Abstract
We document a case of Mycobacterium kansasii, a rare, zoonotic bacterium, in a white-tailed deer (WTD; Odocoileus virginianus) in East Baton Rouge Parish, Louisiana. Grossly, the deer had fibrinous pleuropneumonia with yellow, mineralized nodules scattered throughout the lungs and extending to the pleura. The kidneys were enlarged and had numerous pale foci in the cortex. Microscopically, the pulmonary architecture was replaced by variably sized, multifocal-to-coalescing granulomas with peripheral histiocytes and fewer multinucleate giant cells, and necrotic centers with mineralization and hemorrhage. The latter rarely contained one to a few acid-fast, slender, 7-µm long bacteria, for which beaded morphology was sometimes evident. Similar acid-fast bacteria were also within histiocytes in the kidney. PCR assay of fresh lung sample and subsequent sequencing revealed a non-tuberculosis mycobacterium, M. kansasii. These lesions were similar to those that result from infection with M. bovis in WTD. Both M. bovis and M. kansasii are zoonotic. WTD are a reservoir of M. bovis, which is a major concern in regions in which WTD and cattle can come into close contact.
Mycobacteria incorporate > 180 species of aerobic microorganisms, including those within the Mycobacterium tuberculosis complex (MTC), which can cause pulmonary, disseminated, or cutaneous disease in humans and animals.4,10 Broad classification of mycobacteria includes > 100 species of non-tuberculosis mycobacteria (NTM) that are ubiquitous in the environment and are found in soil, water, dust, and feed material. 4 Many of these NTM are opportunistic pathogens capable of causing clinical disease indistinguishable from tuberculosis-like disease, necessitating identification through bacterial culture or molecular testing. 13 We describe herein findings from postmortem examination of a free-ranging white-tailed deer (WTD; Odocoileus virginianus) diagnosed with infection by Mycobacterium kansasii, a NTM rarely reported in wildlife and more commonly associated with pulmonary disease in immunocompromised humans. 5
In February 2019, a 2.5-y-old WTD buck was found dead and partially submerged in water within a flooded field in northern East Baton Rouge Parish, Louisiana. The carcass was in poor-to-moderate nutritional condition with a body mass of 54.4 kg. Field autopsy revealed fibrinous pleuropneumonia with numerous, randomly scattered, 0.5 to 6.0-cm diameter, firm, yellow nodules with gritty material throughout the pulmonary parenchyma and extending to the pleura (Fig. 1). The kidneys were subjectively enlarged and had numerous, white, mildly elevated foci in the renal cortex. The abomasal mucosa was reddened. Fresh and formalin-fixed (in 10% neutral-buffered formalin) samples of lung, kidney, liver, heart, and spleen were submitted to the Southeastern Cooperative Wildlife Disease Study (SCWDS) at the University of Georgia (Athens, GA) for diagnostic evaluation. Formalin-fixed tissues were routinely processed and stained with hematoxylin and eosin. Additional histochemical stains performed on lung and kidney included Ziehl–Neelsen (ZN) acid fast, Grocott methenamine silver (GMS), and Lillie–Twort (LT) Gram stains. Histologic processing and staining were performed by standard protocols at the Athens Veterinary Diagnostic Laboratory (AVDL) at the University of Georgia, which is accredited by the American Association of Veterinary Laboratory Diagnosticians.
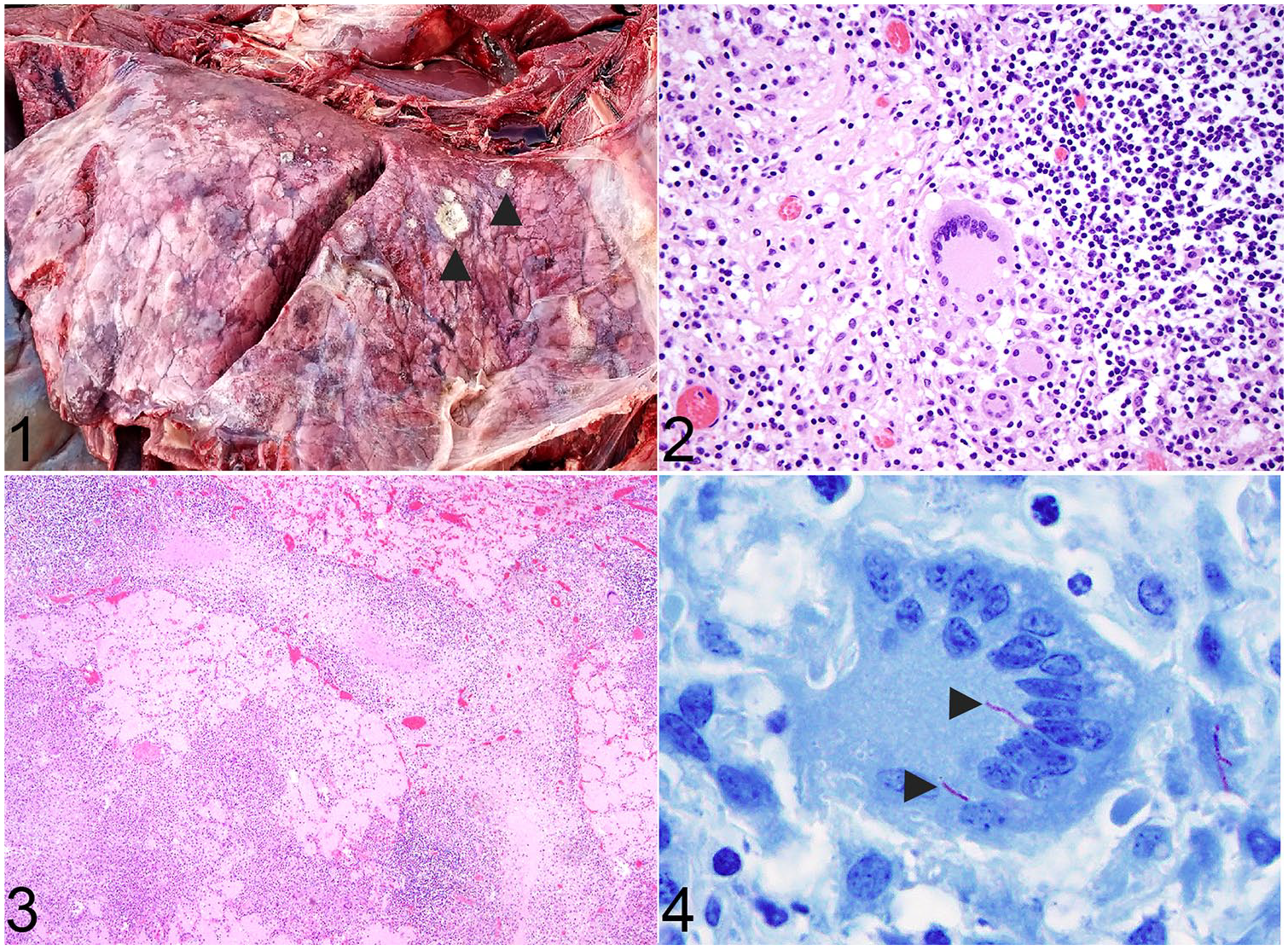
Fibrinous pleuropneumonia caused by Mycobacterium kansasii in a white-tailed deer.
Microscopically, most of the pulmonary architecture was effaced and replaced by variably sized (≤ 5 mm diameter), multifocal-to-coalescing granulomas. Central areas of necrosis within the granulomas often encompassed large, mineralized foci, as well as scattered, mild-to-moderate hemorrhages, surrounded by a rim of macrophages and mature fibrocytes. Rarely, single and small aggregates of elongated, sometimes beaded, ~ 7-µm long bacilli were within the cytoplasm of multinucleate giant cells. The surrounding interstitium was often compressed and obscured by fibrous connective tissue with high numbers of lymphocytes, plasma cells, histiocytes, and occasional Langhans multinucleate giant cells interspersed with fibrin and pyknotic debris (Fig. 2). Adjacent airway lumens and alveoli were filled with edema and variable amounts of pyknotic debris. Lymphocytes and plasma cells occasionally formed nodules within the interstitium. These inflammatory cells also expanded interlobular septa, and less commonly the pleura, along with scattered pyknotic debris, fibrocytes, and edema (Fig. 3). ZN stain confirmed the presence of acid-fast bacteria within the cytoplasm of Langhans multinucleate giant cells (Fig. 4).
Approximately 50% of the renal interstitium was infiltrated by many nodular aggregates of lymphocytes and plasma cells, with fewer histiocytes and Langhans multinucleate giant cells. Inflammatory cell infiltrates were more abundant within the renal cortex compared with the medulla. Small foci of necrosis were occasionally among these inflammatory cells, and acid-fast bacteria similar to those in lung were seen rarely within the cytoplasm of histiocytes. No other bacteria or fungi were evident in the lung or kidney by LT Gram or GMS stains, respectively.
The acid-fast bacteria microscopically observed within lung lesions prompted molecular confirmation of Mycobacterium spp. in fresh lung tissue by PCR assay. The assay targeted the 16S rRNA gene, which is conserved among all Mycobacterium spp., using primers KY18 (5′-CACATGCAAGTCGAACGGAAAGG-3′) and KY75 (5′-GCCCGTATCGCCCGCACGCTCACA-3′). PCR amplifications were carried out in 100-μL reaction mixtures by adding 50 μL of template DNA to 50 μL of a premade amplification master mixture. DNA extraction was performed (QIAamp cador pathogen mini kit; Qiagen, Hilden, Germany) according to the manufacturer’s instructions. Amplification was achieved through denaturing at 95°C for 5 min, followed by annealing for 40 cycles at 95°C for 5 s, 55°C for 5 s, and 68°C for 25 s. Extension was performed at 71°C for 1 min. Sanger sequencing of the PCR product on the SeqStudio sequence was achieved (Applied Biosystems BigDye terminator v3.1 cycle sequencer; Thermo Fisher Scientific, Waltham, MA) and revealed a best match with 100% similarity following comparison search of a 500-bp sequence in BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi) for Mycobacterium kansasii. Aeromonas salmonicida, A. hydrophila, Lactococcus lactis, and Clostridium sporogenes were isolated from the lung following aerobic and anaerobic culture and were considered environmental contaminants, given that the deer carcass was partially submerged in water at the time of recovery. No bacteria were isolated from the kidney. All ancillary tests were performed at the AVDL.
The detection of M. kansasii in a WTD underscores the importance of the detection and identification of Mycobacterium species in wildlife to assess for MTC pathogens, which are transmissible to livestock and have zoonotic potential. The severity of associated pulmonary lesions with evidence of spread to the kidneys in this WTD supports a diagnosis of M. kansasii infection. The NTM M. kansasii cannot be grossly or microscopically distinguished from mycobacteria within the MTC, including the causative agent of bovine tuberculosis, M. bovis.5,14 Further, cross-reactivity among Mycobacterium species, including M. bovis, in antemortem immunologic testing of cattle and cervids has been documented in the United States.15,18
The economic implications of a diagnosis of M. bovis, a federally reportable disease in the United States and Canada (http://www.inspection.gc.ca/animals/terrestrial-animals/diseases/reportable/bovine-tuberculosis/eng/1330205978967/1330206128556) in free-ranging wildlife are grave compared to a diagnosis of M. kansasii infection. The U.S. Department of Agriculture maintains a national eradication program for bovine tuberculosis through regulatory directives and surveillance, including testing of carcasses at federal and state slaughter facilities. Livestock producers with herds containing animals that test positive for M. bovis are required to quarantine the herd and are banned from exporting animal products until further testing and depopulation confirm that the remaining animals are tuberculosis-free. Carcasses with diseased tissue that tests positive for M. bovis are condemned and destroyed (U.S. Code of Federal Regulations, Title 9, https://www.aphis.usda.gov/aphis/ourfocus/animalhealth/animal-disease-information/cattle-disease-information/national-tuberculosis-eradication-program). As of this writing, the incidence of bovine tuberculosis has been significantly reduced in the United States, largely as a result of the National Tuberculosis Eradication Program. However, Michigan continues to harbor bovine tuberculosis because the disease is endemic in WTD populations and continues to sporadically spread to cattle. 12
In WTD naturally infected with M. bovis, the most common lesions are caseous granulomas in the retropharyngeal lymph nodes. Pulmonary and alimentary involvement is also possible, and lesions in the palatine tonsils have been reported.14,19 Transmission can occur through inhalation of aerosols or through ingestion of contaminated soil, water, and feed. Microscopically, M. bovis typically is seen as one or a few granulomas within the affected tissue. Lymphocytes, histiocytes, and multinucleate giant cells surround necrotic foci, similar to the presentation in our case. In Michigan, M. bovis was likely introduced to free-ranging WTD herds from infected cattle in the early-to-mid 20th century. 9 In the early 1900s, cattle in North America were widely infected with M. bovis, prompting the formation of the U.S. surveillance and eradication program. Much progress has been made in decreasing the prevalence of M. bovis cattle in the United States and Canada, but the initial spillover resulted in the establishment of this pathogen in a common, ubiquitous wildlife species, thus hindering M. bovis eradication efforts in cattle. 9
Detection of M. bovis in deer is challenging because herds may harbor M. bovis for years with no apparent antemortem clinical signs and no obvious gross lesions at the time of autopsy or hunter harvest. 9 Increased surveillance and monitoring for M. bovis in susceptible wildlife species such as deer, bison, and elk remain important factors in understanding the epidemiology and distribution of this disease. The potential for cross-reactivity on antemortem M. bovis skin tests makes M. kansasii and other NTM important differentials for livestock producers and state veterinarians.15,18 Based on the lack of previous detection in the face of M. bovis surveillance efforts, M. kansasii is not believed to commonly infect WTD herds in North America.
The NTM M. kansasii identified in this WTD is a slow-growing, beaded Mycobacterium species that is most commonly detected in water sources and in humans as well as in multiple captive and free-ranging wildlife species in the United States.5,17 M. kansasii is a common NTM infection in human immunodeficiency virus–infected humans. The bacterium is less recognized in veterinary medicine and is most often an incidental finding at autopsy or detected in the feces of otherwise healthy animals, including deer. 5 The likely source of infection is the environment, and to date, there is no evidence of animal-to-animal transmission. M. kansasii typically is not considered a threat to domestic animals or wildlife, given that cases causing disease are sporadic with no perceived herd- or population-level effects.
A case of M. kansasii has been reported in a single, hunter-harvested black-tailed deer (O. hemionus columbianus) that had variably sized granulomas adhered to the thoracic wall, lungs, and abdominal viscera. Microscopically, rare acid-fast bacilli were identified, and granulomas contained caseous material surrounded by macrophages, multinucleate giant cells, fibroblasts, and mononuclear cells. 5 Other species in which clinical disease associated with M. kansasii infection has been reported include: domestic (Sus scrofa domesticus) and feral (S. scrofa) swine, cattle (Bos taurus), camels (Camelus dromedarius), a domestic cat (Felis catus), 2 dogs (Canis lupus familiaris), a llama (Lama glama), an alpaca (Vicugna pacos), 2 bonteboks (Damaliscus pygargus dorcas), a goat (Capra hircus), Florida manatees (Trichechus manatus latirostris), rhesus macaques (Macaca mulatta), squirrel monkeys (Saimiri sciureus), ring-tailed coatis (Nasua nasua), and 2 black-bearded sakis (Chiropotes satanas).1–6,8,10,12,13 Immunosuppression with or without identification of a primary cause of disease was often suspected or confirmed in these affected animals. Consistently documented lesions in these cases were granulomas in the pulmonary parenchyma or thoracic wall. In some cases, disseminated granulomas were in various organs (e.g., kidneys, liver, spleen, and lymph nodes), including in our WTD (kidney) and the previously reported black-tailed deer (intestine).
In our case, mycobacteriosis was the primary differential diagnosis, and pulmonary infection was considered severe and potentially fatal in a WTD. Other less likely differentials included bacterial pneumonia with or without an inciting penetrating wound, fungal pneumonia, and parasitic granulomas (e.g., Dictyocaulus viviparus). The latter was ruled out because there was no gross or microscopic evidence of nematodes within the lungs, and infection with D. viviparus rarely causes clinical disease in healthy, adult deer. 11 Several species of fungi, including Aspergillus spp. and Conidiobolus spp., thrive in subtropical climates such as in Louisiana. Aspergillus infection has been identified in WTD but is not commonly reported as a finding or cause of clinical disease. There is also a single report of Conidiobolus incongruus causing disseminated, eosinophilic granulomas in a WTD. 7 Spores or hyphae consistent with fungal infection were not observed microscopically in the examined tissue. Bacteria associated with a foreign body or penetrating wound (e.g., Trueperella pyogenes) were unlikely because there was no evidence of traumatic injury to the thoracic cavity. T. pyogenes is most commonly associated with intracranial abscessation in deer but can cause disseminated disease including pneumonia. 16 The absence of neutrophilic inflammation in examined tissue made infection with other bacteria a less likely differential. The presence of pulmonary granulomas in a cervid strongly suggested mycobacteria as the most likely differential, and molecular testing confirmed the presence of the NTM, M. kansasii.
Molecular testing is crucial for the identification and differentiation of Mycobacterium spp., including distinguishing NTM- from MTC-type bacteria. In free-ranging wildlife, any animal with pulmonary granulomas or fibrinous pleuropneumonia should be tested for Mycobacterium spp., especially if livestock are nearby. Acid-fast bacteria in our case were observed only rarely, which is consistent with previously reported cases.1,5,8 Even in the absence of acid-fast bacteria, Mycobacterium spp. should remain a differential for livestock or deer with pulmonary granulomas. This is demonstrated in a report of disseminated granulomas in 2 black-bearded sakis in a zoologic park with no histologic evidence of acid-fast bacteria; identification of M. kansasii was made solely through PCR. 10 Bacterial culture is another appropriate way to determine species of Mycobacterium; however, in time-sensitive cases, molecular testing is preferred. This is particularly important in cases for which a bacterium within the MTC is a possible differential diagnosis, based on the potential threat to the health of agricultural animals or humans.
Footnotes
Acknowledgements
We thank the Louisiana Department of Wildlife and Fisheries for submission of case material, and all Southeastern Cooperative Wildlife Disease Study (SCWDS) member state wildlife agencies for their support. We also thank the AVDL for assistance with histology and ancillary testing. Logistical support was provided by Jeanenne Brewton and Michelle Willis of SCWDS.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding was provided by the sponsorship of the Southeastern Cooperative Wildlife Disease Study by the fish and wildlife agencies of Alabama, Arkansas, Florida, Georgia, Kentucky, Kansas, Louisiana, Maryland, Mississippi, Missouri, Nebraska, North Carolina, Oklahoma, Pennsylvania, South Carolina, Tennessee, Virginia, and West Virginia, USA. This support was provided in part by the Federal Aid to Wildlife Restoration Act (50 Stat. 917).
