Abstract
Hormone assays that use a solid-phase, automated, chemiluminescent enzyme immunoassay (CEIA) with an alkaline phosphatase–tagged hormone or antibody as a reporter are performed on serum or EDTA plasma in our laboratory. CEIA cortisol results appeared to increase in the presence of excess EDTA. We investigated the effect of the addition of different amounts of EDTA on cortisol concentrations in pooled canine serum samples. The recommended EDTA plasma concentration of 4.1 mmol/L (1.8 mg/mL) did not alter cortisol concentrations when added to serum pools; however, the addition of ≥5.1 mmol/L (2.25 mg/mL) of EDTA increased apparent concentrations of cortisol. Supplementation of serum samples with MgCl2 to 5 mmol/L reversed the effect of EDTA up to a concentration of ~8.1 mmol/L (3.6 mg/mL). Our findings show that CEIA cortisol results on EDTA plasma can be artificially increased if the EDTA concentration exceeds 5.1 mmol/L.
The Endocrine Diagnostic Laboratory at the College of Veterinary Medicine, Auburn University (Auburn, AL) assays samples submitted by veterinarians for the diagnosis and monitoring of a variety of endocrine disorders. Samples may be in transit for several days when sent by mail. Consequently, determination of the sample type and shipping conditions that best preserve immunoreactive hormone concentrations is important. Although total thyroxine concentrations in serum or plasma remain stable for 5 d at −20, 4, or 25°C, cortisol concentrations measured by radioimmunoassay (RIA) are significantly affected by sample type as well as storage conditions. 2 In a prior study, cortisol declined in serum stored for 5 d at 4, 25, or 37°C, whereas concentrations were unchanged in dipotassium ethylenediaminetetraacetic acid (EDTA) plasma stored at 4 or 25°C. Thus, blood drawn for RIA cortisol assay is collected and submitted in EDTA plasma rather than serum at our laboratory.
Given the discontinuance in 2014 of the RIA kits, laboratories have converted to an automated, solid-phase, chemiluminescent enzyme immunoassay (CEIA; Immulite 1000, Siemens Healthcare Diagnostics, Tarrytown, NY) for several assays. 9 The CEIA for canine cortisol has been validated previously. 9 During the transition period in our laboratory, cortisol values obtained using RIA versus CEIA were compared and found to provide similar results. However, cortisol concentrations using the CEIA were substantially higher than those using the RIA in several samples submitted in EDTA Vacutainer tubes (Becton, Dickinson, Franklin Lakes, NJ). Discussion with the submitting client revealed that blood was collected into EDTA-containing tubes and the harvested plasma was decanted into another EDTA-containing tube. A prior publication demonstrated that excess amounts of EDTA reduced concentrations of parathyroid hormone in human plasma samples measured using the same CEIA. 6 These investigators were able to reverse this interference by adding magnesium sulfate to the Immulite assay reagent wedge. We hypothesized that the quantity of EDTA in plasma samples potentially affects cortisol values determined by CEIA, and that addition of magnesium could negate these changes.
High and low cortisol canine serum pools were produced by mixing serum samples submitted to our laboratory within the past 6 mo for cortisol testing. Experiments were performed using serum because the concentrations of EDTA in plasma samples received by the laboratory are unknown. Low pool samples were made from serum with <150 nmol/L cortisol and high pool from those >300 nmol/L. A moderate canine serum pool was also produced (mean ± SD; 183 ± 11 nmol/L).
When filled to optimal draw volume, the concentration of EDTA in plasma can be 3.2–5 mmol/L (1.4–2.2 mg/mL), 3 whereas in commonly used Vacutainer tubes, the final concentration is expected to be 4.1 mmol/L (1.8 mg/mL). K3EDTA (Alfa Aesar, Ward Hill, MA), similar to that contained in Vacutainer tubes, was added to the high and low canine serum pools to achieve EDTA concentrations of 0, 4.1, 5.1, 6.8, 10, and 20 mmol/L. Samples were then assayed for cortisol in replicates of 4 using CEIA. 9
In order to evaluate the impact of magnesium chloride on cortisol values, MgCl2 (Sigma-Aldrich, St. Louis, MO) solubilized in water to 1 mol/L was added to the high and low serum pools to produce concentrations of 0, 5, 6.7, 10, and 15 mmol/L. The ability of 5 mmol/L MgCl2 added to serum to negate the chelating effect of EDTA was investigated in the high cortisol pool. This concentration of MgCl2 was selected because it did not alter cortisol values measured in either high or low serum pools. To reduce effects of dilution after addition of the MgCl2 solution and to eliminate the need to add small volumes of MgCl2 solution to test samples, 7.5 µL of 0.1 mol/L MgCl2 containing red food color (McCormick, Hunt Valley, MD) included at 1:100 (vol:vol) was added to Immulite sample cups and dried down using a SpeedVac concentrator (Thermo Fisher Scientific, Waltham, MA). Serum samples (150 µL) for assay (n = 4 per treatment) were added to the cups and briefly vortexed. The red food dye (along with the MgCl2) immediately entered the serum sample solution subsequent to brief mixing, which ensured proper treatment of each sample.
The moderate cortisol pool sample was used to evaluate the effect of 50% optimal blood draw on cortisol values, with and without the addition of 5 mmol/L MgCl2. A 50% blood-filled EDTA tube would result in an estimated plasma EDTA concentration of 8.1 mmol/L (3.6 mg/mL).
Statistical analyses were performed (Prism 7, GraphPad Software, La Jolla, CA). Normality of the distribution was tested using the Shapiro–Wilk normality test. One-way ANOVA was used for analysis followed by the Dunnett multiple comparisons test. Two-way ANOVA was used to analyze data (main effects were EDTA or MgCl2), followed by Tukey multiple comparisons test (each result was compared with serum lacking both EDTA and MgCl2) and by Sidak multiple comparison test (no MgCl2 vs. with MgCl2 within each EDTA treatment).
The addition of EDTA at concentrations ≥5.1 mmol/L in the low cortisol serum pool or ≥6.8 mmol/L added to the high cortisol serum pool significantly increased the measured concentration of cortisol (Fig. 1). These concentrations are equivalent to filling a blood collection tube to 80% or 60% of the optimal capacity, respectively.
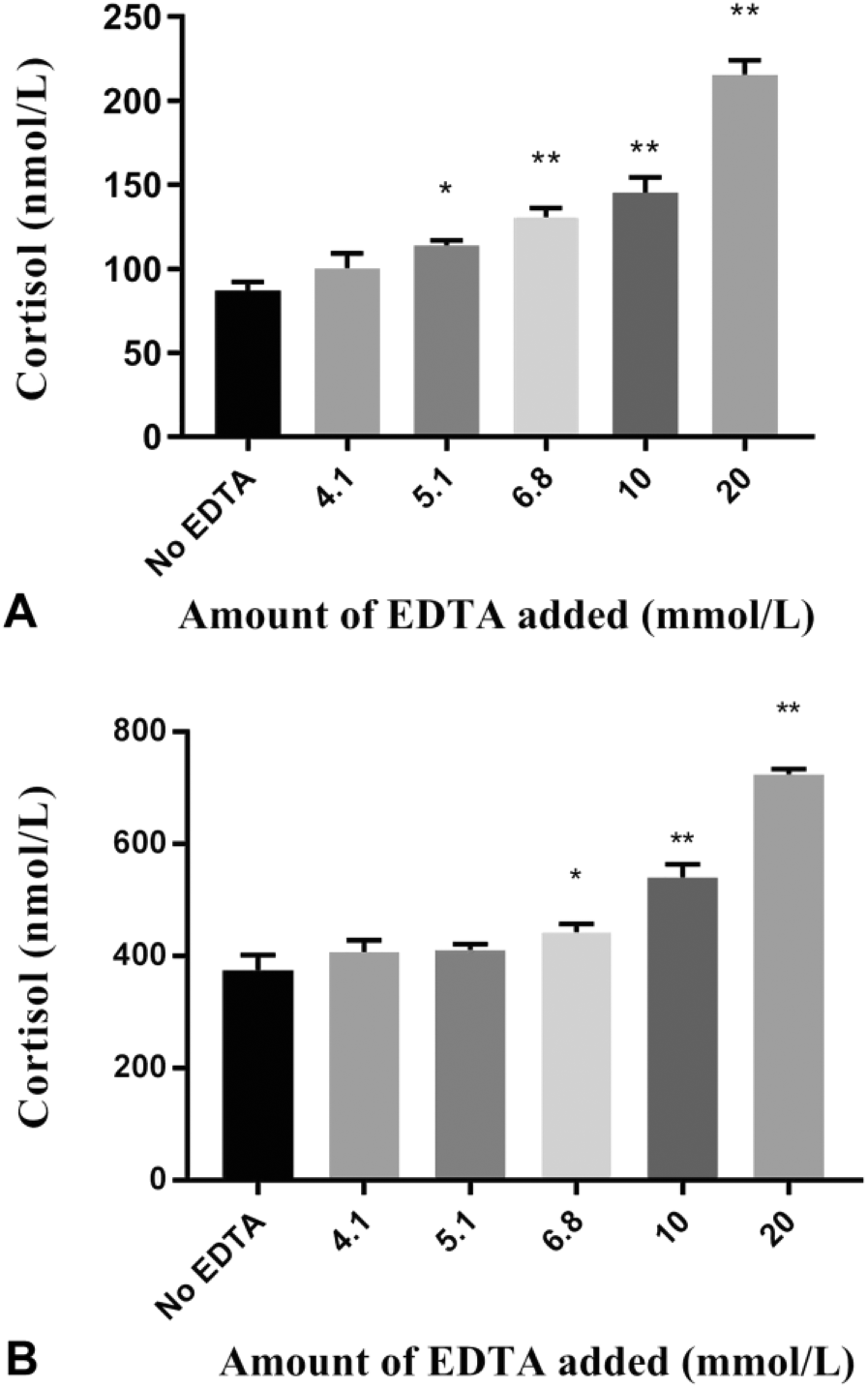
Immunoreactive cortisol concentrations (mean ± SD of 4 replicate measurements) determined using CEIA in
Addition of ≥6.7 mmol/L MgCl2 to the low cortisol pool, or 15 mmol/L MgCl2 to the high cortisol pool, significantly lowered the apparent cortisol concentration (Fig. 2).
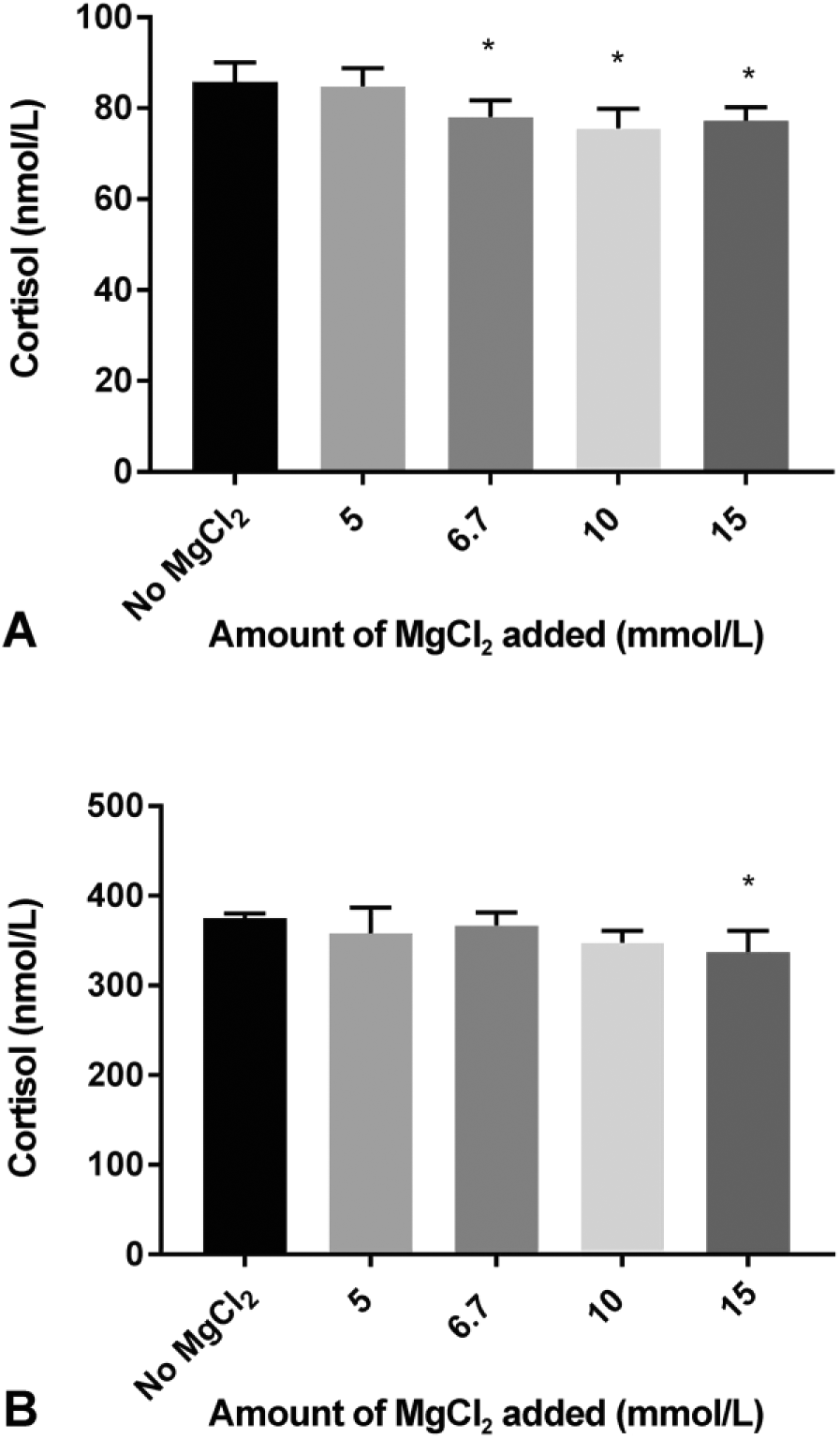
Immunoreactive cortisol concentrations (mean ± SD of 4 replicate measurements) determined using CEIA in
Addition of ≥6.8 mmol/L EDTA to the high cortisol serum pool significantly increased the apparent concentration of cortisol (Fig. 3). Supplementation of 5 mmol/L MgCl2 lowered the cortisol concentration in samples with 6.8 mmol/L EDTA to a value not different from the untreated serum pool. Addition of 5 mmol/L MgCl2 reduced the cortisol concentration in samples with 10 or 20 mmol/L EDTA, but the concentration in the resulting samples (samples containing 10 or 20 mmol/L EDTA plus MgCl2) was significantly (p < 0.01) greater than that in the untreated serum pool.
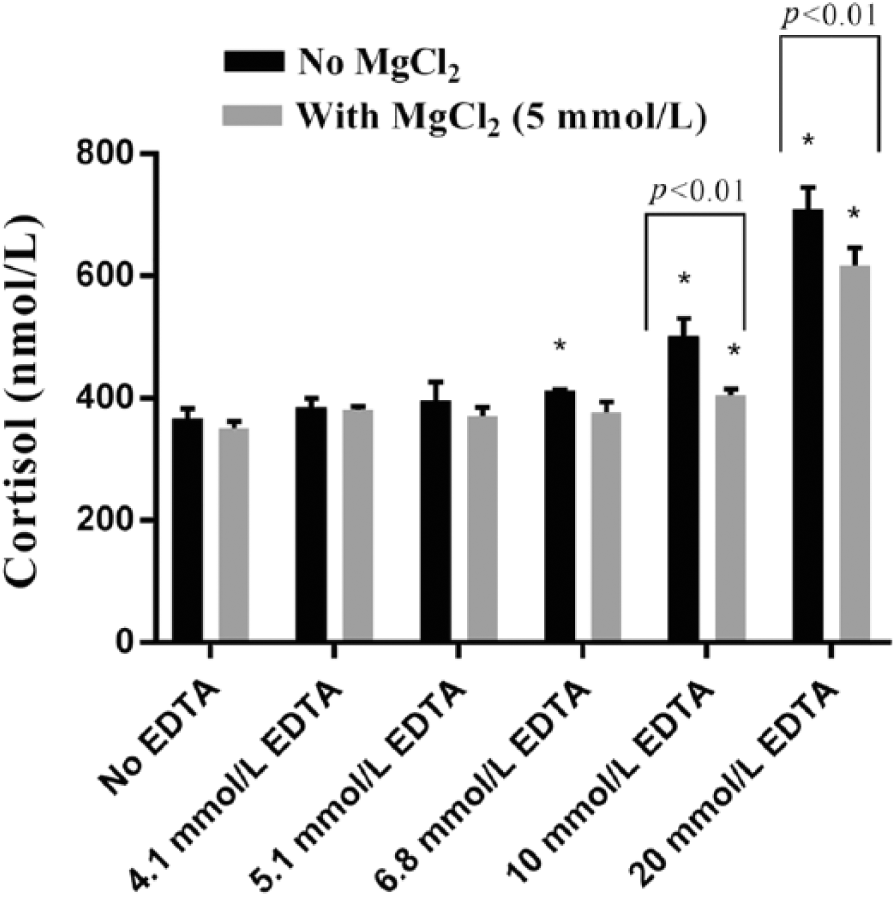
Immunoreactive cortisol concentrations (mean ± SD of 4 replicate measurements) determined using CEIA in a high canine serum pool with or without addition of EDTA and MgCl2. * = p < 0.05 or greater vs. no EDTA and no MgCl2 (first bar). Brackets indicate side-by-side comparison of results from samples with 10 or 20 mmol/L EDTA versus samples with the same amount of added EDTA together with MgCl2 (p < 0.01).
The addition of 8.1 mmol/L EDTA to the moderate cortisol pool significantly increased cortisol from 183 ± 11 to 218 ± 8.5 nmol/L (p < 0.001). However, when both 8.1 and 5 mmol/L MgCl2 were added, results were not significantly different from the untreated pool (192 ± 8.5 nmol/L, p = 0.32).
The CEIA employed in our study uses alkaline phosphatase (AP) as a reporter enzyme. 1 In the case of the cortisol assay, AP is attached to cortisol, and this complex competes with unbound cortisol in a patient sample for binding to an antibody bound to a bead. In this competitive assay, higher amounts of cortisol in the patient sample result in reduced amounts of cortisol–AP complex binding to the antibody; this in turn results in a lowered amount of light generation when a chemiluminescent substrate is added. 9 AP is a metalloenzyme containing both zinc and magnesium, which are necessary for activity. 5 It is generally accepted that EDTA mainly inactivates or inhibits the activity of AP by chelating zinc atoms. 4 In the case of the CEIA for cortisol, it is likely that the presence of a threshold concentration of EDTA chelates zinc (and/or magnesium) in AP, reducing its ability to mediate light generation, 9 leading to the incorrect interpretation that the sample contains a greater concentration of the hormone. A different type of CEIA employs 2 antibodies (2-site test), 1 of which is tagged with AP, whereas the other antibody is bound to a bead in the test unit. 9 In tests using this format, excess EDTA would be expected to result in falsely lower apparent hormone concentration as documented previously using the same CEIA instrument to measure parathyroid hormone in human samples. 6 In 2-site assays, light generation is directly related to the amount of hormone in a patient sample, rather than indirectly as in the cortisol assay.
In veterinary practice, optimal filling of blood collection tubes can be challenging. Practitioners may submit plasma samples for hormone measurement that contain EDTA in amounts above the recommended concentrations. We employed serum containing different concentrations of cortisol to simulate EDTA plasma samples containing excess EDTA. It is conceivable that different results might be obtained if whole blood was used, with the resulting plasma containing various amounts of EDTA.
The issue of introducing an error related to EDTA concentration in a test result using CEIA for cortisol measurement can be avoided by using serum samples for testing. Cortisol in canine serum, however, is subject to potentially greater loss of immunoreactivity than when collected in EDTA plasma.2,8 The rate of loss is dependent on time, storage temperature, and interestingly, the initial concentration of cortisol.2,7 Samples containing higher concentrations of cortisol show a greater rate of loss than those with lower concentrations.2,7 Overall, it would be advisable when submitting serum for canine cortisol measurement to use a rapid means of delivery and include cooling during shipment. When samples are submitted as EDTA plasma, practitioners should be aware of the necessity to fill blood collection tubes to optimal volume to avoid EDTA interference with the cortisol CEIA.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
