Abstract
A unilateral, malformed gnathotheca in a green-winged macaw (Ara chloropterus) was managed for 16 mo with periodic beak trims. Biopsy of the affected beak did not identify an underlying cause; however, at postmortem examination, a densely cellular neoplasm invaded the mandible from the beak, dissecting and effacing the cortex and trabecular bone and inciting periosteal proliferation. There was no evidence of metastasis. Neoplastic cells were strongly immunopositive for cytokeratin AE1/AE3, consistent with squamous cell carcinoma (SCC). A black-headed caique (Pionites melanocephalus) was presented with a similar clinical history, timeline, and gross appearance. Biopsy of the mandibular portion of the lesion resulted in a diagnosis of SCC. Medical management with toceranib at a palliative dose resulted in adverse side effects and was discontinued. Early diagnosis of mandibular SCC may allow for early intervention and development of treatment modalities. Our 2 cases suggest that the mandibular bone, not the beak, may be a preferred biopsy site in the diagnosis of mandibular SCC in psittacines.
Squamous cell carcinomas (SCCs) are relatively uncommon in avian species but are reported most frequently in cockatiels, Amazon parrots, conures, and budgerigars.4,14 Fewer cases of SCC are reported in poultry, hornbills, storks, eclectus parrots, and macaws.1,2,5–7,12,13 SCCs in birds typically arise from single or multiple sites in the skin, mucocutaneous junctions, uropygial gland, and upper gastrointestinal tract.3,14 SCCs originating at the beak occur rarely, but lead to chronic non-healing wounds, beak deformities (cracking, peeling, irregular growth, overgrowth), and potential difficulty in prehending food and water.6,12,14 These neoplasms also cause osteolysis of the underlying bone as a result of local invasion. SCCs often have necrotic areas, therefore secondary infections are common, and up to 22% of pet birds treated for SCC have concurrent fungal or bacterial infections.13,14 We describe herein the clinical history and pathologic findings associated with mandibular SCC in a green-winged macaw (Ara chloropterus) and a black-headed caique (Pionites melanocephalus).
A 22.5-y-old, female, green-winged macaw was treated multiple times over ~16 mo for a non-healing wound at the right gnathotheca. Initially, the macaw was presented because of a 2-cm² ulcerative defect at the right gnathotheca with caudal, yellow-to-brown keratin proliferation. No mandibular osteolysis was seen on radiographs. Several husbandry changes were implemented, including altered diet and increased ultraviolet light. In addition to the maintenance pelleted diet, leafy greens and vegetables, and treats, including nuts, fruits, and seeds, were added to make up 10% and 5% of the diet, respectively. Aerobic culture of the debrided wound produced heavy growths of Pseudomonas aeruginosa, Enterococcus gallinarum, and Klebsiella oxytoca. The wound was debrided every 4–6 wk over a year. In the first 4 mo, the beak appeared to grow over the exposed tissue, but then the lesion began to extend rapidly (Fig. 1). At 15 wk, biopsy of the lesion revealed heterophilic, ulcerative dermatitis and parakeratotic hyperkeratosis. The beak appeared to be healing, with granulation tissue forming between keratin layers 1 wk later. Amikacin (Zoetis; Parsippany-Troy Hills, NJ) and a chelating agent (Tris–EDTA; Tricide; Molecular Therapeutics, Athens, GA) solution (compounded by the UGA Veterinary Teaching Hospital pharmacy, Athens, GA) flushes over the wound every 8 h were implemented at this time. Minor debridement was continued monthly until ~1 y from initial presentation at which time tissue proliferation was noted between the keratin layers. The flushes were discontinued after 7 mo given the potential concern for a neoplastic process or fungal infection. The owner chose to continue debridement and medical management while monitoring food prehension and quality of life. Four months later, ~1 y after initial presentation, the macaw was anorexic for 2 d and was euthanized given its poor quality of life and progression of the lesion to encompass the right gnathotheca.
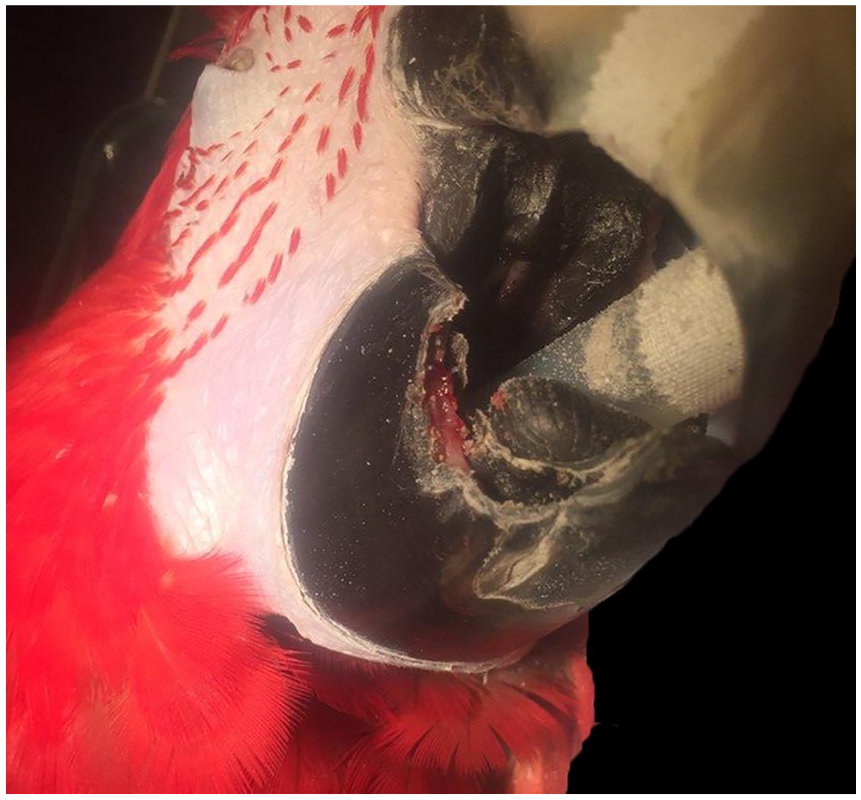
Ulcerative-to-proliferative lesion at the right gnathotheca of a green-winged macaw.
At postmortem examination, the macaw was thin (body condition score 2 of 5) with roughened, thickened, proliferative, irregular keratinization at the right gnathotheca and focally extensive, marked thickening of the right mandible (Fig. 2). Dissection revealed an irregular, off-white to pink, firm-to-hard, ill-defined, rounded, raised mass of ~2 cm diameter immediately caudal to the junction of the right gnathotheca and mandible. The surface of the mandible was mottled tan. No other significant changes were present at postmortem examination, and there was no evidence of metastasis.
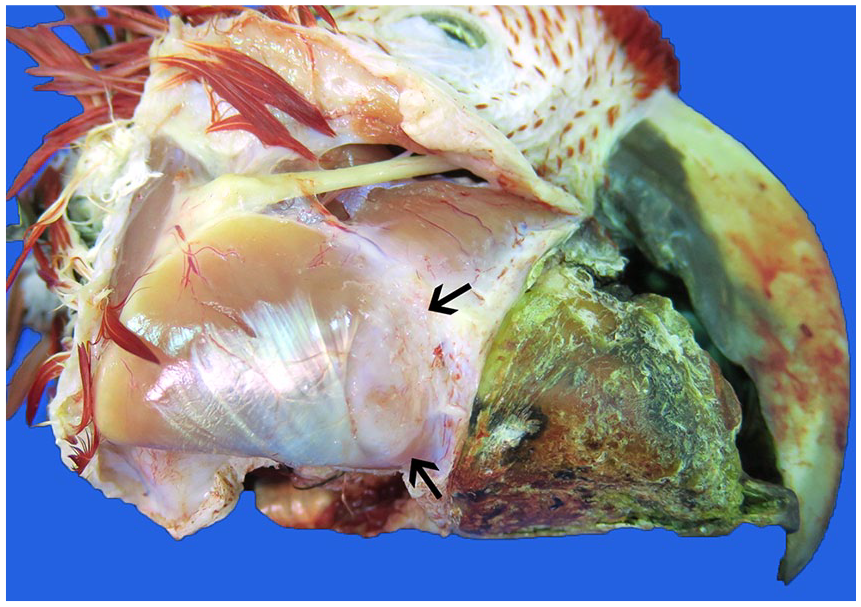
Irregularity of the right gnathotheca caused by mandibular squamous cell carcinoma. The mandibular surface is rounded and mottled by periosteal proliferation (arrows).
Histologically, the mass was unencapsulated and poorly delineated, with invasion and focally extensive effacement of cortical and trabecular mandibular bone (Fig. 3). At the junction of the mandible and beak, the mass produced abundant, disorganized, and degenerate, brightly eosinophilic keratin. The mass consisted of polygonal cells arranged in cords and islands with eosinophilic cytoplasm and central, vesicular nuclei with 1 or 2 basophilic nucleoli, which often surrounded central regions of keratin pearls. There was moderate anisocytosis and anisokaryosis with frequent single cell necrosis and a mitotic count of 27 in 2.37 mm2. Neoplastic cells were diffusely and strongly positive for pancytokeratin by immunohistochemistry (AE1/AE3; Cell Marque, Rocklin, CA; Supplementary Fig. 1). Moderate numbers of inflammatory cells infiltrated the mass, including heterophils, lymphocytes, plasma cells, and macrophages. The lesion was diagnosed as mandibular SCC with secondary gnathothecal disfiguration.
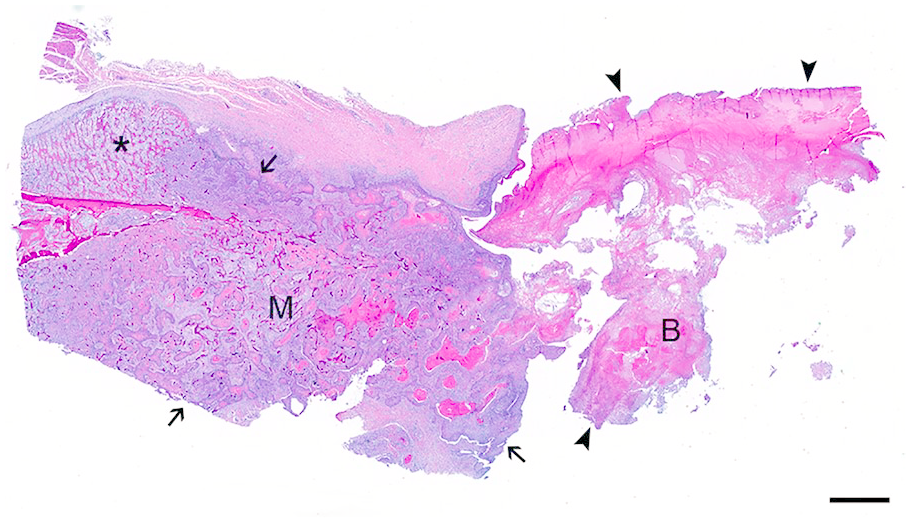
Subgross photomicrograph of the gnathotheca-mandibular junction in a green-winged macaw with mandibular squamous cell carcinoma. Gnathothecal keratin (B) is markedly degenerate and disorganized (arrowheads); the neoplasm is limited to the mandible (M), invades the cortical and trabecular bone (arrows), and incites periosteal proliferation (asterisk). H&E. Bar = 2 mm.
A second case was an 8-y-old, male black-headed caique with chronically progressive, proliferative disfiguration of the right gnathotheca (Fig. 4). Biopsy ~2 mo after initial presentation revealed inflammation and keratin with cocci. Additional biopsies of the beak and mandibular lesions were collected the next month, and a diagnosis of SCC was made. A computed tomography scan revealed a 1.3 × 1.2 × 0.8 cm mass associated with locally extensive mandibular lysis, deformation of the gnathotheca, and mild extension into the oral cavity. Surgical treatment options and palliative radiation therapy were declined given the potential risks and side effects. Toceranib (Palladia; Zoetis) was prescribed at 10 mg/kg once daily as a less aggressive treatment option. Treatment was discontinued after 2 mo because of hypoproteinemia and further progression of the lesion. Hypoproteinemia resolved 2 mo after cessation of treatment. As of 9 mo after diagnosis, the bird has a good quality of life and is maintaining weight with periodic beak trims.

Mandibular squamous cell carcinoma in a black-headed caique. Marked areas indicate biopsy sites with positive (O) and negative (X) detection of the neoplasm.
A potential link exists between hypovitaminosis A and SCC in avian species. 14 Pet birds are commonly fed seed-based diets, which are deficient in vitamin A, leading to hypovitaminosis A and squamous metaplasia of the upper digestive tract, which may progress to SCC if the condition is chronic and not corrected.8,10,14 Although it is possible that the birds in our report may have had historic vitamin A deficiency and squamous metaplasia of the upper digestive tract mucosa and salivary glands, this is speculative and neither were identified clinically or at postmortem examination. Although dietary changes were implemented at the initial presentation of the macaw with the goal to provide balanced nutrition and facilitate food prehension, neoplastic transformation may have already occurred. It is also possible that the initial wound represented a chronic infection and inflammatory process that initiated malignant transformation as the disease progressed, although this is considered less likely given the lack of infectious processes diagnosed clinically or at postmortem examination. 14 Similarly, chronic, persistent trauma and wounds may predispose to SCC and could have contributed to neoplastic transformation, although there was no indication of trauma in the clinical history.
The exact timeline of neoplastic transformation and progression in the green-winged macaw is unknown. Only keratin with superficial colonization by mixed bacteria was present in the biopsy; neoplastic cells were not detected. It is possible that tumor growth was responsible for the initial non-healing wound and infection, which progressed slowly initially and was managed easily with repeated debridement. Tumor growth accelerated to the point of becoming unmanageable. Additionally, the gross changes in the gnathotheca potentially resulted from disruption and dysregulation of the squamous epithelium that produces the keratin of the gnathotheca. The neoplasm in the macaw was located at the junction of the mandible and gnathotheca, extending into the mandible but not the gnathotheca. This pattern of detection reflects the lack of neoplastic invasion of the gnathotheca, potentially because of poor vascular supply within the beak. Eventually, sampling of the mandibular lesion in the black-headed caique led to the detection of the neoplasm. Despite the long clinical course of the disease, no evidence of metastasis was identified in other tissues, which is consistent with previous reports and suggests that early detection and treatment may allow for improved clinical outcome.6,13
Several treatment modalities are described for treating SCC in avian species, although optimal treatment regimens are lacking for most neoplasms. These therapies include chemotherapy, surgery, cryosurgery, radiation therapy, and electrosurgery. 9 The efficacy of these treatment modalities varies with several limiting factors. Surgical removal of SCC, particularly in the beak region, is technically challenging and may compromise quality of life depending on the size and invasiveness of the lesion.6,13 Hypoxia in SCC leads to necrosis and potentially infection, which can lead to poor responses and outcomes with radiation therapies. 10 Given the small number of cases, it is difficult to ascertain appropriate radiation or chemotherapeutic dosing, which may be insufficient for tumor recession. Although avian species tend to tolerate radiation treatment well, SCC in birds may not be as radiosensitive as those in other species, which can make complete remission without surgery difficult to attain.9,11 Intralesional chemotherapy with carboplatin or cisplatin has had mixed success. 9 In the case of the black-headed caique, palliative medical management with chemotherapy was unsuccessful in slowing disease progression and resulted in secondary complications.
Features suggestive of mandibular SCC in these 2 psittacines included unilateral thickening and disfigurement of the gnathotheca, as well as osteolysis and periosteal proliferation of the underlying mandible. Although potentially more invasive, biopsy of the osteolytic regions of the mandible instead of the beak may provide improved chances of detecting and diagnosing mandibular SCC in psittacines. Further work is needed to definitively determine the ideal biopsy site for diagnosis of mandibular SCC in birds. Early detection, combined with the prolonged course of disease and lack of metastasis in these and historic cases, might allow for the development of improved therapeutic techniques in future cases.
Supplemental Material
Supplemental_material_1 – Supplemental material for Mandibular squamous cell carcinoma in two psittacines
Supplemental material, Supplemental_material_1 for Mandibular squamous cell carcinoma in two psittacines by Morgan A. Halley, Justin M. Stilwell, Jessica R. Comolli, Stacey L. Wilkinson, Drury R. Reavill, Stephen Divers, Joerg Mayer and Daniel R. Rissi in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
