Abstract
On December 8, 2008, a male fisher (Martes pennanti) housed in a quarantine enclosure at the St-Félicien Zoo was found dead with multiple skin ulcers on the muzzle and plantar pads. At necropsy, no major findings were found, and a specific cause of death was not determined microscopically. However, at the borders of ulcerated sites, there were increased numbers of koilocytes, with perinuclear vacuolation and nuclear enlargement. A pan-herpesvirus nested polymerase chain reaction (PCR) assay was conducted, and an expected PCR product of 230 nucleotides was obtained within tissues collected from around the skin ulcers. Other tissues, including intestines and pool of lung, liver, and kidney, tested negative. The obtained PCR amplicon was sequenced and was highly related to the partial viral DNA polymerase (DPOL) gene of Mustelid herpesvirus 1. Virus isolation was negative, and no virion was detected by electron microscopy. The pathogenic potential of this novel herpesvirus and its role in the death of the fisher are unknown.
The fisher (Martes pennanti) is a fur animal of the size of a domestic cat with a long tail and short legs, classified in the Mustelidae family. 15 As a generalist predator, the fisher’s diet consists predominantly of North American porcupines, snowshoe hares, squirrels, small mammals, birds, carrion, and, to a lesser extent, fruits and plants.10,11 Fishers have been reported in the forest regions of several states of the United States and provinces of Canada.11,14
In October 2008, 2 captive-born fisher couples, each aged 2.5 years, were bought from a breeding farm in Minnesota, and sent to the St-Félicien Zoo in Quebec, Canada. While in quarantine, a physical exam was performed under anesthesia 6 days after their arrival. Hematology and biochemistry blood tests were normal except for a marginal uremia of 16.9 mmol/l (reference values: 3.57–12.14 mmol/l) detected in one of the males. Fecal and radiographic exams were performed and no significant clinical findings were recognized.
On December 8, 2008, the male fisher that was previously found marginally uremic in October 2008 was found dead in its quarantine enclosure with multiple skin ulcers on the muzzle and plantar pads (Fig. 1). A variety of causes could induce ulcerative skin lesions but with the gross lesions of the muzzle and plantar pads, Feline calicivirus (FCV), Felid herpesvirus 1 (FHV-1), and Canine distemper virus (CDV) as well as frostbite were considered as possible causes. The 3 other healthy fishers had shown no clinical signs of illness.
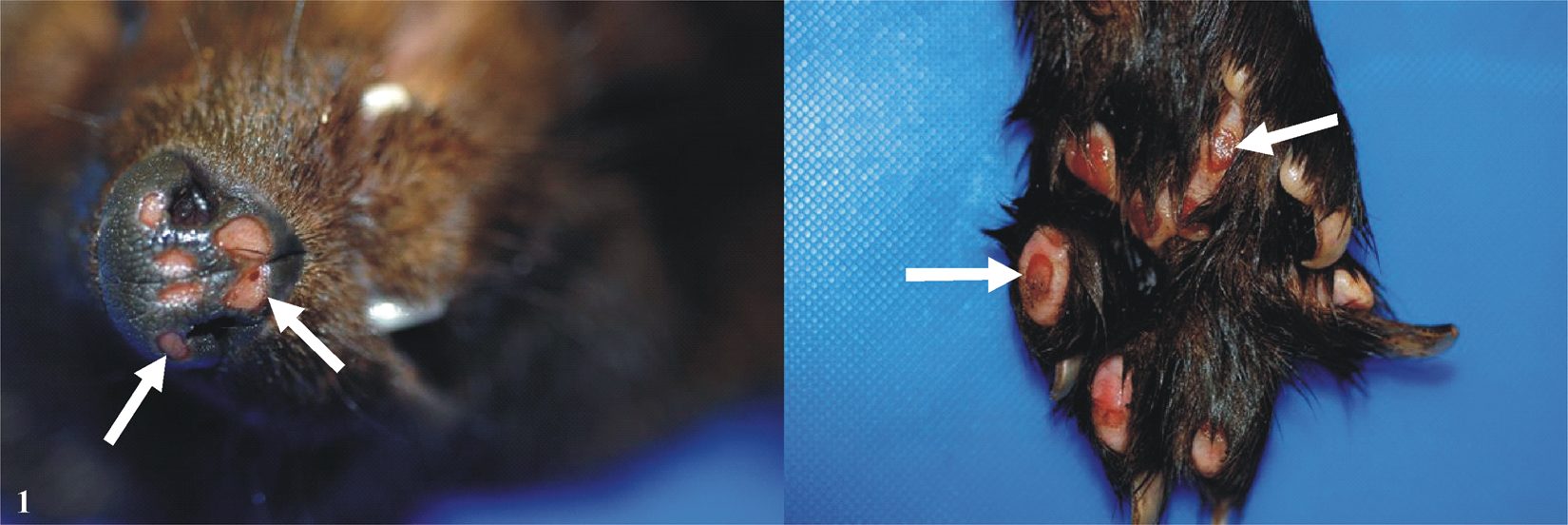
Fisher (Martes pennanti) skin; palmar pads and muzzle ulcer lesions. Enlarged ulcerative lesions of the muzzle can be seen in the left panel and enlarged ulcerative lesions of 1 footpad are illustrated in the right panel. Arrows indicate the ulcers.
On gross examination, the fisher, which had lost 1.68 kg in body weight, had no fat in the body cavities or surrounding the organs. There were patchy losses of hair on both sides of the flanks and on the hips. The most remarkable lesions were sharply demarcated and deep, round to oval ulcerations of approximately 0.5–1.0 cm in size located on all paws and footpads, and on the muzzle (Fig. 1). Selected few grams of tissues (brain, lung, heart, liver, kidney, lymph nodes, spleen, stomach, bladder, intestine, pancreas, adrenals, thyroids, muscles, and skin) were taken for light microscopic evaluation examination, toxicology, a bacteriology, b immunohistochemistry, c electron microscopy (EM) examination, c virus isolation, and polymerase chain reaction (PCR) assays.
A specific cause of death was not determined microscopically. It is noteworthy that endocrine dermatosis might be considered to explain the marginal alopecia. Thus, sections of the skin with alopecia, adrenal, and thyroid glands were examined. Thickness of the epidermis varied from 4 to 5 cells thick to 1–2 cells thick, suggestive of epidermal atrophy. In these regions, the hair follicles appeared atrophied. No cortical hyperplasia or hypoplasia, or nodular hyperplasia, were noticed in the adrenal glands. No remarkable changes were noted in the thyroid. No specific tests were done to assess the hormonal status of the animal. Beneath the ulcers noted grossly, there were occasional fibrin thrombi and mild superficial infiltrate of neutrophils, lymphocytes, and plasmacytes (data not shown). At the margin of the ulcerated sites, the epidermis was thickened with increased numbers of koilocytes, with perinuclear vacuolation (Fig. 2B) and nuclear hypertrophy. Occasionally, there were scattered discrete cytoplasmic basophilic and, rarely, eosinophilic (not shown) inclusion–like material within the vacuoles (Fig. 2C) and areas with large, pale amphophilic intranuclear inclusions, which displaced the chromatin peripherally (Fig. 2C, 2D). No intralesional bacteria were detected following Gram and periodic acid–Schiff stains.
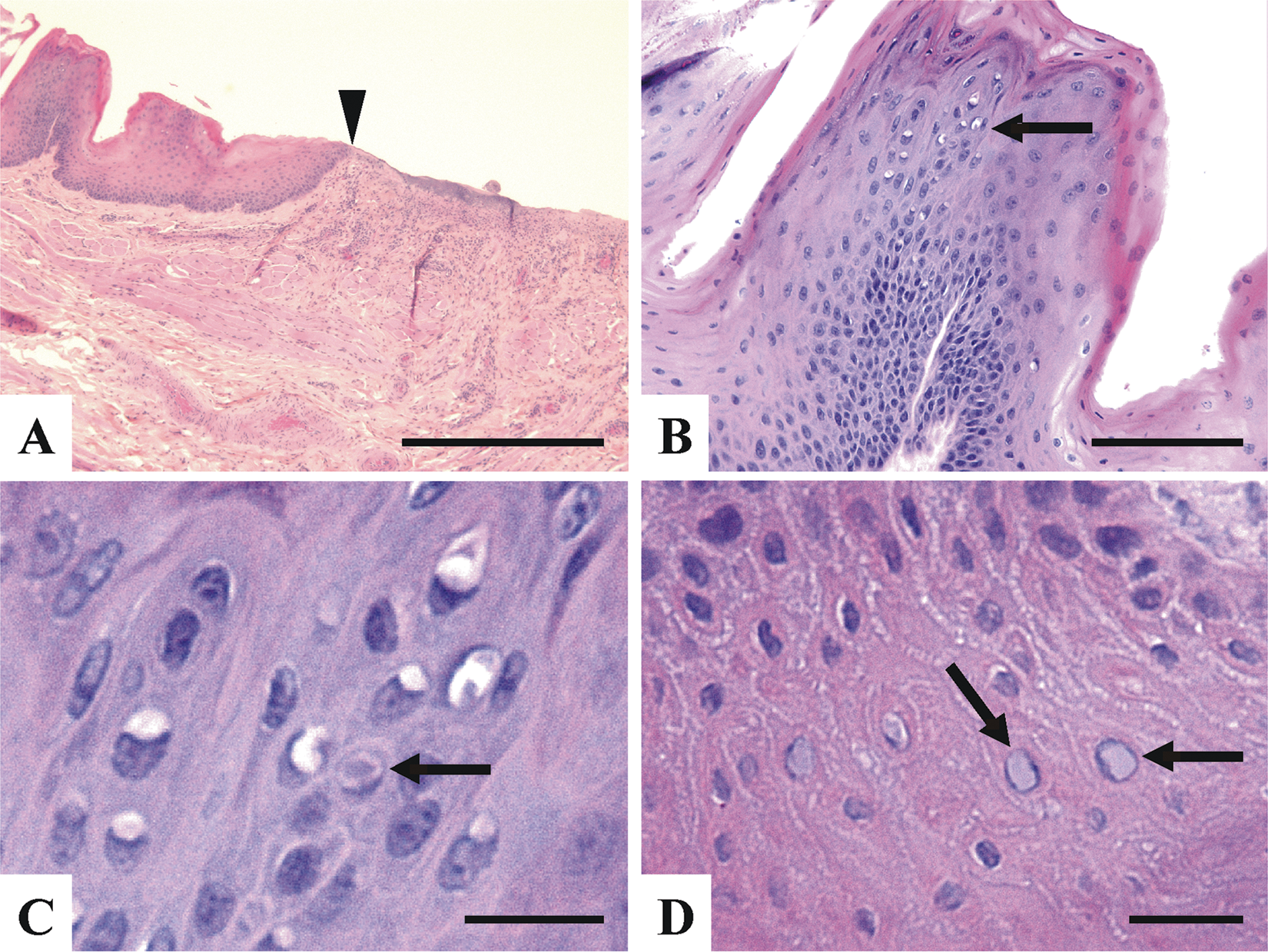
Fisher (Martes pennanti) skin; representative microscopic findings associated with the ulcer lesions.
A heavy metals screen panel (selenium, copper, arsenic, lead, iron, and zinc) performed in the liver was negative. Lung, liver, and colon were sent for aerobic culture on blood agar plate (containing 5% [v/v] defibrinated sheep’s blood) with overnight incubation at 37°C. No pathogenic bacteria were found. Using an avidin–biotin complex immunoperoxidase method, the following antibodies were used to detect viral antigens: 1) rabbit anti-Measles virus, d which detects antigen from several viruses classified within the Morbillivirus species, including CDV; 2) rabbit anti-adenovirus type 2, e which detects antigen from a number of Mastadenovirus species, including Bovine adenovirus A–C, Canine adenovirus, and Equine adenovirus A and B; and 3) monoclonal antibodies specific for FHV-1 (clone FHV7-7) c and FCV (clone FVCS-19). c Consistency of staining for each antigen was confirmed using tissue sections from known positive cases stained in parallel with the fisher tissues. Immunostaining for CDV in the skin and brain and for FHV-1, FCV, and adenovirus in the skin were negative.
Virus isolation was attempted using Madin–Darby canine kidney (MDCK) and Crandell feline kidney (CRFK) cell lines f because they are well known to permit the isolation of several types of viruses.2,4,7,8 The fetal mink lung (ML) epithelial cell line g was also used because it is derived from a member of the Mustelidae family.1 Ten-day-old embryonated eggs h (inoculated into the chorioallantoic sac and the chorioallantoic membrane) were also used for virus isolation. Unfortunately, all virus isolation attempts were negative. Skin lesions and surrounding tissues were ground in a glass tissue homogenizer, prepared accordingly (as cell culture supernatants and allantoic fluids), and then negatively stained for electron microscopy visualization. No virus particle could be observed in any of the prepared samples.
A pan-herpesvirus nested PCR (nPCR) assay was conducted as previously described.3,13 Briefly, DNA was extracted from 1 g of tissue sample, including skin, intestines, and a pool of lung, liver, and kidney, with a commercial kit i according to the manufacturer’s tissue protocol. The PCR i was conducted according to the manufacturer’s specifications, with a set of 3 primers (DFA, ILK, and KG1) as previously described. 13 Nested PCR was then performed under the same conditions using 5 µl of the first PCR reaction and 2 other primers (TGV and IYG), as previously described. 13 An expected PCR product of 230 nucleotides (nt) was obtained only with skin tissues collected from and around the ulcers. The obtained PCR products were sequenced using a standard automated sequencing method, j and a sequence of 219 nt in length was submitted to GenBank Basic Local Alignment Search Tool (BLAST) for comparison. The BioEdit Sequence Alignment Editor software with the ClustalW alignment method k was used for nt and amino acids (aa) comparisons. Nucleotide comparison showed that the nearest nt homology was with the viral DNA polymerase (DPOL) gene of Mustelid herpesvirus 1 (MusHV-1; also known as Badger herpesvirus; order Herpesvirales, family Herpesviridae, subfamily Gammaherpesvirinae, genus Percavirus). 1 The Fisher herpesvirus (FiHV) partial sequence possesses 85.3% nt identity and 91.7% aa identity with the MusHV-1 DPOL gene published sequence (Fig. 3). Compared to MusHV-1, FiHV possesses a total of 23 nt silent mutations over a total of 32 nt mutations (Fig. 3). From a total of 73 deduced aa, 67 aa are identical between FiHV and MusHV-1 (Fig. 3). Attempts to increase the length and yield of the PCR product obtained by the pan-herpesvirus nPCR assay, and to amplify other viral genomic regions of FiHV by designing new PCR primers based on the reported sequences of MusHV-1, were unsuccessful.

Partial nucleotides and deduced amino acids sequences of Fisher herpesvirus (FiHV) viral DNA polymerase (DPOL) gene compared to Mustelid herpesvirus 1 (MusHV-1) sequences.
Frostbite could cause vascular changes followed by epidermal necrosis but will most often affect extremities like the tips of the ears and tail. The footpads lesions were particularly similar to the cutaneous lesions observed when cats are infected with a systemic virulent strain of FCV 9 but immunohistochemistry results were negative for FCV. Canine distemper virus is known to infect animals classified within the Mustelidae family and to induce typical hyperkeratosis skin lesions located at the same sites where the fisher’s ulcerative lesions were observed. Noteworthy, all CDV diagnostic results were negative, and no microscopic lesions associated with CDV infection were found in tissues. Herpesviruses are well known to cause ulcers of the skin and mucosa in several mammals. 6 It is possible that FiHV could be involved in the formation of the skin ulcers, but many animals are known to harbor herpesviruses asymptomatically. 6 Unfortunately, no virus was isolated and observed by EM. If FiHV was the etiological cause of the lesions, it would be expected that virions would be observed by EM within and surrounding the cutaneous ulcers lesions. Noteworthy, the EM technical approach that was used possesses low sensitivity. In spite of the fact that the macroscopic skin lesions were impressive, the histopathological findings were rather mild and not typical of classic herpesvirus diseases (Fig. 2). It is also noteworthy that similar histopathological findings have been previously observed in cases of herpesvirus-associated skin lesions reported in cats and green sea turtles.5,12 Thus, the detection of FiHV DNA only within skin lesions and not in other tissues as well as the absence of other known pathogens in those lesions suggests that FiHV might be associated with the formation of such ulcers, but these findings alone are certainly not conclusive.
Is it possible that the pan-herpesvirus nPCR assay gave a false-positive result? It is believed that if it was the case, then there is a good chance that this PCR assay would be positive for other fisher tissues, which was not the case (data not shown). The novel viral DNA sequence of FiHV was most closely related to the badger herpesvirus (i.e., MusHV-1). 1 Like the fisher, the badger is an animal classified in the Mustelidae family. 15 Thus, it is reasonable to assume that FiHV is a new herpesvirus classified in the subfamily Gammaherpesvirinae. Further fisher cases would have to be studied and additional work is needed to establish the involvement of FiHV in the ulcer lesions and in the death of the animal.
Footnotes
Acknowledgements
The authors are grateful to Dr Dale L. Godson (Prairie Diagnostic Services Inc.) for his help with the immunohistochemistry and EM assays. In addition, the authors are grateful to Cynthia M. Guilbert and Sanela Music for critically reviewing the manuscript. The authors are grateful to Ossama Allam, Guy Fontaine, and Denis St-Martin for their technical support.
a.
Direction du laboratoire d’expertises en analyses alimentaires (DLEAA), Ste-Foy, Quebec, Canada.
b.
Laboratoire d’expertise en pathologie animale du Québec (LEPAQ), Quebec, Canada.
c.
Dr. D. L. Godson, Prairie Diagnostic Services Inc., Saskatoon, Saskatchewan, Canada.
d.
Dr. D. B. Ziola, University of Saskatchewan, Saskatoon, Saskatchewan, Canada.
e.
Lee Biomolecular Research, San Diego, CA.
f.
Dr. S. Dea, Institut national de la recherche scientifique, Institut Armand-Frappier, Laval, Quebec, Canada.
g.
NBL-7, ATCC CCL 64; American Type Culture Collection, Manassas, VA.
h.
Canadian Food Inspection Agency, Nepean, Ontario, Canada.
i.
QIAamp DNA mini kit, Fast Cycling PCR kit; Qiagen Inc., Mississauga, Ontario, Canada.
j.
Sequencing Laboratory, Faculté de médecine vétérinaire, Université de Montréal, St-Hyacinthe, Québec, Canada.
k.
BioEdit Sequence Alignment Editor software version 7.0.5.2, Ibis Therapeutics, Carlsbad, CA.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The current work was supported by the Natural Sciences and Engineering Research Council of Canada (NSERC) discovery grant (to CAG), the Veterinary diagnostic service of the Faculty of Veterinary Medicine, University of Montreal and the Animal Pathology Expertise Laboratory of Quebec’s Ministry of Agriculture Fisheries and Food (MAPAQ).
