammatory cell infiltrates (Fig. 1). Also present were multifocal glial nodules in the gray matter. In the most severely affected areas, there was extensive neuronal necrosis with degenerate and necrotic neurons commonly surrounded by neutrophils and microglial cells, with focal microhemorrhages. In such areas, neurons commonly contained eosinophilic intranuclear inclusion bodies that were surrounded by a clear halo and prominent margination of the nuclear chromatin (Fig. 2).
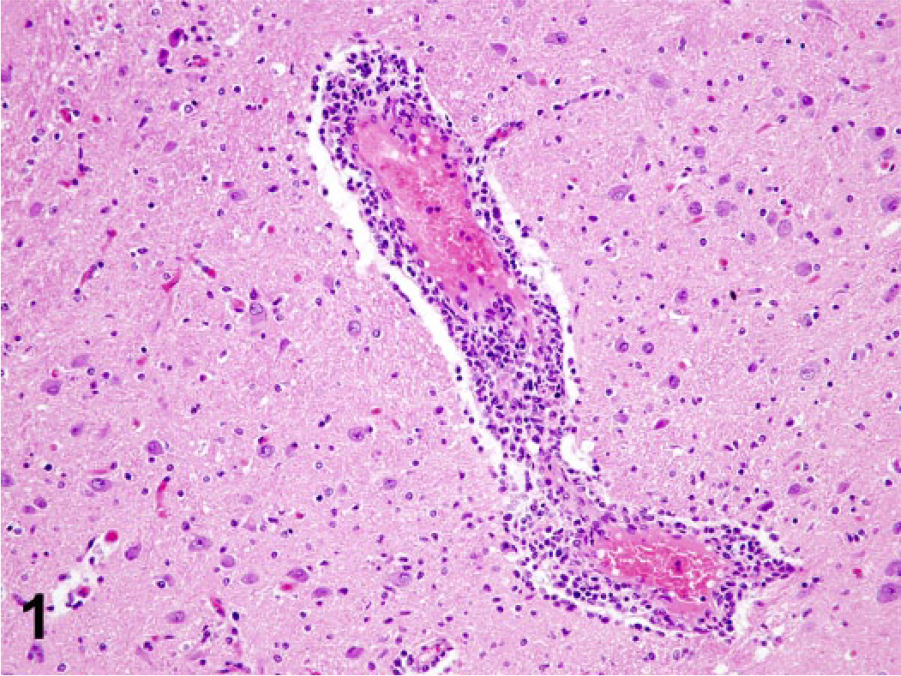
Black howler monkey (Alouatta caraya); brain. Virchow–Robbin space of vessels in affected areas is expanded by lymphocytes, histiocytes, and fewer plasma cells. Hematoxylin and eosin.
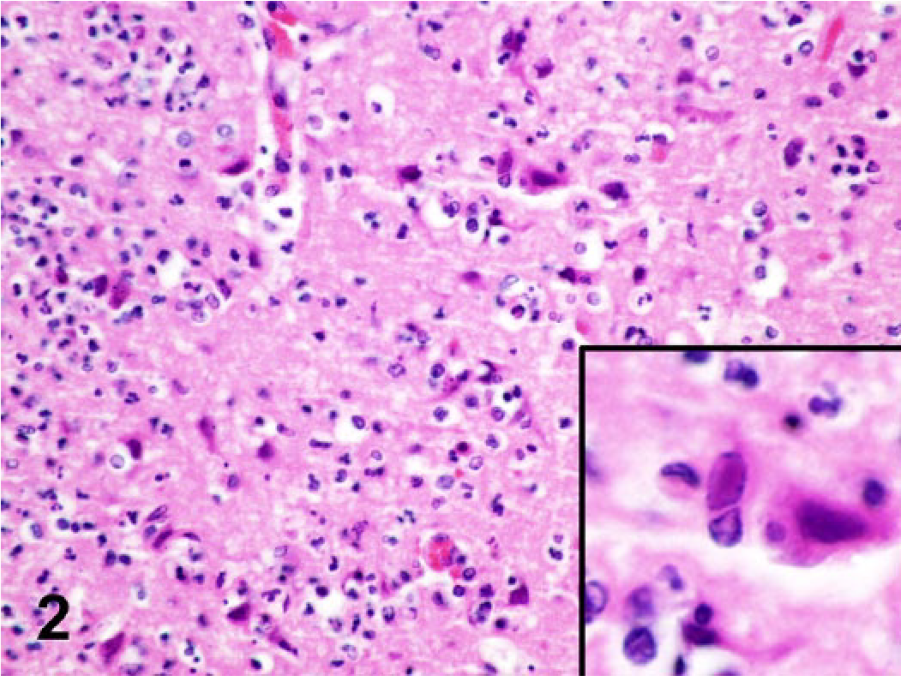
Black howler monkey (Alouatta caraya); brain. Extensive neuronal necrosis with degenerate and necrotic neurons commonly surrounded by neutrophils and microglial cells. Neurons commonly contained eosinophilic intranuclear inclusion bodies that were surrounded by a clear halo and prominent margination of the nuclear chromatin (inset). Hematoxylin and eosin.
For transmission electron microscopy, brain tissue samples that had been fixed in neutral buffered, 10% formalin solution were trimmed into 2-mm pieces and postfixed in 1% osmium tetroxide in 0.1 M sodium cacodylate buffer for 2 hr. Tissues were serially dehydrated in ethanol, infiltrated with propylene oxide and embedded in resin a in plastic molded embedding capsules. b Sections were obtained with a ultramicrotome. c Semithin (1-μm) sections were stained with 1% toluidine blue and examined to identify areas of interest, and ultrathin (90-nm) sections were cut from those areas. These sections were mounted onto 300-mesh copper grids, stained with uranyl acetate and lead citrate, and then observed under a transmission electron microscope. d Numerous hexagonal viral particles, ~100 nm in diameter with a dense DNA core, were observed within nuclei of neurons (Fig. 3).
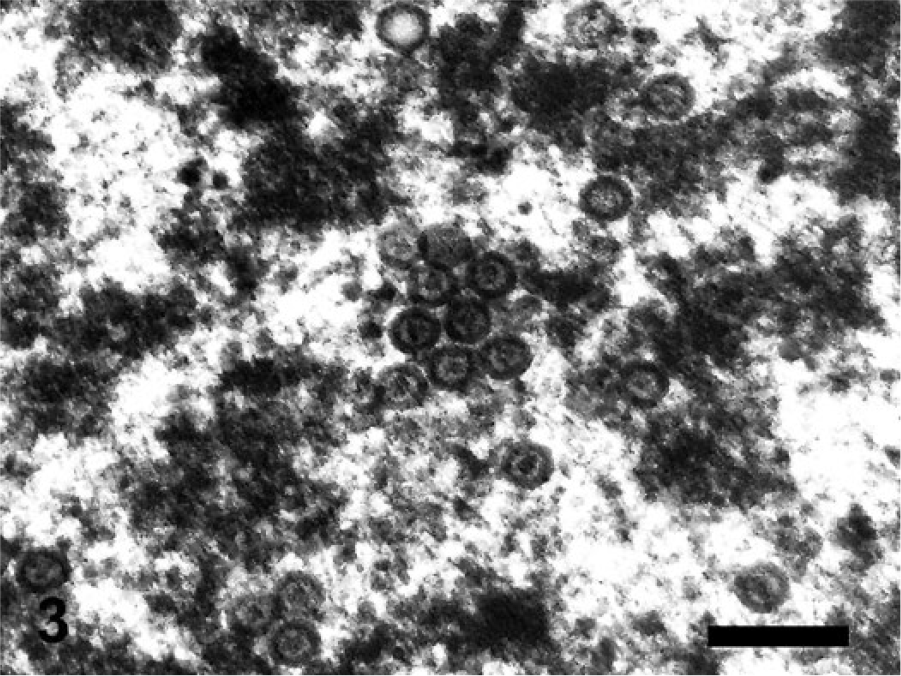
Black howler monkey (Alouatta caraya); brain. Neurons contained numerous intranuclear hexagonal viral particles measuring ~100 nm, and contained a dense DNA core. Electron microscopy. Bar = 300 nm.
Brain tissue was also tested using a consensus herpesvirus polymerase chain reaction (PCR) assay that targets a ~215–315-bp region of the herpesvirus DNA polymerase gene. 10 A PCR product of expected target size was obtained and sequenced. A 156-bp sequence (that excluded primer sequences) was derived and analyzed by BLAST against the GenBank database. 1 The nucleotide sequence was found to be 100% identical to the corresponding partial DNA polymerase sequence of HSV-1.
For immunohistochemical examination of HSV-1, formalin-fixed, paraffin-embedded samples of brain were cut into 5-μm sections. Immunohistochemistry (IHC) was performed on an automated stainer e using a polyclonal goat anti–HSV-1 antibody f at a dilution of 1:100. Deparaffinization and antigen retrieval of formalin-fixed, paraffin-embedded tissues were performed using a pretreatment system g and protein kinase. g Detection of the target was achieved using a commercial detection system, g and the immunoreaction was visualized with DAB (3,3′-diaminobenzidine) substrate g counterstained with hematoxylin. Positive immunohistochemical controls were HSV-1–infected brain tissues from a rabbit for which infection had also been confirmed by PCR. For negative controls, the primary antibody was replaced with homologous nonimmune serum. IHC identified HSV-1–specific antigen in neurons as well as endothelial cells, microglia, and ependymal cells (Fig. 4).
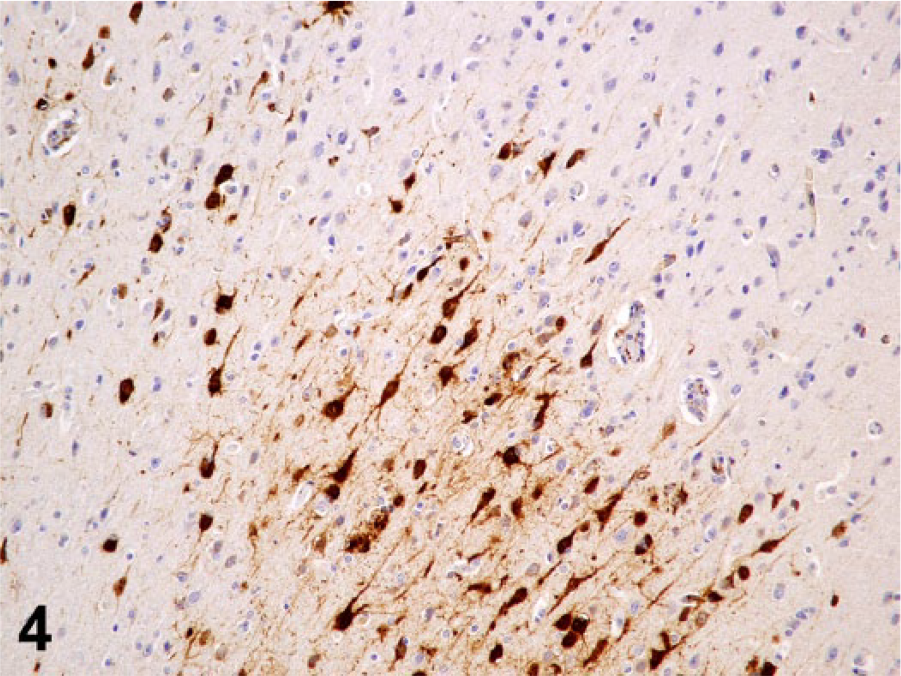
Black howler monkey (Alouatta caraya); brain. Strong immunohistochemical labeling of neurons with Human herpesvirus 1 antigen.
Microscopic examination in combination with IHC, PCR, and electron microscopy confirmed necrotizing meningoencephalitis caused by HSV-1 in a howler monkey. The lesions in the brain were consistent with those described in rodents, including mice, rabbits, and chinchillas, as well as in humans, and showcase the distinct neurotropism of the virus.2,4,7,11–13 Mice and rabbits have been commonly used as models of HSV-1infection to study ocular disease, latency, reactivation, and recurrent disease. 11 HSV-1 and HSV-2 are closely related double-stranded DNA viruses with ~70% genomic homology, and can cause lifelong latent infections in various sensory ganglia neurons in humans.2,9 A seroprevalence of >80% has been reported for HSV-1 in humans, in contrast to 20% for HSV-2.2,9
In humans, HSV-1 is associated with labial or oral lesions, while HSV-2 primarily infects the genitalia.2,9 However, HSV-1 is also the most common cause of sporadic necrotizing encephalitis in humans with an incidence of 1–3 cases per million. 9 Clinically, it is characterized by rapid onset of fever, headache, confusion, and seizures, regardless of the age of the patient. 2 During the progression of the disease, progressive obtundation as well as aphasia or hemiparesis may develop. 2 Untreated, the mortality is close to 70%. 9 HSV-2 also can cause encephalitis in neonates, as a manifestation of disseminated infection. 5 In immunocompetent humans, the primary lesion is severe necrotizing meningoencephalitis in the temporal lobes.2,9 Eosinophilic inclusions with a clear halo and nuclear chromatin margination, known as Cowdry type A inclusions, can be observed in affected neurons. 13
The pathogenesis of HSV-1 encephalitis is still unclear. While reactivation of latent virus may result in morbidity and mortality, only 10% of individuals developing encephalitis have a prior history of herpesvirus infection, suggesting that the encephalitis is not caused by recrudescence. 9 There is also evidence of genetic diversity between strains isolated from cold sores and brain in the same patient. 9 Proposed routes of infection for HSV encephalitis include entry via the olfactory bulb, ocular nerve, trigeminal ganglion, and hematogenous spread.9,12,13 The source and route of infection and disease progression could not be determined in our case; however, anthropozoonotic transmission is possible and has been reported in the rabbit, chinchilla, and marmoset.11–13
Footnotes
Acknowledgements
We thank Mark Stanhope and the staff of the Diagnostic Center for Population and Animal Health virology laboratory (Michigan State University).
Authors’ contributions
KJ Barnes drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. MM Garner contributed to acquisition of data, and critically revised the manuscript. AG Wise contributed to analysis and interpretation of data, and critically revised the manuscript. M Persiani contributed to analysis of data, and critically revised the manuscript. RK Maes contributed to analysis and interpretation of data. M Kiupel contributed to analysis of data; critically revised the manuscript; and gave final approval.
a.
PELCO Eponate 12 resin, Ted Pella Inc., Redding, CA.
b.
BEEM capsules, BEEM Inc., West Chester, PA.
c.
Leica Reichert Jung Ultracut E ultramicrotome, Leica Microsystems Inc., Buffalo Grove, IL.
d.
LEO 906e transmission electron microscope, Carl Zeiss Microscopy GmbH, Jena, Germany.
e.
Autostainer Link 48, Dako North America, Carpinteria, CA.
f.
Chemicon, Temecula, CA.
g.
PT Link, Target Retrieval Solution, EnVision Flex+ detection system, DAB substrate; Dako North America, Carpinteria, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
