Abstract
We determined saliva cortisol concentrations (SCCs) in samples from 36 dogs using a chemiluminescence immunoassay (CLIA) and liquid chromatography–tandem mass spectrometry (LC-MS/MS) before and after a 10-min veterinary visit. The median cortisol concentration of the 77 samples analyzed with CLIA was 1.34 nmol/L, and the concentration of samples analyzed with LC-MS/MS was 1.30 nmol/L. With an intercept (a) of −0.06 (95% CI: –0.16 to 0.02) and a slope (b) of 0.97 (95% CI: 0.90–0.97) the Passing–Bablok regression revealed that there were no significant systematic and proportional differences between the 2 methods. In the Bland–Altman analysis, the bias was 0.08 nmol/L (95% CI: –0.38 to 0.54 nmol/L) between the 2 methods. Comparing the hormone levels in the saliva of 36 dogs before and after a veterinary visit, the CLIA method had a mean difference of 0.03 nmol/L (95% CI: –1.72 to 4.75 nmol/L; p = 0.35) and the LC-MS/MS method a mean difference of 0.12 nmol/L (95% CI 95%: –2.43 to 4.40 nmol/L; p = 0.62). Comparison of cortisol changes before (p = 0.86) and after (p = 0.79) the veterinary visit revealed no differences in using either the CLIA or LC-MS/MS methods. There was also no difference in the assessment of hormone increase or decrease between CLIA (p = 0.72) and LC-MS/MS (p = 0.17). Overall, the measurement of SCC in dogs by either CLIA or LC-MS/MS provided comparable results.
Cortisol is a hormone that is regulated via the hypothalamic–pituitary–adrenal (HPA) axis.15,16 Cortisol is involved in energy metabolism by, among other actions, increasing gluconeogenesis and activating lipolysis. 13 Moreover, it is involved in water and electrolyte balance and thus also influences blood pressure. 34 Cortisol increases the synthesis, and enhances the vasopressor effect, of angiotensinogen. 34 In the kidneys, cortisol increases the glomerular filtration rate. 34 In addition, cortisol has anti-inflammatory and immunosuppressive effects; it inhibits immunoglobulin synthesis, induces apoptosis of lymphocytes, and inhibits the production of inflammatory cytokines.26,27,32,35,36
Cortisol is a well-known analyte in both human and veterinary medicine that is used to assess physical and psychologic stress and pain.4,7 In dogs and cats, psychologic stress caused by an unfamiliar environment or separation from the owner, for example, is a widespread problem in everyday life.2,19 A visit to the veterinary clinic is a particularly stressful situation for some animals, 14 although it is often unavoidable. For example, dogs whose blood was taken in an unfamiliar environment were found to have a greater stress response, based on increased cortisol concentration in their serum, than dogs who were used to having their blood taken and were, therefore, less stressed. 28 In veterinary medicine, cortisol concentrations in the plasma and saliva of dogs are correlated, a correlation also well known in human medicine. 33 For example, 5–10% of the plasma concentrations of free cortisol, which is not bound to proteins, can also be detected in saliva. 33 It is beneficial to have an accurate, rapid test for cortisol concentrations in a relatively noninvasive sample such as saliva to rule out hypoadrenocorticism or to assess animal stress.
The determination of cortisol in dog saliva has been carried out in clinical studies, such as by radioimmunoassay (RIA),25,33 liquid chromatography–mass spectrometry (LC-MS), 23 competitive chemiluminescence enzyme immunoassay (CLIA), 5 or ELISA. 38 In contrast to CLIA, which uses chemiluminescence for detection, ELISA uses an enzyme-linked system; the enzyme catalyzes a reaction that produces a color change, which is measured to determine the amount of the target substance. High-performance liquid chromatography (HPLC) and liquid chromatography–tandem mass spectrometry (LC-MS/MS) are the methods of choice for the determination of cortisol in blood, saliva, and urine, as they are the most sensitive and specific in human medicine.8,30 In human medicine, immunoassays have been validated against LC-MS/MS assays. 6 To date, there have been no reported studies comparing the measurement of saliva cortisol concentrations (SCCs) in dogs using the CLIA and LC-MS methods. Therefore, we compared SCC in dogs by CLIA and LC-MS, and assessed the variation of SCC pre and post veterinary consultation (within each method and between the 2 methods).
Materials and methods
Study design
Our study was approved by the Ethics Committee of the Faculty of Veterinary Medicine, Ludwig-Maximilians-Universität München (AZ 282-18-03-2021). Dogs were included in our study if they had no known pre-existing disease conditions and their owners reported that they had been stressed at the veterinary clinic. We included 36 dogs in our study, 23 females and 13 males, with a mean age of 7 y (range: 2–14 y) and a mean weight of 20 kg (range: 5–42 kg). The animals were fasted for 2 h before the visit. The standardized veterinary visit (10 min on the treatment table) included a general examination (assessment of general condition, determination of capillary refill time, auscultation of heart and lungs, palpation of lymph nodes and abdomen, and measurement of rectal body temperature).
A saliva sample was collected from each dog by a veterinarian both before and after the general examination for our cortisol measurement study. Sample 1 was taken in front of the clinic where dogs were unstressed; sample 2 was taken immediately after the 10-min examination on the treatment table. Two days later, each dog underwent a second standardized veterinary visit, in which cortisol was again measured in the dogs’ saliva before (sample 3) and after the veterinary visit (sample 4) with the same standardized procedure, giving a total of 4 samples per dog (n = 77 of 144 included). A saliva sample for comparison between the 2 methods was included if sufficient saliva could be obtained for the CLIA and LC-MS/MS methods (250 µL). Where it was possible to analyze both pre- and post-test samples from the same dog by CLIA and LC-MS/MS (n = 20), the hormone concentrations measured by both methods was also compared.
A commercial salivette (Salimetrics) remained in the patient’s mouth for ~60 s, was then placed in an appropriate tube (Salimetrics), centrifuged, transferred to an Eppendorf tube using a pipette, and stored at −20°C up to 6 mo until analyzed. Samples were sent refrigerated to the Faculty of Psychology at the Technical University of Dresden, Germany, where cortisol measurements were performed by a competitive chemiluminescence immunoassay (CLIA IBL cortisol saliva ELISA, cat. 30172091; Tecan) using an absorbance microplate reader (Sunrise; Tecan) and LC-MS/MS as reported in previous studies. 10
The CLIA is a variation of an ELISA, namely a competitive assay with a luminescent signal. The unknown amount of antigen (cortisol) in the sample and a known amount of enzyme-labeled antigen (E-Ag) compete for the binding sites of the antibody bound to the wells of the microtiter strips. After incubation, unbound E-Ag is removed by washing. The intensity of the luminescence emitted after the addition of the luminescent substrate solution is inversely proportional to the antigen concentration in the samples. The results for the samples can be determined directly from the standard curve. 21 The CLIA was validated by the manufacturer for human serum cortisol; the between-run imprecision had a CV of 2.9–3.9% at 0.007–0.063 μg/dL, raising to 5.0% at 0.003 μg/dL.21,23
The LC-MS/MS that we used consisted of a HPLC coupled to a MS (C-20AD pumps; Shimadzu), SIL-20AC autosampler (Shimadzu), column temperature oven (CTO-20AC; Shimadzu), triple quadrupole tandem mass spectrometer (API 5000 Turbo-ion-spray; ABSciex), column (4.6 × 50 mm, Chromolith Speed ROD RP-18e LC; Merck), and LC column (3.0 × 75 mm, 2.2 µm, Shim-pack XR-ODS; Shimadzu). The LC separates the compounds to be analyzed based on their rates of adsorption to a columnar matrix; the MS detects, identifies, and quantifies the compounds based on specific multiple reaction monitoring settings derived from individual compound mass:charge ratios. 3 The internal standard was a deuterated cortisol. LC-MS/MS was performed in the same laboratory using the same chemicals, reagents, and instrumentation as well as liquid chromatography methodology. 10 This method has also been developed based on human saliva. Deuterated internal standard samples of cortisol-d4 were obtained from Toronto Research Chemicals. Calibration standards were prepared by spiking blank samples with standard solutions to final concentrations of 0, 0.001, 0.01, 0.05, 0.1, 0.5, 1, 5, and 10 ng/mL for cortisol. Mean intra-assay CV was 8.2%, and mean inter-assay CV was 7.3%. 10 To evaluate the matrix effect and method recovery, 3 blank saliva samples with 5 replicates each were spiked with pure standard samples at 3 different concentrations. The reaction was compared to the response of a neat standard solution.
Statistical analysis
Cortisol data were log-transformed due to a non-normal distribution and studied via robust mixed-effects models to compare cortisol concentration between either CLIA and LC-MS or between times (before and after the veterinary visit) in CLIA and LC-MS groups. After log transformation, the following model assumptions were investigated: 1) the normality of results was checked by the Shapiro–Wilk normality test, 12 2) the homogeneity of variances between groups was checked with the Bartlett test, 17 and 3) the heteroskedasticity (constancy of error variance) was checked with the Breusch–Pagan test. 18 Homogeneity of variances is checked between samples; heteroskedasticity checks whether the residuals of the model on the y-axis spread with increasing fitted values on the x-axis. Thus, we considered them separate assumptions and checked them via a “performance” package (R statistical software, http://www.r-project.org/). If the assumptions were satisfied, we used a mixed-effect model. 11 If the assumptions were not satisfied, a robust mixed-effect model regression should have been applied. 9 However, because assumptions were not satisfied, all analyses were conducted via the (parametric) robust approach, which is based on the robustification of the scoring equations and an application of the Design Adaptive Scale approach.20,22 All differences between particular groups, namely
SCC measured with CLIA vs. with LC-MS/MS
SCC measured before the veterinary visit using CLIA vs. LC-MS/MS
SCC measured after the veterinary visit using CLIA vs. LC-MS/MS
SCC measured before vs. after the veterinary visit using CLIA
SCC measured before vs. after the veterinary visit using LC-MS/MS
were assessed after model-fitting by the estimated marginal means (R package - emmeans). The Tukey p-value correction for multiple comparisons was applied; the alpha (significance) level of 0.05 remained unchanged. Passing–Bablok regression and Bland–Altman analysis were applied to compare 2 groups, CLIA and LC-MS/MS. Results with p ≤ 0.05 were considered statistically significant; results with p of 0.05–0.1 were considered a tendency. In the case of multiple comparisons, a correction was applied as a p-value adjustment, instead of adjusting the alpha level, so that each p-value was multiplied by the number of tests, while the alpha level of significance was left unchanged. Data analysis was performed using R v.4.3.1 (https://www.r-project.org/).
Results
Comparison of SCC (n = 77) using CLIA and LC-MS/MS
A direct comparison of cortisol measurements using CLIA and LC-MS/MS was carried out on 77 saliva samples (Fig. 1); the median SCC of all 77 samples analyzed with CLIA was 1.34 nmol/L and that of the 77 samples analyzed with LC-MS/MS was 1.30 nmol/L. According to the Bland–Altman analysis, there was a bias of 0.08 (95% CI: –0.38 to 0.54) between the 2 methods. The Passing–Bablok regression analysis revealed an intercept (a) of 0.06 (95% CI: 0.16–0.02) and a slope (b) of 0.97 (95% CI: 0.90–1.05; Fig. 2). Consequently, there were no systematic and proportional differences between the 2 methods.
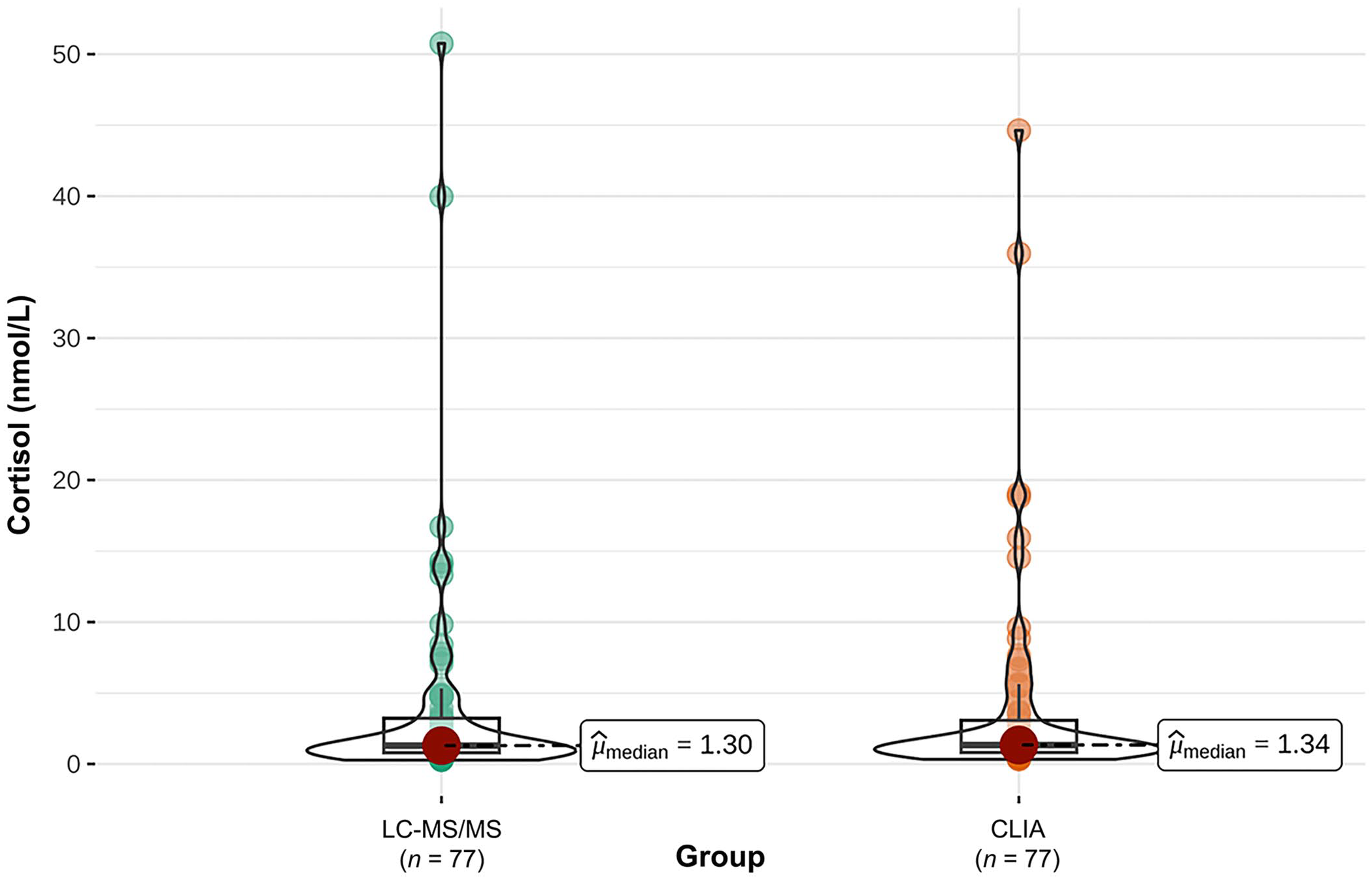
Cortisol concentration (nmol/L) in 77 dog saliva samples evaluated by liquid chromatography–tandem mass spectrometry (LC-MS/MS) and by chemiluminescence immunoassay (CLIA). Central bars = medians.
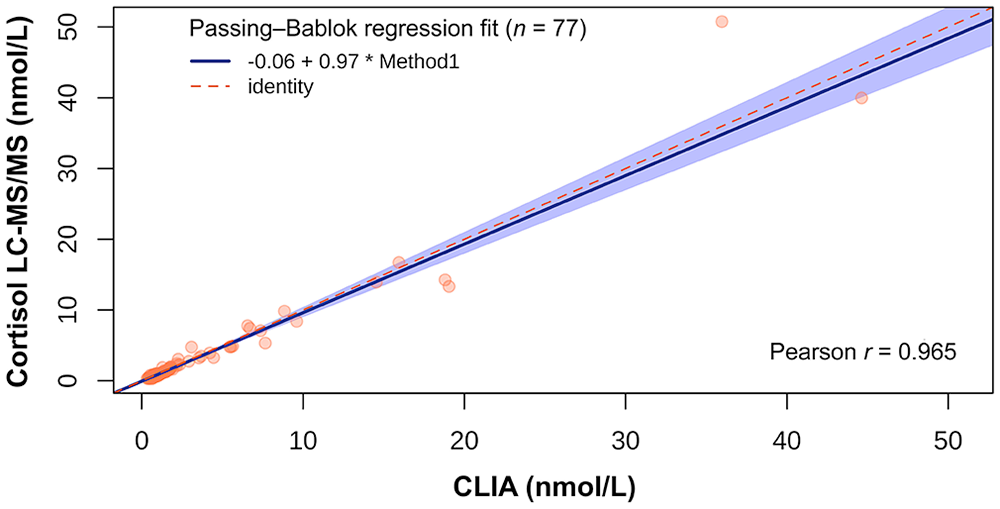
Passing–Bablok regression analysis of saliva cortisol concentrations of 36 dogs using chemiluminescence immunoassay (CLIA) and liquid chromatography–tandem mass spectrometry (LC-MS/MS).
Comparison of SCC fluctuations (n = 20) using CLIA and LC-MS/MS
Comparing the SCC before and after the 20 veterinary visits, the CLIA method had a mean difference of 0.03 nmol/L (95% CI: –1.72 to 4.75; p = 0.35) and the LC-MS/MS method a difference of 0.12 nmol/L (95% CI: –2.43 to 4.40; p = 0.62). At the time before the veterinary visit, the difference of SCC (log-transformed) between CLIA and LC-MS/MS was 0.102 nmol/L (95% CI: 0.082–0.466; SE = 0.14; p = 0.465) and after the veterinary visit the difference was 0.038 nmol/L (95% CI: –0.223 to 0.325; SE = 0.14; p = 0.783; Fig. 3). Comparison of cortisol changes before (p = 0.86) and after (p = 0.79) the veterinary visit revealed no differences by using either the CLIA or LC-MS/MS methods. There was also no difference in the assessment of hormone progression between CLIA (p = 0.17) and LC-MS/MS (p = 0.72). In the Bland–Altman blot (Fig. 4), despite a few outliers, the 2 methods appear to be comparable.
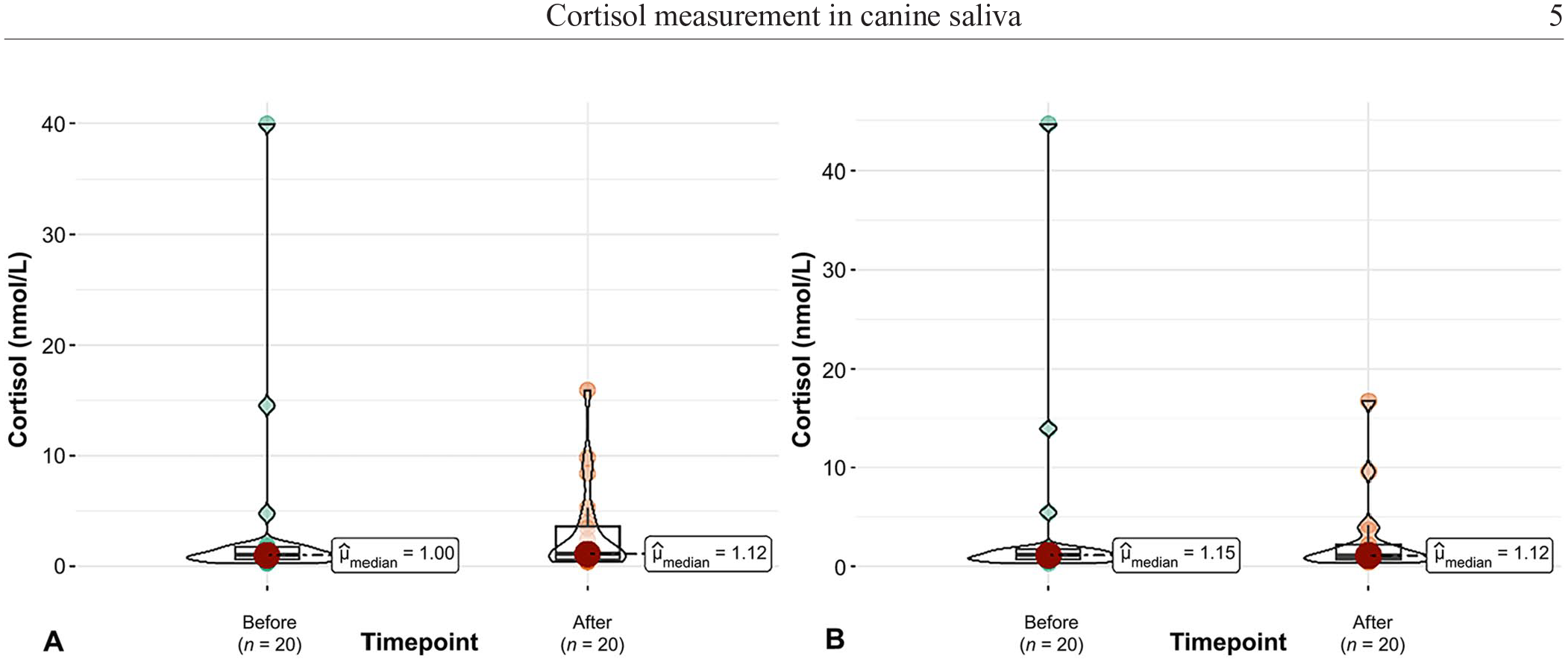
Cortisol concentrations (nmol/L) in the saliva of 20 dogs before and after a veterinary visit, determined by
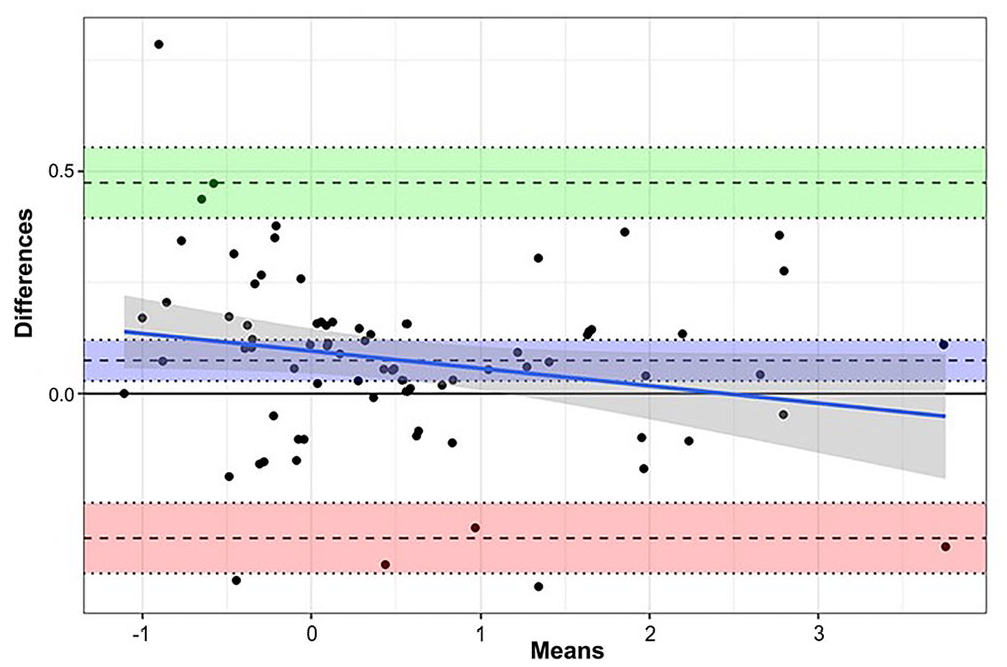
Bland–Altman comparison of saliva cortisol concentrations of 36 dogs determined by chemiluminescence immunoassay and liquid chromatography–tandem mass spectrometry.
Discussion
Overall, the Passing–Bablok regression revealed that there were no significant systematic and proportional differences between the CLIA and LC-MS/MS methods. Comparable studies on canine SCC from other researchers are not available. We did not investigate whether the slight difference between CLIA and LC-MS/MS for the determination of basal cortisol levels is clinically relevant. To date, there are no established RIs for basal cortisol in dog saliva. However, as the diagnosis of hyperadrenocorticism is mainly based on dynamic function tests, such as the ACTH stimulation test and the low-dose dexamethasone suppression (LDDS) test, clinical relevance of these differences is unlikely.
Cortisol measurement in children’s saliva by immunoassay yielded 2.39 times higher values than by LC-MS/MS 1 and thus significantly higher than in our study; such differences may be due to different methods or manufacturers. In contrast, in our study, despite some outliers in the Bland–Altman blot, the 2 methods appear to be comparable. Unfortunately, there is a lack of comparable literature on analyses of canine saliva. As part of the development of the LC-MS/MS method that we used, others 10 determined an intra-assay CV of 4.3–10.8% using 7 steroid hormones, including cortisol, although only human saliva was analyzed. The authors reported that cortisol had positive matrix effect behavior, which suggests that the respective responses in saliva were higher than those in the mobile phase, indicating enhancement effects of ionization, which could be largely corrected by the use of internal standards. In another human medical study in which salivary cortisol was analyzed using LC-MS/MS, the intra-assay CV was 4.3–11% 30 ; differences in SCC are therefore conceivable.
In 2012, 24 in a human medical study of cortisol in saliva, a nonlinear relationship was described between various immunoassays and LC-MS/MS results. Cross-reactions between matrix proteins in saliva and antibodies in immunoassays were identified as the main reason for the discrepancy in results, which can have a positive bias on the determination of cortisol concentrations. The authors of that study therefore recommended that LC-MS/MS should be preferred to immunoassays or that cortisol concentrations measured by immunoassays should be adjusted using appropriate conversion tables. 24
Using a combination of single-line solid-phase extraction and LC-MS/MS, 10 particularly high sensitivity and specificity were achieved for cortisol measurement; matrix effects were 6–14% but could be corrected using an internal standard. We used the same procedure for the saliva samples in our study; thus, the matrix effects that we found were negligible.
For the CLIA method that we used, cross-reactivity of 1.73% was reported by the manufacturer. Studies discuss the clinical relevance of cross-reactivity. 21 Cross-reactivity may be the reason for different results compared to the LC-MS/MS method. 21
There are clear differences in the metabolome of dog and human saliva; 25 lipids can be detected in dog saliva that cannot be detected in human saliva. 31 Such physiologic differences highlight the need for species-specific validation of methods for the determination of cortisol in saliva.
The advantage of a saliva sample is that it is less invasive than a blood sample and less stressful for most patients, especially in veterinary medicine. In human medicine, salivary cortisol is used to diagnose Cushing disease and Addison disease. For example, blood samples have been replaced in clinical studies by saliva samples as part of a LDDS test or an ACTH stimulation test. 29 Such studies have not yet been reported in veterinary medicine. However, a clinical study in dogs showed that patients with hypercortisolism had significantly higher salivary cortisol concentrations than healthy dogs. 37 Cortisol was measured in blood and saliva by immunoassay; the intra-assay variability was 5–17.7% for the measurement of salivary cortisol. 37
There is a lack of specific RIs in small animal medicine that can be used to diagnose diseases such as hypo- and hyperadrenocorticism using salivary cortisol. The next step would therefore be to establish RIs for SCC in healthy dogs using CLIA and/or LC-MS/MS. In follow-up studies, cortisol concentrations in blood and saliva could also be compared as part of ACTH and LDDS testing for hypo- or hyperadrenocorticism to investigate whether saliva can be used to discriminate between healthy and diseased dogs. Because such tests are primarily used to assess temporal changes, the CLIA method, which is usually less expensive, could be used.
We only examined SCC twice at 10-min intervals. Follow-up studies with additional times may be of interest to assess temporal changes in SCC. In addition to species, factors such as breed may also influence results.
Footnotes
Acknowledgements
We thank Prof. Clemens Kirschbaum and his team at the Technical University Dresden, Germany, who carried out the measurement of the SCC using CLIA and LC-MS/MS. We are grateful for this cooperation. Many thanks to Dr. Jeremie Korchia for his technical expertise and enormous support which contributed to the preparation and writing of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
