Abstract
Although herpesviruses such as cyprinid herpesvirus 3 (CyHV-3) can establish lifelong latent infections, little is known about latency conditions in farmed koi populations in China. We used nested polymerase chain reaction targeting the TK gene and an indirect antibody ELISA to screen asymptomatic fish obtained from southern China for evidence of CyHV-3 infection. CyHV-3 DNA could be detected either in peripheral blood leukocytes or from gills of asymptomatic koi. Most koi sera did not contain anti–CyHV-3 antibodies; however, 5 samples were ELISA positive, providing evidence of prior CyHV-3 infections. These findings suggest that koi may survive CyHV-3 infections and become virus carriers.
Cyprinid herpesvirus 3 (CyHV-3) is the etiologic agent of koi herpesvirus disease (KHVD) causing morbidity and mortality in the common carp and koi Cyprinus carpio L. populations globally.4,10 CyHV-3, previously named koi herpesvirus (KHV) or carp interstitial nephritis and gill necrosis virus,10,15 is the type species in genus Cyprinivirus, family Alloherpesviridae, which also includes Cyprinid herpesvirus 1 (CyHV-1), Cyprinid herpesvirus 2 (CyHV-2), and Anguillid herpesvirus 1 (AnHV-1). 5 To date, KHVD has occurred in many countries or regions worldwide, including Asia,7,16,18 Europe, 9 and North America. 10 It was first identified in the United States and Israel in 1998, 10 resulting in tremendous economic impact and remains a threat to the common carp and ornamental koi industries as well as wild populations. As early as 2002, CyHV-3 was detected in imported koi in mainland China. 12 Thereafter, mortality events caused by CyHV-3 were reported in some regions, for example, Guangzhou city in southern China.6,11
Investigations of CyHV-3 infections conducted in a wild population of common carp in Japan showed that transmission of CyHV-3 between virus carrier and naive common carp occurred up to 2 y after an outbreak. 19 A 2013 survey showed that CyHV-3 latent infections were present in white blood cells of apparently healthy wild common carp and commercial koi obtained from Oregon, USA. 20 Further investigations demonstrated that B-cells are the major latency sites of CyHV-3. 14 An additional study demonstrated the persistence of CyHV-3 in polymorphic granulocytes from different fish after >2 y. 3 These fish contained CyHV-3 DNA but were asymptomatic, indicating that they were potential virus reservoirs. Asymptomatic hosts may pose a threat to the global common carp and ornamental koi breeding industries through international trade.
The common carp is one of the most economically valuable species in aquaculture and is one the most important fish species in China. Colorful ornamental koi, an inbred subspecies of common carp, are in high demand in China and other nations as pets and for competitive exhibitions. Consequently, the health status of both these fish species is crucial to maintenance of healthy aquaculture facilities as well as ecosystems. However, reports of CyHV-3 in farmed koi populations without any previous history of KHVD in China are lacking. We therefore examined cultured koi obtained from several cities in southern China to detect CyHV-3 latent infection using molecular methods and anti–CyHV-3 antibodies by ELISA. These findings will lay the foundation for further understanding of KHVD epidemiology in southern China.
Apparently healthy koi (3–4 mo, 15 ± 5 cm in length) without a history of KHVD were obtained from aquarium markets and commercial koi farms in Guangzhou, Foshan, and Dongguan located in southern China during June to September, 2015. All koi were housed in separate 0.25-L tanks with filtered circulating water maintained at 20°C to 24°C.
Fish were anesthetized (Tricaine, Tokyo Chemical Industry, Tokyo, Japan) before experiments in accordance with the Chinese Academy of Fishery Sciences Animal Care and Use Committee. Gill swabs were obtained from koi maintained in the laboratory 1 wk postcollection. The gill swabs were transferred to 1.5-mL centrifuge tubes containing180 μL of tissue lysis buffer containing proteinase K and incubated at 56°C from 1 h to overnight until tissue was lysed.
Blood was collected by caudal vein puncture using a syringe previously coated with anticoagulant (EDTA-K2, Sigma-Aldrich, Shanghai, China) and then transferred to a commercial blood collection tube. Blood samples were diluted 6-fold with cell culture medium (Medium199/EBSS, GE Healthcare Life Science, HyClone Laboratories, Logan, UT) and then layered onto lymphocyte separation medium (Ficoll-Paque PLUS, GE Healthcare, Uppsala, Sweden; density 1.077 ± 0.002 g/mL), according to the manufacturer’s instructions. The tubes were centrifuged for 40 min at 1,800 × g at 4°C, and the white cell band was collected and washed twice with phosphate-buffered saline (PBS) by centrifugation at 1,800 × g at 4°C for 10 min. The peripheral blood leukocytes (PBL) concentration was adjusted to 107 cells/mL using PBS.
Total DNA was extracted from PBL and gill swabs according to the manufacturer’s instructions (Cell/blood/tissue DNA kit, Tiangen, Beijing, China). Polymerase chain reaction (PCR) amplification of the thymidine kinase (TK) gene was carried out using 5 μL of template DNA and PCR primers (Table 1) and reaction conditions as previously reported. 1 We performed nested (n)PCR using primers designed by The Centre for Environment, Fisheries and Aquaculture Science (pers. comm.; Table 1). Two μL of the first-round PCR products were used as template in the nested round. Amplified products were visualized by electrophoresis in 1.5% (w/v) agarose gels with ethidium bromide staining and ultraviolet illumination. Sangon Biotech (Shanghai, China) carried out the DNA sequence analysis of the amplified products. BLAST software (http://blast.ncbi.nlm.nih.gov/Blast.cgi) was used to analyze DNA sequence information.
Primers used in the current study.

An ELISA procedure was used to investigate the presence of anti–CyHV-3 antibodies in fish serum and followed the guidelines of an established procedure (unpublished). In brief, microtiter plates were coated with 100 μL of purified CyHV-3 (3 μg/mL) diluted with coating buffer (pH 8.6), and incubated overnight at 4°C. The wells were blocked with 10% skim milk (Biosharp, Hefei, China) and then washed 3 times with PBS supplemented with (PBST; Tween 20, Amresco, Solon, OH). Koi sera were added at 1:300 dilution, and the plates were incubated at room temperature for 2 h. The mouse anti-carp immunoglobulin (Ig)M monoclonal antibody (Aquatic Diagnostic Lab, Scotland, UK) diluted with 1:200 PBST and the goat anti-mouse IgG horseradish peroxidase (Sigma-Aldrich, St. Louis, MO) was incubated at room temperature for 1 h and 0.5 h, respectively. PBST was the abluent for each step after incubation with antibody. Finally, TMB (3,3′,5,5′-tetramethylbenzidine; Biopanda Diagnostics, Dundonald, UK) solution was used and stopped by 2 N H2SO4. Color intensity was measured using a microplate reader at 450 nm using standard procedures. To determine the appropriate optical density (OD) cutoff value for ELISA, 10 sera from specific pathogen–free carp and 10 sera from a CyHV-3–negative farm were used. The OD values measured from the negative fish were 0.110–0.160; furthermore, the mean values of negative carp sera with OD450nm + 3 × SD + 20% security were always <0.2. Therefore, the OD value of each sample that was lower than a mean of 0.2 was interpreted as negative (unpublished).
We initially tested gill swabs or PBL from koi randomly obtained from Guangzhou, Foshan, and Dongguan using direct PCR of the CyHV-3 TK gene. The results showed that there were no positive samples from both gill swabs and PBL as judged by ethidium bromide staining of agarose gels (see top panel of Fig. 1A and 1B for samples from Guangzhou). Interestingly, CyHV-3 DNA was present in samples from either gill swabs or PBL as judged by nPCR using the reaction mixtures from the TK gene amplifications as templates (bottom panel of Fig. 1A and 1B). In some cases, both PBL and gill swabs were positive by nPCR (G1, G6, G10, and G12). Moreover, 2 gill swabs (G2 and G3) were positive, but their PBL counterparts were negative. In contrast, positive samples were found in 4 PBL samples (G5, G8, G9, and G11) that were not positive in gill swabs, suggesting that these fish had latent and not active infections. The remainders of the PBL or gill swab samples were considered negative for CyHV-3 DNA. We detected 348-bp amplicons from samples in all the study areas. No positive sample was detected by single-round PCR in samples from both PBL and gill swabs regardless of source; however, we detected 40–60% positive samples in gill swabs and 60–67% positive samples in PBL using nPCR (Table 2).
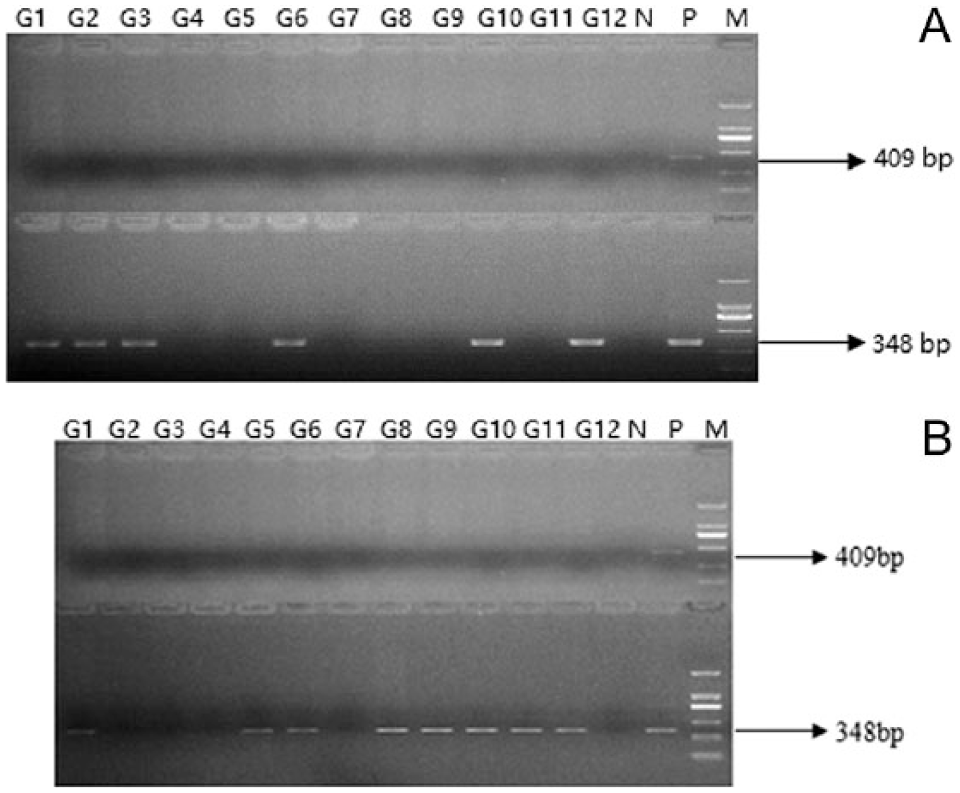
Cyprinid herpesvirus 3 DNA detection in koi (Cyprinus carpio L.) samples obtained from Guangzhou city, China (G1–G12) using TK-specific nested PCR.
Results of PCR by TK or TK-nested in gill swabs or peripheral blood leukocytes (PBLs).*
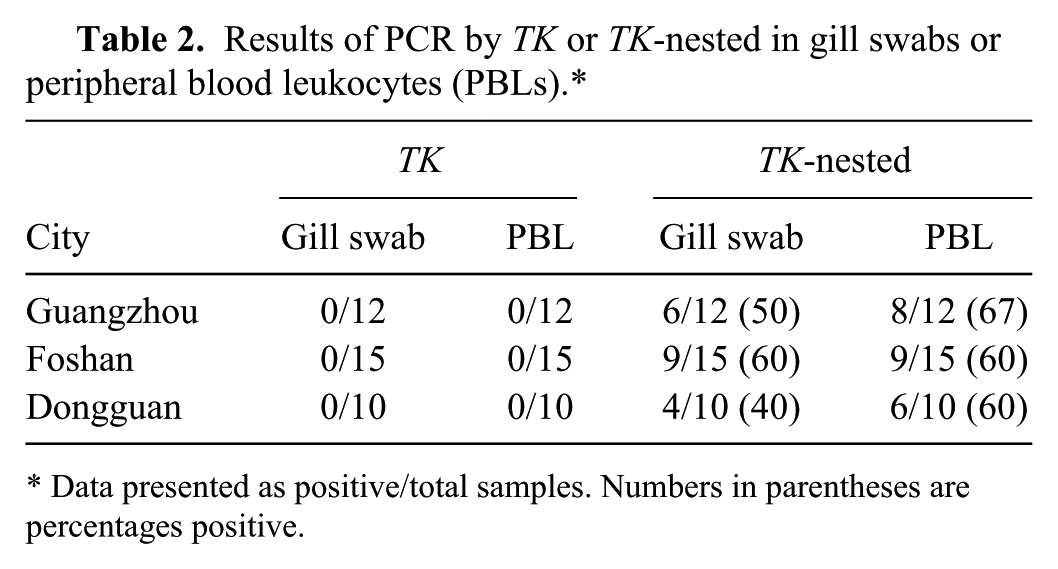
Data presented as positive/total samples. Numbers in parentheses are percentages positive.
The amplicons from the nPCR reactions were sequenced and found to be nearly identical to the TK gene of previously reported clinical isolates (KHV-U, KHV-J, KHV-GZ1301, KHV-GZ11; Supplemental Fig. 1, available at http://vdi.sagepub.com/content/by/supplemental-data). These data indicated that CyHV-3 DNA or virus was present in koi that displayed no clinical signs of disease. This analysis encompassed samples from 37 fish from 3 different regions in southern China.
We tested the sera from our koi fish samples for the presence of anti–CyHV-3 antibodies by indirect ELISA. A prior active infection was evidenced by the positive result. Only 1 serum sample (G3) originating from Guangzhou, 2 serum samples (F3 and F7) from Foshan, and 2 serum samples (D1 and D6) from Dongguan were confirmed as weakly positive; all others were considered negative (Fig. 2). ELISA values were positive in 8% of sera from Guangzhou, 13% from Foshan, and 20% from Dongguan.
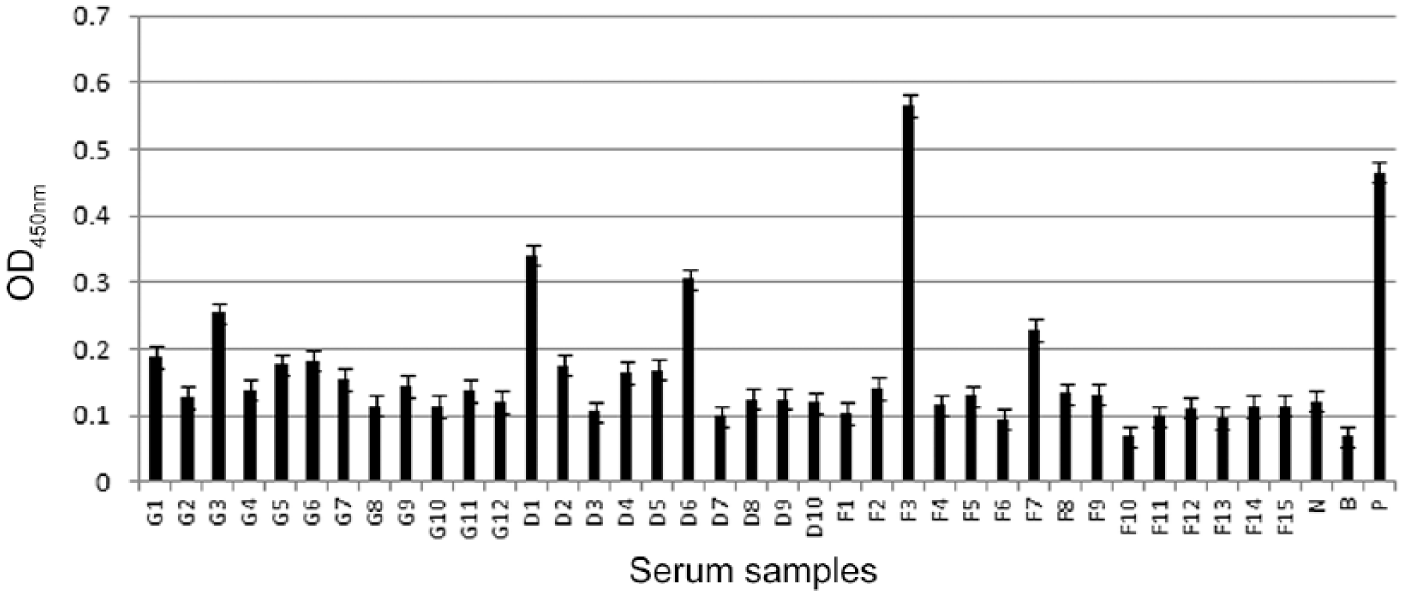
Assessment of koi (Cyprinus carpio L.) serum for anti–cyprinid herpesvirus 3 antibodies from 3 Chinese cities using indirect ELISA. G1–G12, D1–D10, and F1–F15 = sera of koi carp obtained from Guangzhou, Dongguan, and Foshan, respectively; OD = optical density; N = negative serum; P = positive control, serum collected from acutely infected koi; B = blank control.
These findings suggested that the TK gene–specific assay was not sufficiently sensitive for viral DNA detection. All samples showed negative results after the first round of PCR. However, when this step was coupled with a second nPCR amplification step, we could detect viral DNA in these asymptomatic fish. The percent positives were quite high in PBL samples with 67%, 60%, and 60%, as well as in gill samples with 50%, 60%, and 40%, respectively, in Guangzhou, Foshan, and Dongguan. Therefore, nPCR using the newly designed primers may be used as a reliable and effective protocol for testing the latently infected populations of susceptible hosts at low viral DNA concentrations.
Previous studies have suggested that CyHV-3 can establish latent infections in B-cells, as do other lymphotropic herpesviruses. 14 Reactivation can occur by stressors such as netting 2 and temperature fluctuations.8,17 In our study, CyHV-3 DNA was detected in PBL samples, indicating that these fish harbored latent virus. CyHV-3 DNA was also detected in gill swab samples with or without PBL samples, which collectively suggested that the fish had low-level reactivation of CyHV-3 in the gills during handling. These fish may act as the source of koi herpesvirus under stress conditions, especially given that latent virus is not easy to eliminate. For some koi with high economic value, elimination or quarantine of latently infected fish is a viable control strategy to prevent possible virus reactivation and further spread.
The presence of anti–CyHV-3 antibodies indicated that the fish had been infected previously. However, the percent antibody-positive in serum was much lower than that of DNA positives from PBL or gill swabs. According to a previous report, 13 wild-type or attenuated CyHV-3 induced an increased titer of antivirus antibodies between days 7 and 14 in virus-inoculated fish, and the titer of the antiviral antibodies increased up to 20–40 d postinfection (dpi), then gradually decreased over 150 d. In our study, all the samples were collected from asymptomatic fish without a history of KHVD. It was speculated that the fish may have been infected with a low dose of virus or an attenuated virulent strain for several months or even longer, and thus antibodies in most samples were undetectable. Furthermore, a major biological property of herpesviruses is that they are able to establish lifelong latent infection, which is characterized by the absence of production of infectious virus particles, but presence of intact viral genomic DNA. A previous report demonstrated that B-cells are the major latency sites of CyHV-3. 14 Viral DNA could be detected during CyHV-3 latency, whereas the antibody against virus decreased to an undetectable level over time, which could explain why 8 of 12 PBL samples were DNA positive, but only 1 of 12 were antibody-positive in sera, for example, in samples from Guangzhou. Although antibody assessment is used routinely in mammalian and fish screening procedures, the limited sensitivity of this method hinders its value. Until the sensitivity can be increased, current recommendations are for the use of molecular testing combined with ELISA.
Footnotes
Acknowledgements
We thank Dr. Lin Jin (Department of Biomedical Sciences, College of Veterinary Medicine, Oregon State University) for providing valuable information about CyHV-3 latency detection.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Key Technology R&D Program (2013BAD12B02), earmarked fund for Oceanic and Fishery Science Development of Guangdong Province in China (A201501B13) and China Agriculture Research System (CARS-46).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
