Abstract
An 18-year-old, neutered, male Vietnamese pot-bellied pig (Sus scrofa) was treated for chronic, intermittent nasal discharge and sneezing. The animal was diagnosed with severe periodontal disease (grade IV), an oronasal fistula, and multiple tooth root abscesses via dental examination and computed tomography of the skull. Dentistry was performed, including multiple tooth extractions, and antibiotic therapy was initiated. Eighteen months later, the animal was evaluated for lethargy, anorexia, and a firm, 12 cm × 12 cm mass between the 2 rami of the mandible. Laboratory testing revealed moderate anemia, severe leukocytosis, and hyperglobulinemia. Skull radiographs indicated osteomyelitis of the mandible and soft-tissue swelling. A fine-needle aspirate and biopsy were taken, and results were consistent with squamous cell carcinoma. Treatment with piroxicam and antibiotics was initiated as needed to control signs of pain and secondary infection, respectively. Three months after diagnosis, the pig was euthanized due to cachexia and severe depression secondary to squamous cell carcinoma. On postmortem examination, the right mandibular area contained multiple, coalescing, irregular masses extending from the ramus rostrally to the mandibular canine teeth and ventrally within the intermandibular space, completely obliterating the normal anatomy. An open midshaft fracture was present on the right mandible. On histopathology, the masses were confirmed as locally invasive and destructive squamous cell carcinoma. No evidence of metastasis was noted in regional lymph nodes or in any of the distant sites evaluated.
An 18-year-old, neutered, male Vietnamese pot-bellied pig was presented to the Zoological Medicine Service, College of Veterinary Medicine, Kansas State University (Manhattan, KS), from a local zoologic collection for chronic, intermittent nasal discharge and sneezing of 4 months' duration. On physical examination, bilateral nasal discharge was present. The animal was anesthetized, and computed tomography (CT) of the head, thoracic radiographs, graphs, blood collection for complete blood cell count (CBC) and serum biochemical profile, nasal swabs for culture and sensitivity, and a complete dental examination and cleaning were performed.
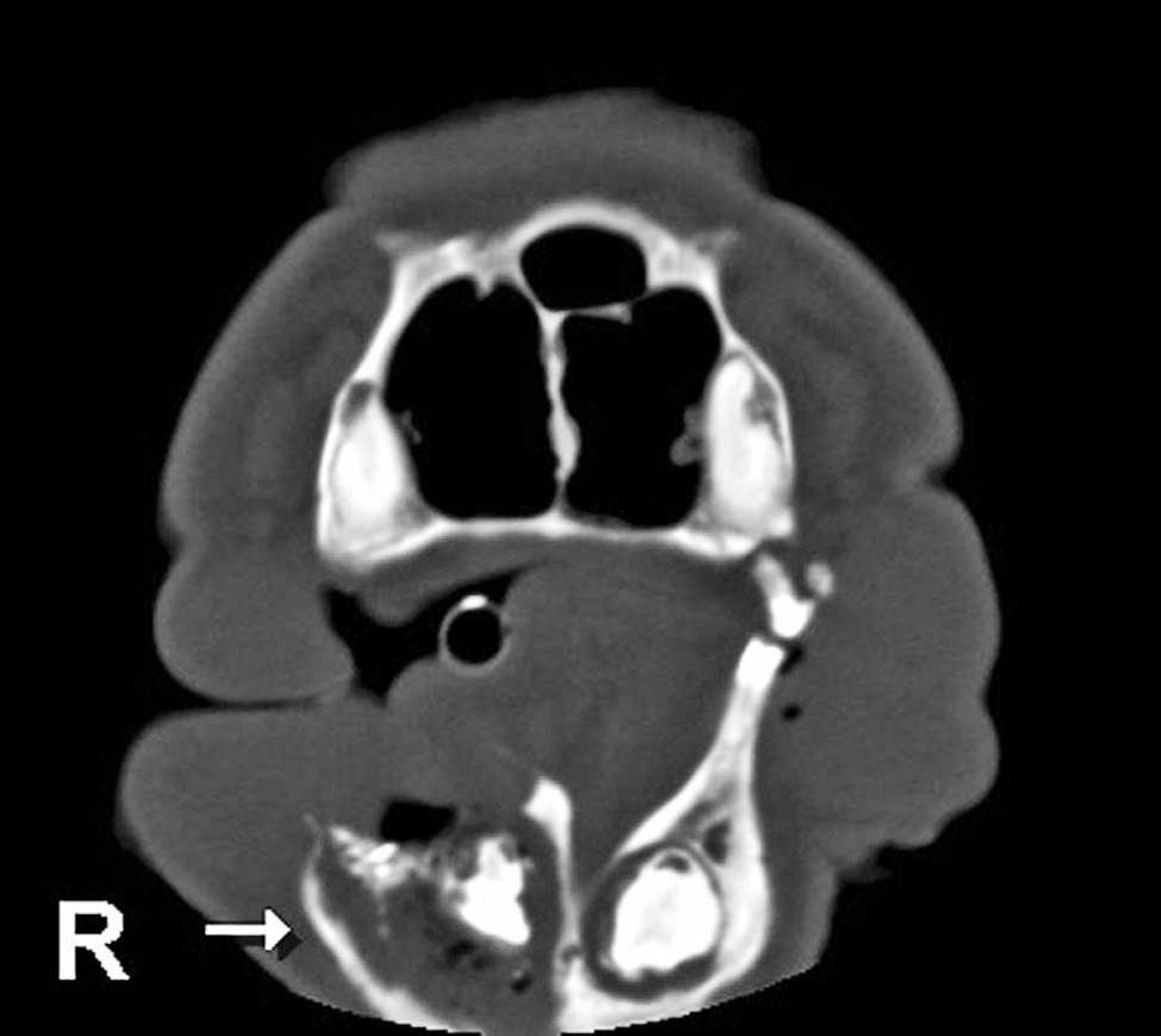
Computed tomography of the head at initial presentation showing osseous destruction with increased soft-tissue density and gas pockets surrounding the right (R) mandibular canine tooth (arrow).
The CT images showed an increased soft-tissue density associated with the tooth root at the caudal nasal passage and sphenoid palatine sinus on the left side. Osteolysis was present at this site. Although the exact maxillary tooth involved was difficult to discern, the location of the osseous destruction suggested a tooth root abscess of the left maxillary molar. The increased CT density continued into the caudal nasal passage ventrally on the left side, conforming to the wall of the nasal passage. An expansile lesion involving the rostral right mandible around the root of the canine tooth along with increased soft-tissue density and gas pockets were present, indicating a tooth root abscess of the right mandibular canine (Fig. 1). Likely potential causes for the osteolysis seen on CT included osteomyelitis and lysis caused by neoplastic disease. Based on CT, these 2 processes could not be distinguished from one another. However, due to the presence of a likely source for an infection, osteomyelitis secondary to the abscessed teeth was thought to be the most likely cause for the osteolysis.
Thoracic radiographs were unremarkable. Laboratory test results were compared with reference values from the International Species Information System. a The CBC revealed a mild leukocytosis (15.2 × 103 cells/μl; reference interval: 6.1–14.3 × 103 cells/μl) and a moderate normo-cytic, normochromic anemia (packed cell volume of 25%; reference interval: 34–49%). During dental examination, the animal was diagnosed with grade IV periodontal disease. An oronasal fistula was present at a right maxillary tooth (108), necessitating extraction of multiple maxillary teeth (107–109). The resulting wound was closed using a gingival flap. Although suppuration was present at the location of the right mandibular canine tooth, this tooth was not extracted due to the possibility of fracturing the weakened mandibular bone. The animal recovered from dentistry without complications and was treated with amoxicillin b while the laboratory results were pending.
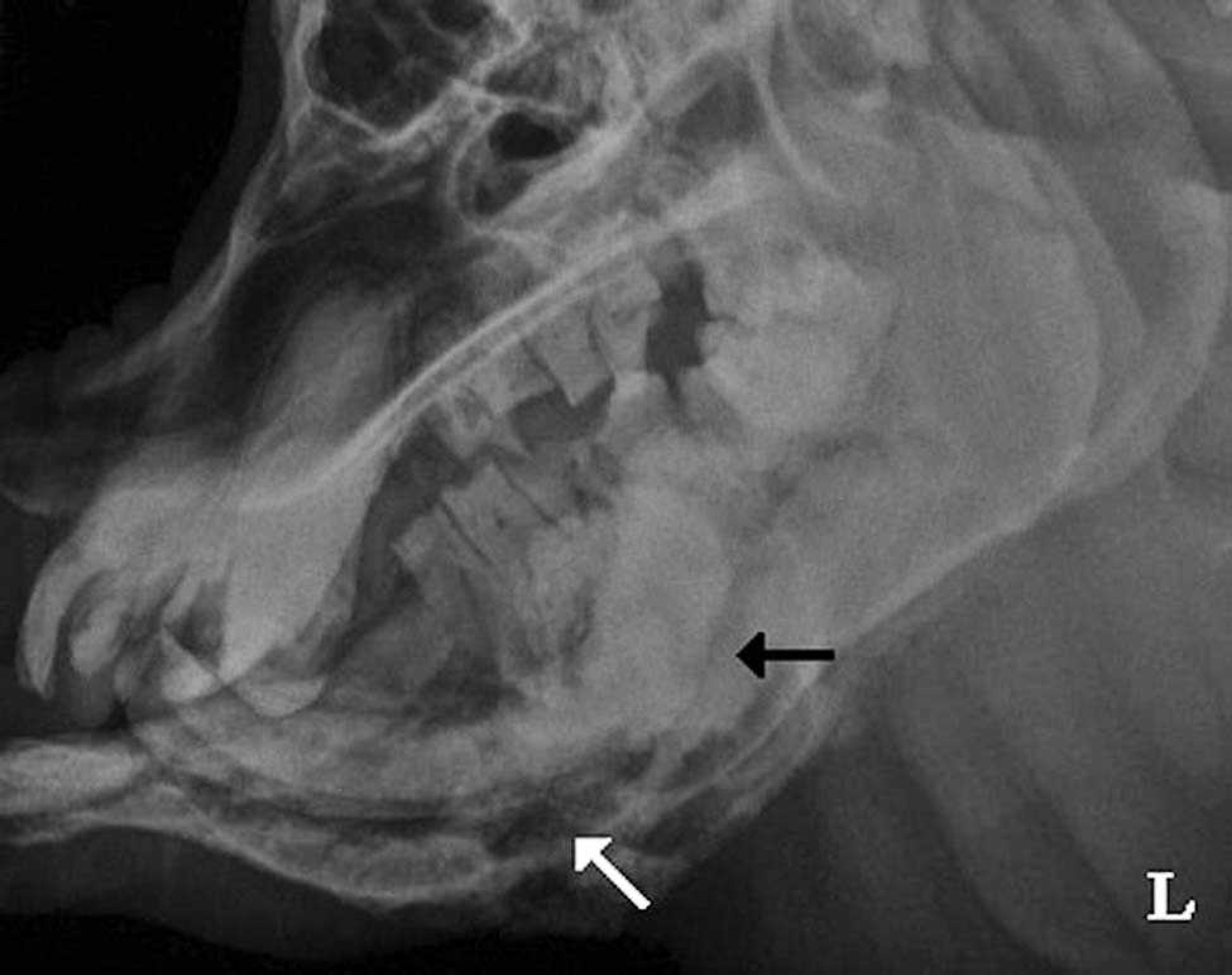
Left (L) lateral radiograph of the skull 18 months after initial presentation showing the moth eaten osteolysis, indistinct bony cortex, and periosteal bone proliferation (black arrow) associated with the rostral to midaspect of the right hemimandible and thinning of the ventral cortex of the left hemimandible with cystic lucency throughout (white arrow).
Aerobic and anaerobic cultures taken from nasal swabs and oronasal fistula yielded Pasteurella sp., Pseudomonas sp., and Peptococcus asaccharolyticus from the nasal swabs. All of these bacteria were sensitive to amoxicillin. Peptostreptococcus prevotii and an alpha Streptococcussp. were isolated from the oronasal fistula swab and were also susceptible to amoxicillin.
Eighteen months after the tooth extractions were performed, the pig was presented again for continued nasal discharge, increased lethargy, difficulty prehending food, and a submandibular mass that had been noted by the keepers. On physical examination, the animal was depressed and very thin, with a body condition score of 1.5 out of 5. The submandibular mass was firm and unassociated with the skin. The mass was approximately 12cm × 12 cm in size and consisted of multiple smaller nodules within the larger mass on palpation. Due to mandibular bone bordering the mass, the third dimension of size could not be determined.
A fine-needle aspirate of the submandibular mass was obtained under manual restraint. The aspirate had high-nucleated cellularity, abundant erythrocytes, and many platelet clumps. Nucleated cells consisted of many well-differentiated squamous epithelial cells, some of which had condensed, pyknotic, or fragmented nuclei and abundant light blue cytoplasm. Occasional clusters of epithelial cells with moderate anisocytosis, moderate anisokaryosis, and high nuclear to cytoplasmic ratio were seen. Nuclei were oval, measured 12–20 μm in diameter, had granular chromatin, and contained 1–2 prominent nucleoli (up to 9 μm). The cytoplasm was light grey to dark blue and occasionally contained punctuate vacuoles. Leukocytes appeared increased in proportion to the amount of blood present. No microorganisms were observed. Although histopathologic examination of the mass was recommended to confirm the morphologic diagnosis, the cytologic examination of the aspirate was consistent with squamous cell carcinoma. Piroxicam c (0.25 mg/kg, orally) was administered daily due to its anti-inflammatory, analgesic, and potential antineoplastic effects.
Two days after the fine-needle aspirate, the pig was anesthetized for a surgical biopsy of the submandibular mass. Blood was obtained for a CBC and serum biochemical profile. The laboratory test results were compared with reference intervals developed by the International Species Information System, a as had been performed previously. The CBC indicated a leukocytosis (24.3 × 103 cells/μl) and a normocytic, normochromic anemia (packed cell volume of 17%). The serum biochemical profile revealed moderate to severe hypoalbuminemia (2.1 g/dl; reference range: 3.7–4.7 g/dl) with hyperglobulin-emia (5.9 g/dl; reference interval: 2.3–4.3 g/dl). Urinalysis and fecal examination were within normal limits. The pig recovered from the diagnostic procedures and anesthesia without complications. The animal was administered amoxicillin b while culture results were pending.
On histopathologic examination, the biopsy tissue contained small groups of anaplastic stratified squamous epithelial cells that did not have a basal layer adjacent to the deep connective tissue. The cells were disorganized, and there was premature keratinization. In one area, the cells were beginning to form keratin pearl-like structures. Nuclei were large and hyperchromatic. The diagnosis of squamous cell carcinoma by fine-needle aspirate was confirmed histologically.
Five days after the surgical biopsy, the animal was manually restrained while radiographs of the skull were performed to assess the degree of mandibular involvement with the neoplasm (Fig. 2). At the rostral to mid aspect of the right hemimandible, bone lysis and an indistinct cortex were observed. At the midbody of the mandible on the ventral aspect, columnar periosteal new bone production was visualized. In addition, thinning of the ventral cortex of the left hemimandible extended from the midshaft to the rostral end of the bone. Cystic lucency was noted throughout the left hemimandible, and soft-tissue swelling was noted ventral to the mandible and larynx.
The culture and sensitivity results yielded Streptococcus pneumoniae, beta-hemolytic Streptococcus sp., Pseudomo-nas aeruginosa, Citrobacter sp., and Fusobacterium necro-phorum.The Citrobacter sp. and P. aeruginosa were resistant to amoxicillin, but all bacteria were susceptible to imipenem. Based on these results, the antibiotics were changed to a combination of imipenem-cilastatin sodium, d marbofloxacin, e and long-acting ceftiofur. f The diet was modified to soften the food to reduce potential pain that might have been associated with eating. Although significant improvement was not seen with any of the aforementioned treatments, the animal was still considered to have an appropriate quality of life.

Gross appearance of the disarticulated mandibu-lar portion of the oral cavity showing the ends of an open fracture of the necrotic right hemimandible (black arrow) and part of the oral squamous cell carcinoma (white arrow).
Approximately 5 months later, the animal was presented again for severe weight loss and inappetence. On physical examination, the pig had lost 11.4 kg over the 5-month period (from 40 kg to 29 kg). The pig was reluctant to stand and was having increasing difficulty prehending food. An increased number of nodular areas were palpable within the submandibular mass. The decision was made to euthanize the animal due to its decline in quality of life.
On complete postmortem examination, there were multifo-cal to coalescing, irregular, nodular masses up to 5 cm in diameter located lateral to the right mandible within the oral cavity. These masses were tan to brown and completely obliterated and replaced the normal anatomy of the caudal two-thirds of the mandible. The surfaces of the masses were ulcerated, and the majority contained necrotic centers. Gingival recession was severe with almost complete exposure of the tooth roots of both the right mandibular premolars and molars. A complete open fracture, which was associated with several of the oral masses, was located in the midshaft of the right hemimandible (Fig. 2). In the area of the mandible surrounding the fracture, major bone loss was present, and the exposed portion of the bone was necrotic. The premolars and molars associated with the right mandibular fracture and surrounding necrotic bone were absent or tenuously attached. Externally, the ventral mandible was distended by multiple palpable masses. The masses extended from the ramus of the right mandible rostrally to the mandibular canine teeth and ventrally within the intermandibular space.
Multiple abscesses were within the nodular masses, as well as along the fourth left maxillary premolar, along the root of the left mandibular canine tooth, and within the left submandibular lymph node. A slab fracture of the fourth left maxillary premolar was also present. There were no premolars or molars caudal to the first premolar on the right maxillary arcade, and severe periodontal disease involved all the remaining teeth on the right and left arcades.
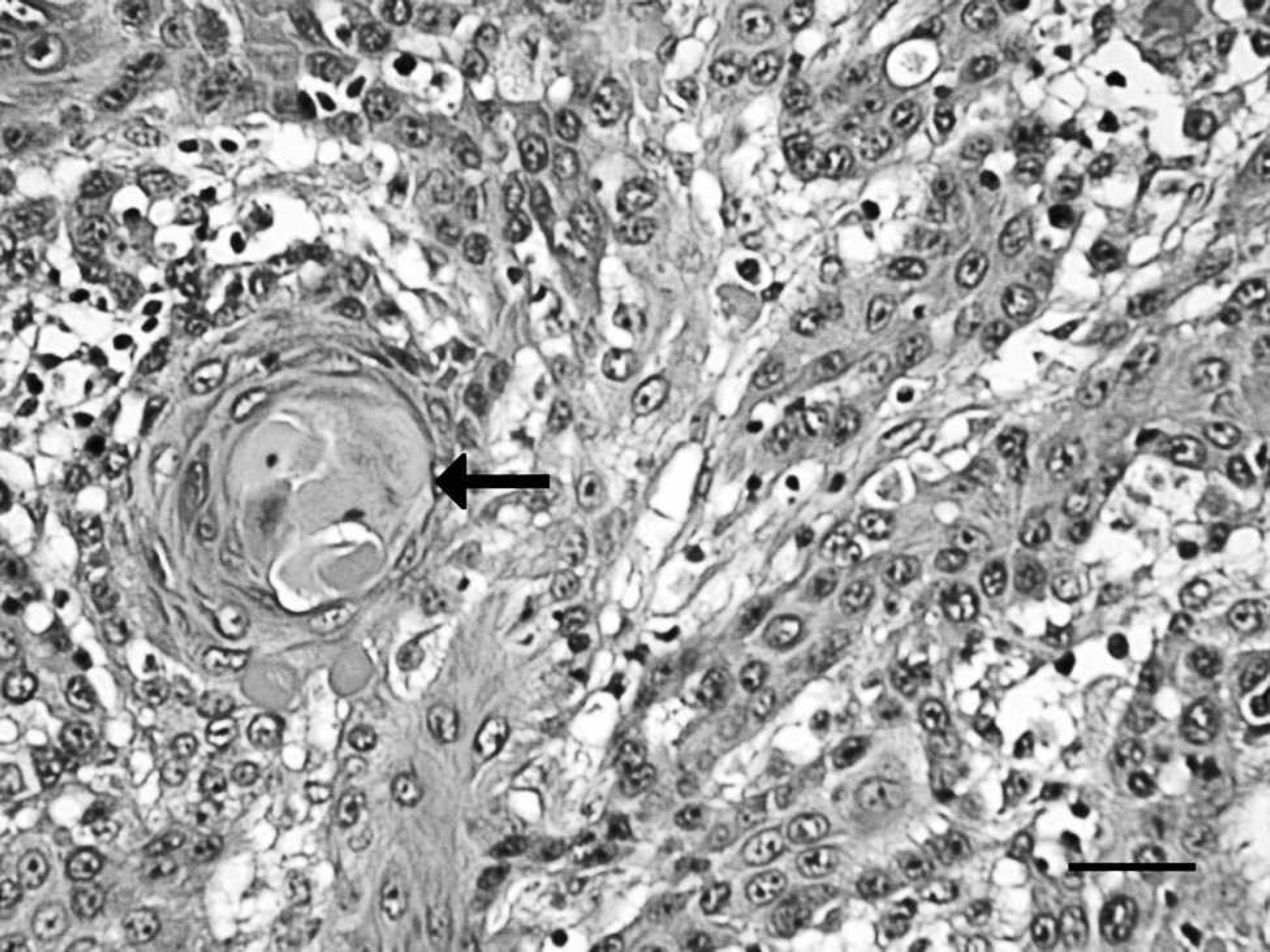
Photomicrograph of the oral squamous cell carcinoma showing prominent keratin pearl formation (arrow) and the moderate anisocytosis and anisokaryosis of the neoplastic squamous cells. Hematoxylin and eosin stain. Bar = 30 μm.
Other necropsy findings included several white, slightly raised plaques up to 1 cm in length on the intima of the aorta within 2 cm of the aortic valve. The stomach and small intestine were devoid of contents. The spiral colon and the rectum contained moderate amounts of formed feces. Multiple cysts measuring up to 5 mm in diameter were present in the cortex and medulla of the kidneys. The trachea, lungs, heart, liver, pancreas, esophagus, gastrointestinal tract, adrenal glands, and urinary bladder were grossly normal.
Histopathology of the oral and mandibular masses showed poorly demarcated, densely cellular, infiltrative neoplastic tissue composed of anastomosing trabeculae, nests, and islands of neoplastic squamous epithelial cells (Fig. 3). The neoplastic cells had distinct cell borders, moderate to large amounts of lightly eosinophilic cytoplasm with occasional individual cell keratinization, and oval nuclei with stippled to vesicular chromatin and 1–2 prominent nucleoli. Moderate anisocytosis and anisokaryosis were present. Mitotic figures averaged 3 per 400× field of view. Multifocal, concentric lamellae of aggregated keratin (keratin pearls) were present (Fig. 4). Areas of necrosis were characterized by abundant eosinophilic cellular debris, and the overlying epithelium was often ulcerated. These characteristics were consistent with squamous cell carcinoma and subsequent necrosis. Microscopically, squamous cell carcinoma was not observed in the submandibular lymph nodes or in any other tissues and organs examined.
Multifocal glomerular tufts were segmentally expanded and replaced by large amounts of eosinophilic, homogenous to waxy material, which stained orange with Congo red dye and demonstrated green birefringent under polarized light, indicating amyloid deposition. Deposits of amyloid also were observed in the cortical and medullary interstitium. In addition, minor amyloid deposits were present in the adrenal glands, spleen, and liver, indicating systemic amyloidosis.
Mild to multifocal atherosclerosis was noted in the aorta. The tunica intima was markedly thickened by clusters of poorly stained cholesterol clefts. Mandibular lymph nodes, skeletal muscle, heart, lung, cerebrum, cerebellum, small intestine, large intestine, and stomach were histologically normal.
Neoplasms that have previously been reported in Vietnamese pot-bellied pigs include endometrial adenocar-cinoma, uterine leiomyoma, squamous cell carcinoma, and hepatocellular carcinoma. 2,6–8 Neoplastic diseases are infrequently reported in domestic swine because of the short life span of most production animals. As the number of domestic swine kept as pets as well as the number of exotic swine that are maintained in zoo collections increases, the population of geriatric swine will also increase. Due to the increased incidence of neoplastic diseases in any geriatric population when compared with juvenile populations, this will likely lead to an increase in the diagnosis of neoplastic disease in swine species kept as pets or maintained in zoologic collections.
Squamous cell carcinoma is a common neoplastic disease in domestic animals. 3,4,6 It is considered to be a malignant neoplasm, because the neoplasms are locally invasive and have excessive and rapid growth. 4,6 Although squamous cell carcinoma does not commonly metastasize to distant sites, it frequently metastasizes to regional lymph nodes. 4,6 Due to the lack of reported cases of oral squamous cell carcinoma in swine, it is difficult to predict whether metastasis to distant sites or to regional lymph nodes will be a common finding. It is likely that the squamous cell carcinoma in swine will behave in a similar manner to that in other animals, regardless of the species affected. In a previous case of oral squamous cell carcinoma in a potbellied pig, metastases were reported in the regional lymphatics. Therefore, evaluation for regional metastasis is indicated in cases of oral squamous cell carcinoma in swine, as is done in other domestic species.
Despite the long-standing clinical history and extensiveness of the oral lesions, no evidence of metastasis to regional lymph nodes or to any other distant sites was present in the current case. However, the neoplasm was still highly aggressive and invasive, as is frequently seen with squamous cell carcinoma in other domestic species. The invasiveness in the present case was significant enough to lead to the long-standing open fracture with necrotic bone of the right mandible, as well as complete obliteration of the normal anatomy associated with the caudal two-thirds of the right mandible.
Due to the aggressive and destructive nature of squamous cell carcinoma, ulceration of the tumor surfaces and secondary bacterial infections are common. 4,6 A necropsy finding in the current case associated with the invasive nature of squamous cell carcinoma was the destruction of adjacent tissues, including surrounding periodontal structures. The weakening of these structures likely contributed to secondary bacterial infection and abscessation of multiple tooth roots, as well as severe periodontitis of all the remaining teeth. Multiple ulcerations of the tumor surfaces were present in the current case, along with necrotic centers within the oral neoplastic nodules. Abscesses within the tumor, as well as around the tooth roots and submandibular lymph node, are likely a result of secondary bacterial invasion associated with ulceration and weakened immune function at the neoplasticsites. The mandibular fracture, periodontitis, and tooth root abscesses likely played the most significant role in the dysphagia and associated weight loss seen in the present case, although cancer cachexia was likely involved as well.
Although the etiology of squamous cell carcinoma has not been fully described, chronic physical irritation has been postulated as a potential trigger for neoplastic change. 6 In the pot-bellied pig in the present case, the 2-year history of severe periodontal disease may have contributed to the neoplastic transformation of these lesions into squamous cell carcinoma. Dental disease was severe (grade IV) and had already led to the abscess formation at the right mandibular canine tooth and the left maxillary molars at the time of initial diagnosis. Severe osteomyelitis and suppuration from the right mandible were present 18 months before the formation of the palpable masses around the ventral mandible. Although these conditions were treated with dental cleaning, tooth extractions, and antibiotics, the animal continued to have intermittent nasal discharge and difficulty prehending food for 2 years after treatment. This indicates that the oral cavity had active inflammation from the time the periodontal disease was first noted until the diagnosis of squamous cell carcinoma 18 months later.
Accurate diagnosis of squamous cell carcinoma requires histopathologic examination of biopsy material or cytologic evaluation of fine-needle aspirates. Other ancillary tests, such as survey radiographs, CT, ultrasound, and magnetic resonance imaging, may help determine the extent of the disease and assess the size and location of a neoplasm, but they cannot positively distinguish between osteomyelitis and neoplastic disease. Therefore, multiple diagnostic tests are often employed to diagnose the neoplasm and determine the severity of clinical disease.
The prognosis for oral squamous cell carcinoma is generally guarded to poor, regardless of the species of animal affected. Few treatment modalities have been shown to be effective, although early radical surgery has been shown to improve the prognosis in some species. 1,7 Piroxicam was administered in the current case due to its potential antineoplastic effects. This nonsteroidal anti-inflammatory drug has been evaluated for antineoplastic effects in both dogs and humans. Multiple mechanisms have been postulated for the antineoplastic effects of nonsteroidal anti-inflammatory drugs; however, the specific mechanisms of action have not been determined. 1,9 In canine oral squamous cell carcinomas, response rates to piroxicam administration are similar to those reported for other cytotoxic treatments. 9 The antineoplastic effects of piroxicam have not been evaluated in swine; however, it was suspected that the analgesic and anti-inflammatory effects of this drug would be beneficial to the overall quality of life in the pot-bellied pig of the present report. Due to the severity of the disease at the initiation of treatment and the relatively short duration of treatment prior to euthanasia, evaluation of any antineo-plastic effects of piroxicam in the current case would be difficult. However, the keepers did report that the piroxicam appeared to make the pig more comfortable.
In summary, the current case report presents the clinical and pathologic findings associated with a highly invasive oral squamous cell carcinoma in a Vietnamese pot-bellied pig. With increased popularity of miniature pigs as pets over the last decade, it is likely that neoplasms will be increasingly diagnosed and reported as these animals age. Squamous cell carcinoma has an extremely poor prognosis, and treatment options are limited. However, prompt diagnosis with possible radical resection early in the course of disease may offer the best chance for control of the neoplasm and prolongation of an appropriate quality of life for the patient.
Acknowledgements. The authors thank the staff at the Sunset Zoological Park in Manhattan, Kansas, for their care and concern of the patient presented in this case report and Dr. Mark Ruder for his help with collecting the information for this publication.
Footnotes
a.
b.
Amoxi-Drop® (amoxicillin), GlaxoSmithKline, Research Triangle Park, NC.
c.
Feldene® (piroxicam), Pfizer Labs, New York, NY.
d.
Primaxin® I.V. (imipenem-cilastatin sodium), Merck & Co. Inc., Whitehouse Station, NJ.
e.
Zeniquin® (marbofloxacin), Pfizer Animal Health, Exton, PA.
f.
Excede® (ceftiofur), Pfizer Animal Health, New York, NY.
