Abstract
The addition of ethylenediamine tetra-acetic acid (EDTA) to serum can affect the measurement of cortisol by chemiluminescent enzyme immunoassay (CEIA); addition of magnesium chloride (MgCl2) may reverse the effects. However, similar characteristics for thyroxine (T4) measurement are unknown. We measured cortisol and T4 in paired EDTA-anticoagulated plasma and serum samples from 50 dogs. Additionally, both hormones were measured in 15 samples of each type after the addition of MgCl2. Samples were collected under routine clinical conditions; therefore, specific EDTA concentrations in plasma samples were unknown. Cortisol and T4 values were significantly different comparing plasma and serum samples in the absence of MgCl2. For cortisol and T4, EDTA-plasma concentrations were 51.2% and 43.7% higher than serum, respectively (p < 0.001 for both). The addition of MgCl2 to plasma significantly decreased the measured cortisol concentrations (p < 0.001) but not T4 (p = 0.44). After addition of MgCl2, cortisol concentrations in EDTA-plasma were no longer significantly different from serum, whereas T4 concentrations in EDTA-plasma remained significantly different from serum. In the clinical setting in which tubes may be underfilled, use of EDTA-plasma significantly increases the measured concentration of cortisol and T4 obtained by CEIA. Addition of MgCl2 to EDTA-plasma can overcome the effects of EDTA when measuring cortisol, but not T4. Thus, T4 should not be measured in EDTA-plasma.
Keywords
Introduction
Hyperadrenocorticism is a common endocrine disease in dogs; hypoadrenocorticism is a less common disease in dogs but can be fatal if misdiagnosed. The adrenocorticotropic hormone (ACTH) stimulation test, low-dose dexamethasone suppression test, and assessment of baseline cortisol concentration are screening tests in which cortisol concentrations are measured to rule in or out hyperadrenocorticism and hypoadrenocorticism.2,12 Accurate cortisol measurements are crucial for diagnosis. Hypothyroidism is a common canine endocrinopathy, with measurement of thyroxine often employed for both diagnosis and monitoring of therapy.
Previously, radioimmunoassay (RIA) was most commonly used by veterinary reference laboratories to measure cortisol and thyroxine (T4). Given that cortisol is more stable with time and temperature variation when stored as ethylenediamine tetra-acetic acid (EDTA)-plasma compared with serum, use of EDTA-containing tubes was recommended for collection of samples for cortisol measurement, particularly with samples shipped to our laboratory (Auburn University Endocrine Diagnostic Laboratory, Auburn, AL). 3 Use of RIA for veterinary hormone measurement is declining, being increasingly replaced by nonradioactive methodologies such as chemiluminescent enzyme immunoassay (CEIA). However, EDTA can interfere with CEIA measurement of cortisol 10 and parathyroid hormone. 8
EDTA concentrations in commonly used Vacutainer tubes (BD, Franklin Lakes, NJ) is expected to be 4.1 mmol/L (1.8 mg/mL) if the tubes are filled to their designated fill capacity. Underfilling of tubes increases the final EDTA concentration, and excess EDTA interferes with the CEIA measurement. For example, cortisol concentrations were significantly increased in canine serum samples when EDTA had been added to a concentration ≥ 5.1 mmol/L (2.25 mg/mL). Addition of magnesium chloride (MgCl2; 5 mmol/L) reversed the effect of EDTA. 10 In that study, known amounts of EDTA were added to serum samples before cortisol measurement. Although this permitted determination of EDTA concentration in samples, it did not reflect sample collection in clinical situations. Underfilling of EDTA-containing tubes is anticipated to be common when used in clinical situations. Although use of serum as the sample type obviates the concern over EDTA interference, a variety of sample types are typically submitted to diagnostic laboratories. Samples for T4 measurement are usually collected as serum; nevertheless, on occasion, EDTA-plasma is analyzed. The effect of EDTA on T4 measurements has not been reported, to our knowledge.
Our first objective was to determine if sample type, namely serum or EDTA-plasma, impacted values of either cortisol or T4 determined using CEIA. Our second objective was to determine whether addition of MgCl2 influenced the effect of EDTA. Our hypothesis was that plasma samples collected in the presence of EDTA would show increased apparent concentrations of both cortisol and T4 compared with serum and that addition of MgCl2 would negate the influence of EDTA.
Materials and methods
Animals
Paired EDTA-anticoagulated plasma and serum samples were obtained from 50 dogs. Samples for phase I (5 dogs) and II (45 dogs) were collected from July to December 2015 and from August to November 2017, respectively. Blood was collected into plain tubes or EDTA-containing tubes (BD) and obtained from the Auburn University Clinical Pathology Laboratory from Auburn University Veterinary Teaching Hospital submissions. Signalment and reason for hospital visit were not considered. Inclusion criteria were that EDTA-anticoagulated plasma and serum samples were obtained from the same blood draw and at least 1.5 mL and 1.0 mL of sample remained for testing in phases I and II, respectively. Exclusion criteria included having received oral thyroxine, prednisone, prednisolone, hydrocortisone, dexamethasone, or methylprednisolone within the previous 24 h or an injection of ACTH within the previous 4 h. Because samples used were convenience samples obtained in the course of routine standard clinical patient care, and blood used was the residual remaining after laboratory testing was completed, specific client consent and approval by the Clinical Research Review Committee or an Institutional Animal Care and Use Committee were not required as per institutional policy. The EDTA concentration in plasma samples was unknown because total blood volume varied in each tube.
Phase I
Given that blood was collected from the Clinical Pathology Laboratory after being sampled for testing purposes in phase II, some EDTA-containing tubes may not have been centrifuged for up to 24 h. Phase I was performed to ensure that the plasma concentrations of cortisol and T4 were not affected by delayed centrifugation. Paired red-top and EDTA-containing tubes from 5 dogs were obtained immediately after collection. EDTA-containing tubes were centrifuged immediately or after being stored at room temperature for 30 min, 3 h, 6 h, or 24 h. Serum collection tubes were allowed to clot at room temperature and centrifuged within 30 min of collection. Serum and EDTA-plasma were collected after centrifugation and stored at −20°C until use.
Phase II
Paired canine EDTA-plasma and serum samples were obtained from an additional 45 dogs in a clinical setting. The exact EDTA concentration in the plasma samples was unknown. The serum samples collected in phase I as well as the EDTA-plasma samples that had been centrifuged immediately were also included for a total of 50 dogs in phase II. Cortisol and T4 concentrations were measured in the paired serum and EDTA-plasma samples. In the last 15 samples, MgCl2 was also added to both serum and EDTA-plasma to a final concentration of 5 mmol/L as described previously, 10 and cortisol and T4 measured after the addition.
Hormone assays
Cortisol and T4 were measured using a previously validated,11,14 solid-phase, automated, CEIA (Immulite 1000; Siemens Healthcare Diagnostics, Tarrytown, NY). The assay coefficient of variation (CV) was < 20% for both. 14 Samples were assayed in duplicate, and the average concentration was used for statistical analysis.
Statistical analyses
For the paired measurements of cortisol and T4, the mean CV and the 95% confidence intervals (CIs) were calculated for each hormone. Normality of data distribution was tested using a Shapiro–Wilk normality test. Statistical tests were used accordingly. In phase 1, cortisol data were analyzed using a one-way repeated measures ANOVA on ranks; T4 data were analyzed using a one-way repeated measures ANOVA. In phase 2, for the full group of 50 dogs, concentrations of cortisol in serum and EDTA-plasma were compared using a Wilcoxon signed-rank test, and concentrations of T4 were compared using a paired t-test. The difference between the concentration measured in serum and in EDTA-plasma was calculated for both cortisol and T4. A Pearson product moment correlation was calculated to determine correlation between the difference in the serum and EDTA-plasma concentrations and the serum concentration. The correlations for cortisol concentrations and for T4 concentrations were compared with a statistical test using the r-to-z transformation.
For the subgroup of 15 dogs for which MgCl2 was added, cortisol concentrations in plasma with and without MgCl2 and in serum without MgCl2 (i.e., the 3 groups of clinical interest) were compared using a one-way repeated-measures ANOVA on ranks. A post-hoc test using the Dunnett method was performed with EDTA-plasma concentrations without added MgCl2 as the control group. To determine if the addition of MgCl2 directly affected the measured concentration, cortisol concentrations measured in serum were compared to cortisol concentrations measured in serum with MgCl2 added using a Wilcoxon signed-rank test. T4 concentrations in plasma with and without MgCl2 and in serum without MgCl2 (i.e., the 3 groups of clinical interest) were compared using a one-way repeated-measures ANOVA. A post-hoc test using the Dunnett method was performed with the concentrations in EDTA-plasma without MgCl2 as the control group. To determine if the addition of MgCl2 directly affected the measured concentration, T4 concentrations measured in serum were compared to T4 concentrations measured in serum with MgCl2 added using a paired t-test. All analyses were performed using statistical software (SigmaPlot 12.0, Systat Software, San Jose, CA; Prism v.8.2.0, GraphPad, San Diego, CA; SAS 9.4 for Windows, SAS Institute, Cary, NC). Significance was set at p ≤ 0.05.
Results
Mean CVs for all paired measurements and 95% CIs were calculated for cortisol and T4. For paired cortisol measurements, the mean CV was 5.5% (95% CI: 4.7–6.3%). For paired T4 measurements, the mean CV was 5.6% (95% CI: 4.2–6.9%).
Phase I
Five dogs were included in phase 1. Breeds in the study population included 2 Great Danes, 2 mixed-breed dogs, and 1 Golden Retriever. Two dogs were spayed females, 1 dog each was an intact female, castrated male, and intact male. Median age of the dogs was 8 y (range: 3–10 y). No significant difference was detected in cortisol nor T4 concentrations over time in EDTA-plasma samples (p = 0.251 and p = 0.139, respectively; Supplementary Table 1).
Phase II
The EDTA-plasma samples used from phase 1 were the samples that were centrifuged immediately after collection. Breeds represented in the additional 45 dogs included 5 mixed-breed dogs; 4 each of Miniature Pinscher and Labrador Retriever; 3 each of Boston Terrier and Miniature Schnauzer; 2 each of German Shepherd dog, Miniature Dachshund, Border Collie, Great Dane, and Yorkshire Terrier; and 1 each of Golden Retriever, German Short-Haired Pointer, Boxer, Cairn Terrier, Vizsla, Havanese, English Springer Spaniel, Bichon Frisé, American Cocker Spaniel, Samoyed, Cavalier King Charles Spaniel, Pug, Jack Russell Terrier, American Staffordshire Terrier, and Miniature Poodle. Nineteen dogs were castrated males, 23 dogs were spayed females, and 2 dogs were intact females. Neuter status of one male dog was not recorded. Median age of the dogs was 9 y (range: 0.25–17 y).
Cortisol concentrations measured in EDTA-plasma were significantly higher than those measured in serum (Fig. 1; p < 0.001). Cortisol concentrations in EDTA-plasma were a median 51.2% higher (range: 3.0–393%) than serum. In the subgroup of 15 dogs for which MgCl2 was added, cortisol concentrations were significantly different between plasma and serum samples if MgCl2 was not added (p < 0.001; Fig. 2). The addition of MgCl2 to plasma significantly decreased the measured cortisol concentration in EDTA-plasma (p < 0.001), and the cortisol concentrations in EDTA-plasma with MgCl2 added were a median 4.9% higher (range: 8.8–29.8%) than serum. After the addition of MgCl2, the cortisol concentrations measured in EDTA-plasma were no longer significantly different from serum (p = 0.080). Cortisol concentrations measured in serum with and without MgCl2 were not significantly different (p = 0.60).
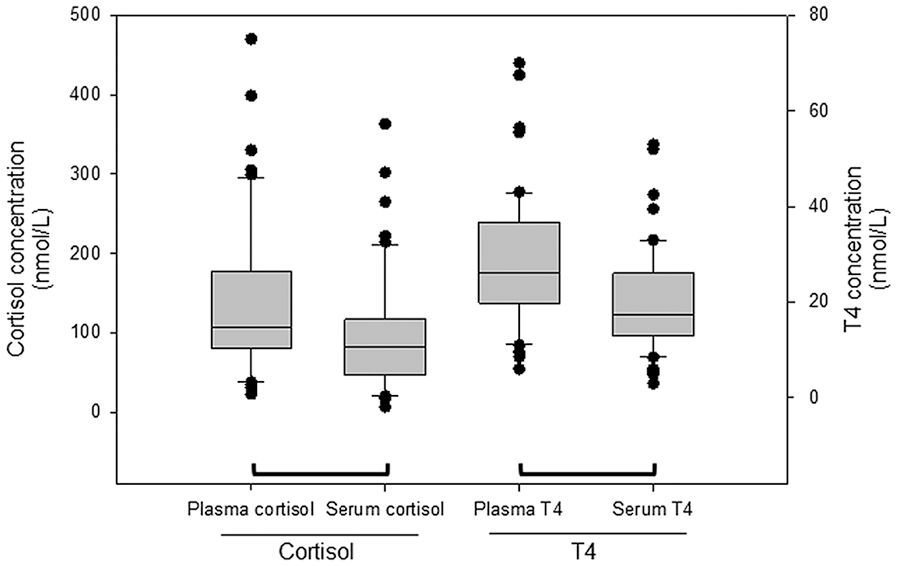
Cortisol and T4 concentrations measured in EDTA-plasma and serum (n = 50 dogs). Values connected by a bracket are significantly different (Wilcoxon signed-rank test for cortisol data, p < 0.001; paired t-test for T4 data, p < 0.001). Each box represents the interquartile (i.e., 25–75th percentile) range; the horizontal line within the box represents the median value; the bars represent the 10–90th percentile; the circles represent outlying datum points.
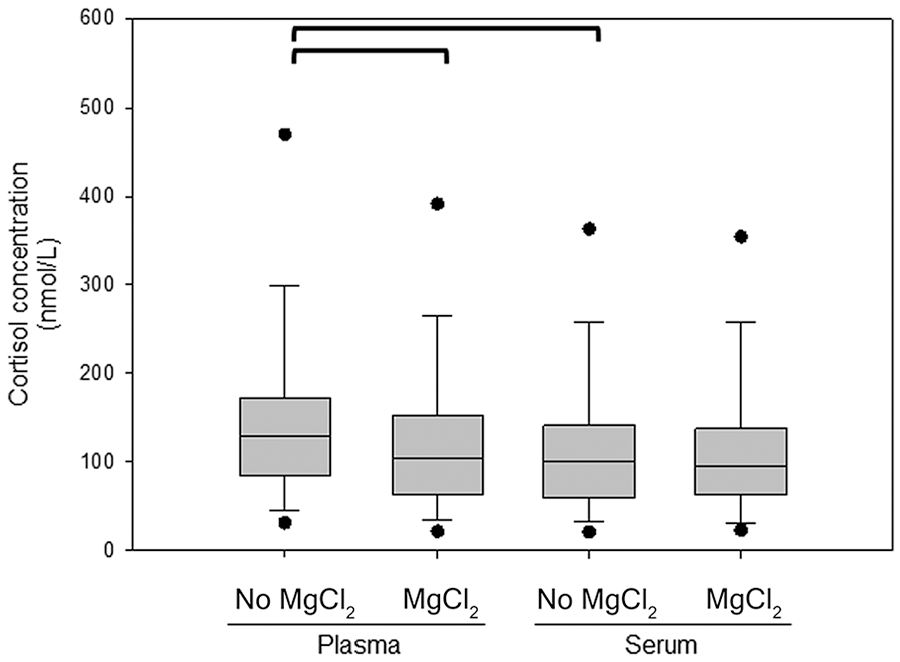
Cortisol concentrations measured in EDTA-plasma and serum with and without 5 mM MgCl2. Values connected by a bracket are significantly different (p < 0.001). Concentrations in plasma with and without MgCl2 and in serum without MgCl2 were compared using a one-way repeated measures ANOVA on ranks. A post-hoc test using the Dunnett method was performed with the concentrations in serum without added MgCl2 as the control group. Cortisol concentrations in serum with and without MgCl2 were compared using a Wilcoxon signed-rank test. See legend of Fig. 1 for additional information.
The T4 concentrations measured in EDTA-plasma were significantly higher than in serum (p < 0.001; Fig. 1). The median difference between the T4 concentrations in serum and EDTA-plasma was 43.7% (range: 0–183%). In the subgroup of 15 dogs for which MgCl2 was added, T4 concentrations were significantly different between plasma and serum samples if MgCl2 was not added (p < 0.001; Fig. 3). The addition of MgCl2 to EDTA-plasma did not significantly change the measured T4 concentration (p = 0.44), and the median difference between the T4 concentrations in serum and EDTA-plasma with the addition of MgCl2 was 43.5% (range: 0–65.4%). After the addition of MgCl2, the T4 concentrations measured in EDTA-plasma were still significantly different from serum (p < 0.001). The T4 concentrations measured in serum with and without MgCl2 were not significantly different (p = 0.38).
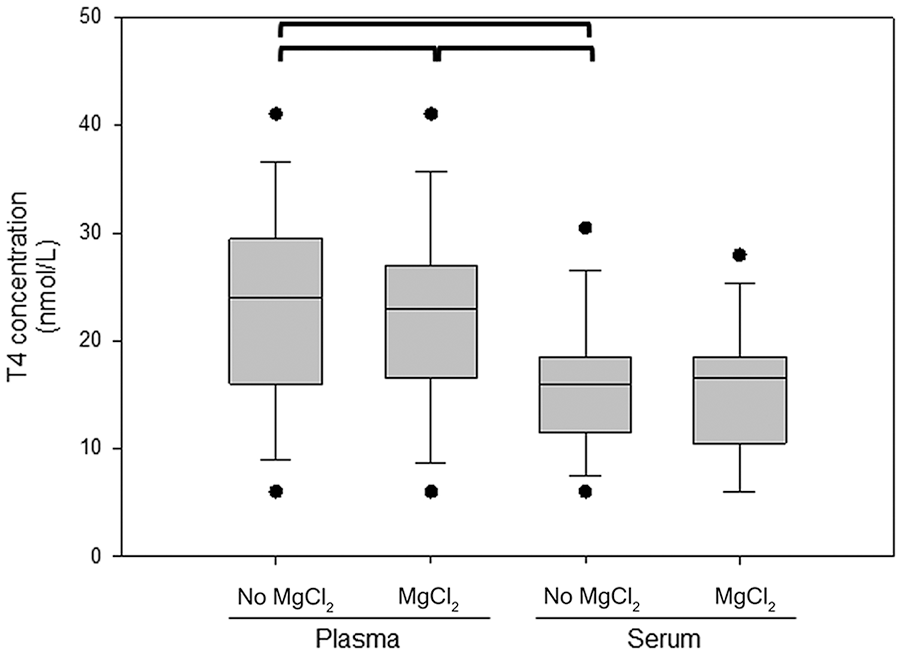
T4 concentrations measured in EDTA-plasma and serum with and without 5 mM MgCl2. Values connected by a bracket are significantly different (p < 0.001). Concentrations in plasma with and without MgCl2 and in serum without MgCl2 were compared using a one-way repeated measures ANOVA. A post-hoc test using the Dunnett method was performed with the concentrations in serum without added MgCl2 as the control group. T4 concentrations in serum with and without MgCl2 were compared using a paired t-test. See legend of Fig. 1 for additional information.
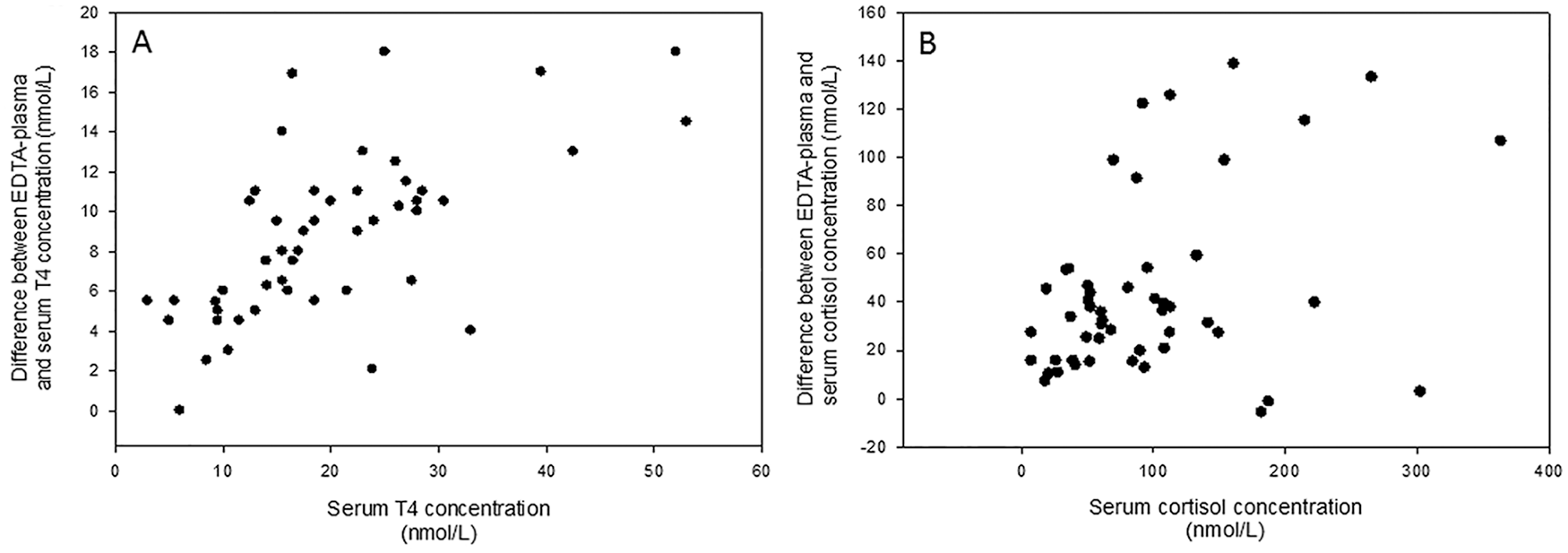
To determine why the addition of MgCl2 had different effects on the measurement of cortisol and T4, we assessed correlations between the hormone concentration in serum and the difference between serum and plasma. Given that the same samples were used to measure cortisol and T4, if the only factor affecting the plasma-EDTA concentration was the amount of EDTA, the correlation should have been the same for both hormones. The calculated difference between the serum and EDTA-plasma concentrations for cortisol was significantly correlated with the serum concentration for cortisol (p = 0.008, r = 0.372). The difference between the serum and EDTA-plasma concentrations for T4 was significantly correlated with the serum concentration for T4 (p = < 0.001, r = 0.639; Fig. 4). The correlation coefficients were not significantly different from each other (p = 0.077).
Discussion
Cortisol and T4 concentrations are commonly evaluated in canine patients. The presence of EDTA can falsely elevate the cortisol concentrations as measured by CEIA technology if sample tubes are underfilled. 10 We found that use of EDTA-plasma to measure cortisol if MgCl2 is not added or to measure T4 could lead to false interpretations.
Alkaline phosphatase (AP) is the reporter enzyme used in the CEIA technology that we employed. 1 It is a metalloenzyme that contains zinc and magnesium ions, which are necessary for its activity. 7 EDTA, if present, chelates zinc atoms and inhibits AP activity. 6 In the CEIA for cortisol and T4, AP generates light in an amount inversely proportional to the hormone concentration; the less light generated, the higher the apparent hormone concentration. Because EDTA decreases light generation, its presence leads to the incorrect interpretation that the sample contains a greater hormone concentration. 10 Addition of MgCl2 to EDTA-plasma samples theoretically could overcome the effect of EDTA because the magnesium would displace the zinc from EDTA, allowing the zinc to bind AP and enzyme activity to return. Indeed, MgCl2 overcame the effect of EDTA on measurement of cortisol in dog serum to which EDTA had been added 10 and of parathyroid hormone in human EDTA-plasma. 8 We found that addition of MgCl2 to dog EDTA-plasma samples overcame the effects of EDTA when measuring cortisol, but not when measuring T4.
The reason for the difference between T4 and cortisol is unclear. The correlation between calculated differences in EDTA-plasma and serum concentrations was not significantly different for T4 and cortisol. Given that the same samples were used to measure cortisol and T4, if the only factor affecting the plasma-EDTA concentration was the amount of EDTA, the correlation should have been the same for both hormones. Because the difference between the correlations approached significance (p = 0.077), it is possible that the interaction between the hormone and the antibody may be different for the 2 hormones. We chose a concentration of 5 mM MgCl2 given that higher concentrations negatively affect the cortisol assay (i.e., decrease the apparent concentration of cortisol). 10 It is possible that higher MgCl2 concentrations are needed to overcome the effect of EDTA for T4 samples while not interfering with the assay.
One weakness of our study was lack of uniformity in EDTA concentrations in the EDTA-plasma tubes. Quantitation of EDTA in plasma requires methodologies such as capillary electrophoresis–mass spectrometry. 13 Although addition of MgCl2 overcame the effects of EDTA in our study overall, we are not confident that its addition would be effective for all samples. Addition of 5 mM MgCl2 should be sufficient if a tube is at least half-filled. 10 If a tube is filled to < 50% of maximum, addition of 5 mM MgCl2 may not be adequate. Unfortunately, in a clinical setting, the amount of tube filling is often unknown. Thus, even if MgCl2 is added to EDTA-plasma for cortisol analysis by CEIA, and if the reported concentration is near an important decision-making threshold, repeat testing or further investigation may be warranted (e.g., the use of a baseline cortisol concentration of 55 nmol/L [2 µg/dL] to determine if further testing for hypoadrenocorticism is warranted).5,9 Another limitation of the study was the relatively small number of dogs used.
EDTA-plasma cannot be used to measure T4 by CEIA based on current knowledge. Cortisol concentrations are most stable in EDTA-plasma, 3 and EDTA-plasma can be used without assay modification if the tube is completely filled. 10 If tubes are filled to at least 50% of maximum, EDTA-plasma can still be used to measure cortisol by CEIA provided that the laboratory is aware of the underfilling and can add MgCl2 to the assay. Samples for measurement of endogenous ACTH require special handling and should be collected in EDTA tubes. 4 Further study is warranted to determine the effect of EDTA on the apparent concentration of endogenous ACTH when measured by CEIA and whether addition of MgCl2 to the assay is warranted.
Supplemental Material
Supplemental_material – Supplemental material for Effect of EDTA on measurement of cortisol and thyroxine by chemiluminescent enzyme immunoassay in dogs
Supplemental material, Supplemental_material for Effect of EDTA on measurement of cortisol and thyroxine by chemiluminescent enzyme immunoassay in dogs by Dana A. Schechter, Hollie P. Lee, Robert J. Kemppainen and Ellen N. Behrend in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Dr. Philippe Gaillard for advice regarding statistical evaluation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
