Abstract
Squamous cell carcinoma (SCC), which is a neoplasm that usually arises from the integument, is reported uncommonly in pet birds. Only a few reports of SCCs in the alimentary tract of birds, including psittacines, have been published, and a detailed description of the pathology is not available in the literature. We present here 12 cases of alimentary SCC in psittacines. The average age of the birds was 22.2 y (range: 15–29 y), and affected species included 4 Amazon parrots (Amazona sp.), 3 cockatiels (Nymphicus hollandicus), 3 macaws (Ara sp.), 1 conure (Eupsittula sp.), and 1 Senegal parrot (Poicephalus sp.). Frequent clinical complaints included regurgitation, dysphagia, dyspnea, lethargy, and/or weight loss. SCC primarily affected the oral cavity in 6 of 12 cases, the crop alone in 2 of 12 cases, the crop and esophagus in 1 of 12 cases, the proventriculus alone in 1 of 12 cases, and the crop, esophagus, and proventriculus in 2 of 12 cases. Histologically, alimentary SCCs were locally invasive and often resulted in mucosal ulceration. Although there were no metastases in any of our cases, poor clinical outcomes were frequent and associated most commonly with complete effacement of the alimentary segment and severe inflammation with opportunistic bacterial infection. Our review of the literature records commonly affected species, variability of gross presentations and clinical signs, plausible etiologies, and current diagnostic developments.
Squamous cell carcinomas (SCCs) are reported uncommonly in pet birds, with only a few cases documented in a variety of avian taxa, including psittacines.1,3,5,8,9,15,25,37 The majority of documented cases of SCCs in psittacines occurred in parrots (Amazona sp.), cockatiels (Nymphicus sp.), and budgerigars (Melopsittacus sp.), followed by conures (subfamily Arinae), cockatoos (Cacatua sp.), African grey and eclectus parrots (Psittacus erithacus and Eclectus roratus, respectively), and macaws (Ara sp.).1–4,6,7,9,12–16 SCCs in birds can occur in any location, typically in rostral mucocutaneous junctions, uropygial gland, choana, distal wing (phalanges and patagium) and, less commonly, the alimentary tract.3,9,10,19,26 Compared to their integumentary counterparts, alimentary SCCs are poorly studied in terms of specific location, gross and histopathologic features. We present here a retrospective study of 12 alimentary SCCs in psittacines and a review of the literature.
Materials and methods
We searched the database of the California Animal Health and Food Safety Laboratory System (CAHFS) to identify laboratory or field autopsy and biopsy cases of psittacines with a diagnosis of SCC, from January 1, 1990, through June 24, 2020. Inclusion criteria for our study were 1) psittacine carcasses or tissues submitted for gross and histologic examination, and 2) a diagnosis of SCC in any part of the alimentary system. The final report for each case was reviewed, and the signalment, clinical history, manner of death, and gross and histologic findings were recorded.
For histopathology, hematoxylin and eosin (H&E)-stained slides were retrieved from the archives, or recuts were obtained from 10% formalin-fixed, paraffin-embedded tissues. Although tissues sampled varied on a case-by-case basis, laboratory and field autopsies generally included sections of the neoplasm, brain, peripheral nerves, heart, trachea, larynx, air sacs, thyroid glands, lung, liver, esophagus, crop, proventriculus, ventriculus, pancreas, intestines, spleen, kidneys, gonads, and skeletal muscle. Biopsies included the neoplasm and associated tissues. All tissues were fixed in 10% neutral-buffered formalin for 24–72 h and processed routinely to produce 4 μm-thick H&E-stained sections. For carcasses with oral SCCs, samples of the head were demineralized by immersion in a commercial fixative/demineralizing solution for several days, before routine histologic processing. All slides that included the SCCs were re-examined by one of the authors (J. Asin) to provide a uniform descriptive and systematic analysis of the histologic findings. On each slide, mitoses were counted in 10 contiguous high-power fields (HPF) to obtain a mitotic count (MC) in 2.37 mm2. In addition, neoplastic cell arrangement, keratin pearls, fibroplasia or fibrosis (interpreted as desmoplastic response or as granulation tissue), and necrosis and/or ulceration were evaluated.
Additional testing was carried out following CAHFS standard operating procedures and varied depending on the clinical history, client requests, gross findings, and/or the pathologist’s criteria. These included: aerobic, anaerobic, and fungal culture of liver, lung, kidney, cloaca, and neoplastic tissue (nasal cavity, crop, esophagus, proventriculus); Salmonella spp. culture of liver, lung, intestines, and/or neoplastic tissue; avian influenza A virus and virulent Newcastle disease virus (Avian orthoavulavirus 1) PCR on pharyngeal and cloacal swabs; periodic acid–Schiff (PAS) and Gram stains of selected sections; immunohistochemistry for avian bornavirus (genus Orthobornavirus) on proventriculus and brain; Chlamydia sp. fluorescent antibody test (FAT) on liver, air sac, and spleen smears; heavy metal screen (including arsenic, cadmium, copper, iron, lead, manganese, mercury, molybdenum, zinc) and selenium concentrations in liver and/or kidney; vitamin A and E concentrations in liver; and fecal flotation parasitologic tests.
Results
We found 12 cases of psittacines with SCC in different areas of the alimentary tract in the CAHFS database. Specifically, 8 were autopsied in CAHFS laboratories, 2 in the field, and 2 cases were biopsy specimens (Table 1).
Demographics, taxonomic distribution, clinical history, and type of death of psittacines diagnosed with squamous cell carcinoma in different locations of the alimentary tract.
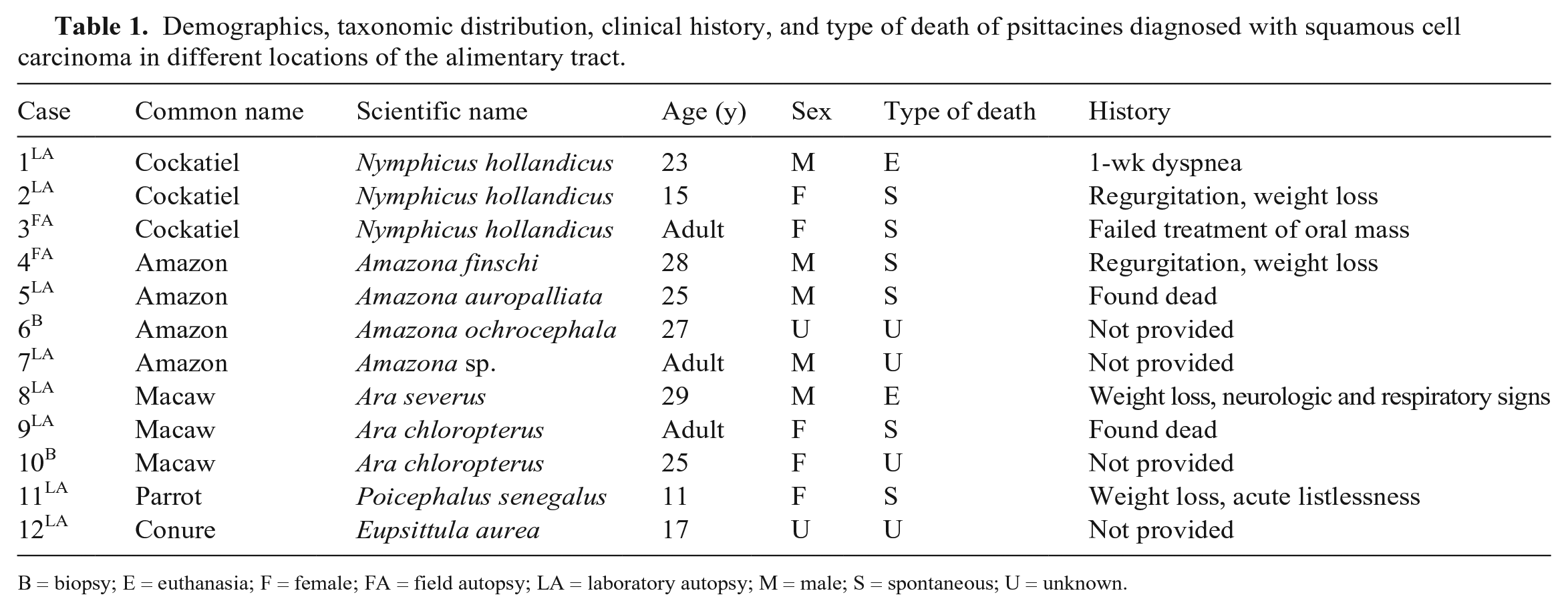
B = biopsy; E = euthanasia; F = female; FA = field autopsy; LA = laboratory autopsy; M = male; S = spontaneous; U = unknown.
The oral cavity was the most frequently affected segment (6 of 12 cases), and for those with available gross descriptions, neoplasms in that area were: 1) light-yellow, multilobulated, cauliflower-like masses arising from the hard palate (case 9), 2) prominent mats mimicking caseous exudate (case 12), or 3) yellow lobular material replacing and expanding the glossal mucosa and extending to the pharyngeal region (case 1; Fig. 1). No additional gross descriptions, except that they were located in the oral cavity and choana, were available for biopsy cases 6 and 10, or for field autopsy case 3.
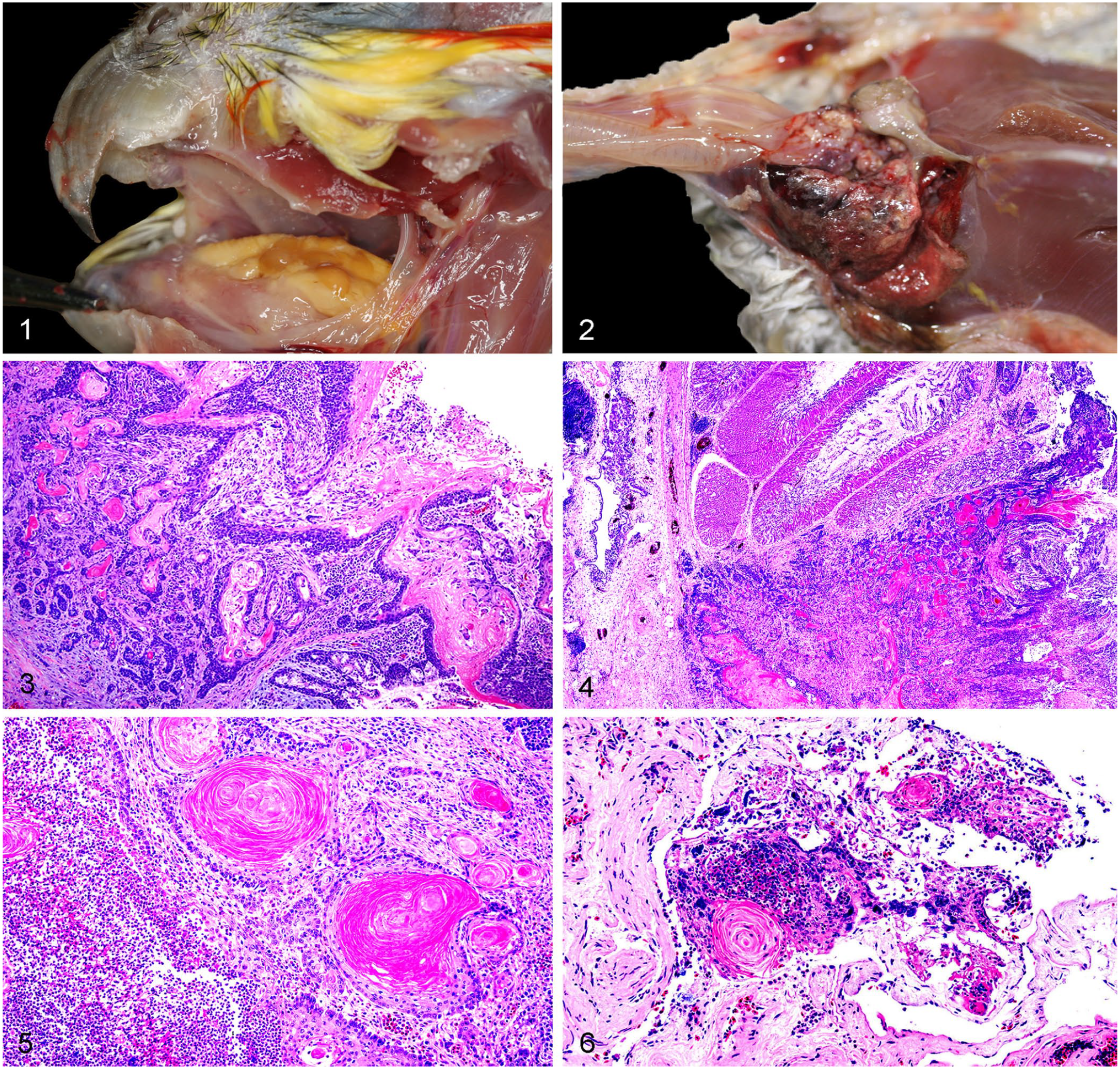
Alimentary squamous cell carcinoma (SCC) in psittacines.
The crop was the second most frequently affected segment (5 of 12 cases), either alone (cases 2, 11) or also affecting the esophagus (case 5), or both the esophagus and the proventriculus (cases 7, 8). SCCs affecting the crop and associated organs were described as a hemorrhagic, tan, and friable mass filling the crop and esophageal outflow tract with associated paintbrush mucosal hemorrhages of the distal esophagus (case 2), as a yellow-tan, protuberant, distal esophageal intraluminal mass (case 5), as a gray, distal esophageal mucosal thickening (cases 7, 11), or as an inconspicuous thickening of the junction of the esophagus and crop reaching the proventriculus, overlain by a granular or roughened mucosal surface resulting in deviation of the crop to the right and marked diffuse compression atrophy of the right lung (case 8). In one of the cases located in the crop (case 2; Fig. 2), the tumor could be palpated through the skin, where it also produced dark-red to purple discoloration. In a field autopsy case (case 4), the esophagus was grossly dark-blue but histologically normal, and intramural SCC was found in the proventriculus. All carcasses were described as thin or emaciated, with moderate to markedly atrophied pectoral musculature and scant fat deposits.
Histologically, SCCs were characterized by neoplastic cells arranged in anastomosing cords and nests (Fig. 3), frequently reaching deep into the submucosa (Fig. 4, Table 2). Although most neoplasms had keratin pearls (Fig. 5), lack of keratin pearls was noted in 2 cases (cases 9, 11). MC was variable and ranged from 3 mitoses/2.37 mm2 in case 9, to 46 mitoses/2.37 mm2 in case 8. In most cases, there was ulceration overlain by a mat of fibrin trapping necrotic debris, erythrocytes, and inflammatory cells. In case 8, large amounts of fibrosis surrounding smooth muscle bundles and a single keratin pearl were observed at the periphery of the atrophic lung (Fig. 6). Although no metastases were observed in any case, neoplastic cells were observed within the vasculature of case 11. Additionally, numerous, up to 5-µm diameter, thin-walled blastospores and blastoconidia arranged in short chains (pseudohyphae) and individual yeast forms were observed in case 8 by PAS stain, and were interpreted as Candida sp. as documented previously. 26 Despite the known association of proventricular adenocarcinomas with Macrorhabdus sp. in budgerigars, 29 no yeast forms compatible with this organism were identified in any case. Mixed bacteria, predominantly represented by coccobacilli and cocci, were observed in all cases, and were admixed with yeast in cases 4 and 10.
Major histopathologic findings of alimentary squamous cell carcinomas (SCCs) in psittacines submitted for autopsy or biopsy examination.
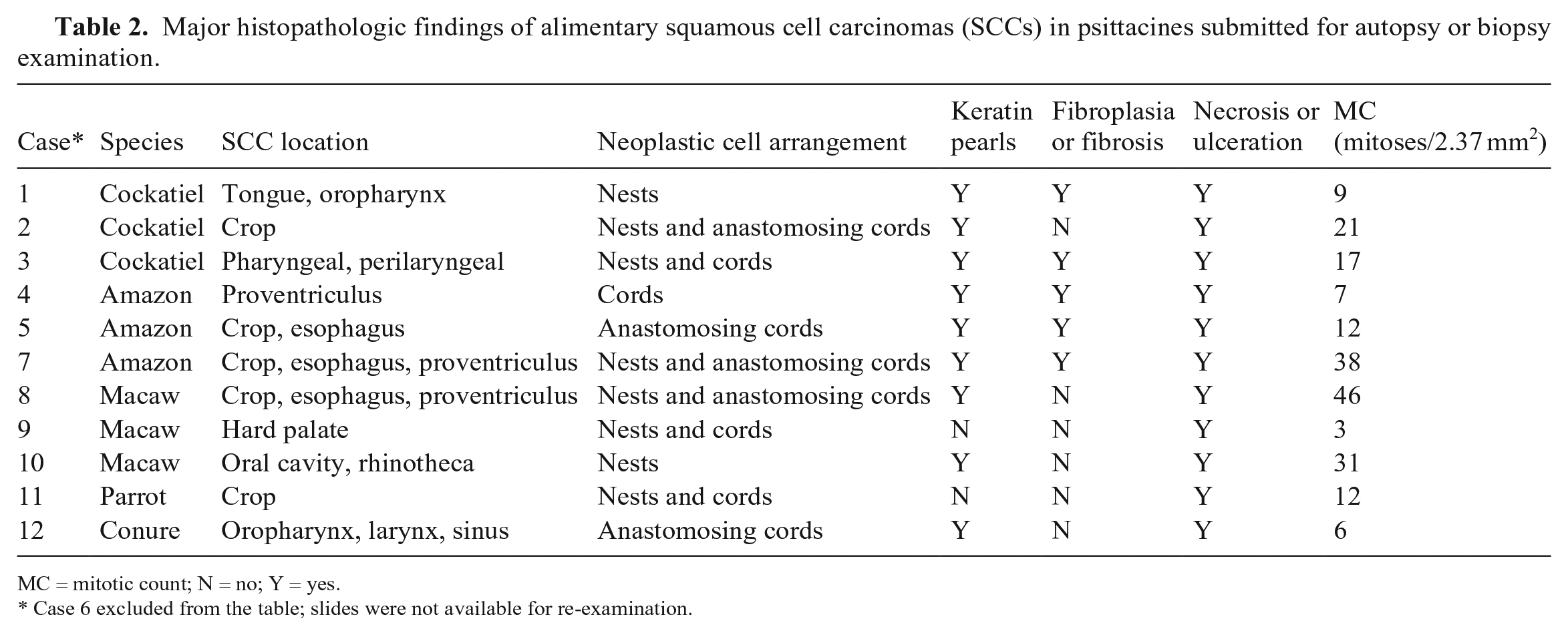
MC = mitotic count; N = no; Y = yes.
Case 6 excluded from the table; slides were not available for re-examination.
Common opportunistic bacteria isolated from neoplastic tissues included Streptococcus sp. and Staphylococcus sp. (case 2), Escherichia coli (cases 8, 9), or mixed flora (cases 5, 8, 9). No fungi were isolated in the 2 cases in which fungal culture was attempted (cases 5, 9).
The cadmium concentration was above expected in the liver and kidney of case 9 (8 and 43.8 ppm, respectively). Concentrations of the other heavy metals were considered adequate in case 9 and the other 2 tested birds (cases 2, 8). Hepatic concentrations of vitamin A were elevated (673 ppm) in case 12.
Avian influenza A virus and virulent Newcastle disease virus PCR was negative in all cases tested (cases 1, 2, 8, 11, and cases 8, 11, respectively). Avian bornavirus immunohistochemistry was negative on both brain and myenteric plexuses in the only case tested (case 8). No parasites or parasite eggs were found in feces via fecal flotation in cases 8 and 11. Chlamydia sp. FAT was negative in cases 1 and 2.
Discussion
Pet birds, especially psittacines, are overrepresented in the literature of avian neoplastic diseases. This is likely a reflection of their popularity as companion animals, their longevity, and the higher likelihood of patient submission for treatment compared to non-psittacine companion birds (e.g., finches). Avian alimentary SCCs are considered uncommon neoplasms in general; most reports refer to poultry.2,11,40 There are a few, scattered reports of alimentary SCC in psittacines that include an ingluvial SCC in a cockatiel, mandibular SCCs in a green-winged macaw (Ara chloropterus), a black-headed caique (Pionites melanocephalus) and a Buffon’s macaw (Ara ambigua), and esophageal SCCs in 2 Amazon parrots (Amazona aestiva and Amazona farinosa).10,18,19,35 Retrospective reviews of case submissions to veterinary diagnostic laboratories also revealed a low prevalence of these neoplasms, including cases of oral SCC in an unspecified psittacine and in a red-shouldered macaw (Diopsittaca nobilis).6,10,26
In our case series, alimentary SCCs occurred in mature psittacines, with a median age of 25 y (average 22.2 y). This is consistent with published reports, in which SCC-affected psittacines had a median age of 22.2 y (average 21.5 y).6,18 No sex predilection was noticed in our study nor discussed in the literature.
Regurgitation, dysphagia, or observation of an oral mass were common clinical complaints in our study as well as in the literature. 42 In alimentary SCC cases, especially in those affecting the crop, dysphagia can be attributed to either reduction of the ingluvial and/or esophageal lumina because of the physical presence of a mass, or it might have a neurogenic cause if atony of the upper digestive tract occurs as a result of invasion of parasympathetic nerve branches. 18 Dilation of the crop might also prompt consideration of proventricular dilation disease, which is caused by avian bornavirus infection. 12
In our study, half of the cases arose from multiple sites in the oral cavity, including the choana. Oral SCCs (not including choana) were diagnosed in 25% of cases in a large-scale clinical survey 42 ; however, SCCs in other locations within the alimentary tract were not included in that study. The species distribution in the cited large-scale clinical survey 42 and other studies18,24,35 matches the findings of our case series, with Amazon parrots and cockatiels significantly overrepresented compared to other psittacines.10,42
The crop was the second most commonly affected segment of the alimentary tract. Therefore, it could be hypothesized that an ingluvial SCC might have been the primary focus in the cases with esophageal and/or proventricular involvement. Despite marked local invasiveness, no evidence of metastasis was reported, which is consistent with the documented biological behavior of SCCs in birds.6,9,26,30 In case 8, it was not clear if the single keratin pearl found in the periphery of the atrophied lung was part of a metastatic focus or simply a point of attachment of the overlying neoplasm. Metastatic alimentary SCC was found in only one report in the literature, which involved a raptor (Montagu’s harrier, Circus pygargus), with clusters of neoplastic cells that formed micrometastases in the lungs and caused osteolysis in the right tibia and radius. 31 Metastasis of SCCs is more prevalent in other species, such as humans, horses, and dogs, frequently reaching the lungs, liver, spleen, and less commonly the bones.4,33,39
As reported elsewhere, 42 necrosis was a prominent histologic feature of alimentary SCCs, most likely related to the rapid turnover of neoplastic keratinocytes. This was the most likely cause of concurrent, opportunistic bacterial or fungal infections in up to 22% of affected birds.
As in other species, conventional diagnostic pathology methods such as cytology and histology are utilized for the diagnosis of SCC in psittacines. Additionally, the use of novel molecular markers such as cyclooxygenase-2 (COX-2), a molecule key in tumor growth, or E-cadherin, a cell adhesion molecule, has been investigated given their prognostic and diagnostic value; the latter seems to be particularly useful to differentiate SCC from mucosal or skin papillomas in small samples. 14 A report concluded that there was a significantly greater proportion of positive cytoplasmic labeling of COX-2 and negative membranous labeling of E-cadherin in SCCs of psittacines compared to papillomas. 14
The etiology of SCC in psittacines has not been determined, although there is speculation about the role of excessive UV light exposure, chemicals, genetics, diet, reused litter, and infectious agents.7,25 In some domestic mammalian species, the transition of viral papillomas to SCC is infrequent but well-established.22,23 However, papillomaviruses do not seem to play a role in the development of mucosal papillomas in psittacine birds. 38 Evidence is mounting toward the association of certain herpesviruses (i.e., Psittacid alphaherpesvirus 1) with internal papillomatosis of parrots. 36 However, definitive evidence of the association between viral infection and SCC in psittacines is lacking.
Other predisposing factors for SCC development, such as hypovitaminosis A or chronic wounds, have been discussed, 34 although these are yet to be proven in animals. The bulk of this information is derived from the human literature, which has investigated the association of epithelial disorders with vitamin A deficiency, 17 and the carcinogenic potential of chronic tissue insult.14,16 Pet bird diets are usually based on seeds, which are not appropriate sources of vitamin A, and thus may lead to hypovitaminosis A and subsequent squamous metaplasia of the upper alimentary tract. If not corrected, this might progress to malignant transformation.28,42 Vitamin A was only quantified in case 12, a conure with an oral SCC. Interestingly, squamous metaplasia in glandular tissue of the external ear was detected in this bird (data not shown). Although histologic lesions in this conure were suggestive of hypovitaminosis A, hepatic concentrations (673 ppm) were considered more than adequate when extrapolating normal poultry ranges (60–300 ppm), and according to a study in Australian psittacines in which hepatic vitamin A concentrations were 33–823 ppm. 21 The correlation of hepatic vitamin A levels with true deficiency, and the potential role in SCC development in psittacines, remains to be elucidated.
Above expected hepatic and renal concentrations of cadmium were found in case 9. Cadmium is an environmental pollutant linked to proximal renal tubular epithelial and testicular necrosis in birds and mammals, with ascribed multi-organic carcinogenic effects in humans.13,20,27 No renal or hepatic lesions were observed in case 9; thus, the clinical significance of this finding to the case or its association with SCC development is undetermined. Detecting cadmium in tissues of adult psittacines is not unusual because it tends to accumulate in the body as the animal ages. A specific cadmium source in case 9 was not identified, but possible cadmium sources for captive birds may include cage material or food dishes (cadmium is found in metal plating and alloys), accidental exposure via small cadmium-nickel batteries, and contaminated feed or drinking water (cadmium can accumulate in plants treated with contaminated fertilizers or sewage sludge). 41
Multiple therapeutic approaches have been described for avian SCC; however, favorable treatment regimens have not been well-established for most tumors. 32 Treatments include surgery, cryosurgery, electrosurgery, radiation, and intralesional therapy. Ingluviotomy was attempted in a cockatiel with ingluvial SCC and, although the bird survived the surgery, complete excision was not possible because neoplastic cells reached the tracheal adventitia, adjacent nerves, and vasculature. 18 Despite the radioresistance of SCCs and low resection success, these neoplasms have a low metastatic rate, and clinical outcome and treatment options might be improved by early detection. 9
In general, SCCs had poor long-term prognosis and were identified as the cause of death in over 30% of SCC submissions in psittacines according to a retrospective review performed in a veterinary diagnostic laboratory. 9 In our study, type of death was available for 8 cases; alimentary SCC was identified as the cause of death in 5 cases, in 2 additional cases SCC led to euthanasia, and 1 bird died under anesthesia. Thus, SCC was the most likely contributor to ill-health and death in most of our cases.
There are some limitations to the interpretation of the results in our study, such as the relatively small number of birds included, or the heterogeneity of the ancillary tests requested among the cases. Nevertheless, these limitations do not interfere with one of the main goals of our work, which was to raise further awareness among avian practitioners and pathologists about the varied gross presentations, nonspecific clinical signs, microscopic appearance, species affected, and possible risk factors of SCCs in psittacines. Despite the local aggressive behavior of SCCs, which makes complete resection difficult, an early diagnosis may translate into improved treatment success, hence widening the spectrum of management and therapeutic approaches as well as improving prognostic outcomes.
Footnotes
Acknowledgements
We thank the pathologists who coordinated the cases included in our study: Drs. Barbara Daft, Hailu Kinde, Rocio Crespo, Janet Moore, H. L. Shivaprasad, and Mark Anderson. We also thank Lucy Gomes for her kind assistance with data mining, and Dr. Robert Poppenga for useful explanations about the heavy metals panel.
Declaration of conflicting interests
The authors declare no potential conflicts of interest related to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
