Abstract
An adult female Cape porcupine (
Keywords
Aspergillosis is a mycosis that affects a diverse range of animal hosts.2,4,5,8
A 17-y-old, 13 kg, female Cape porcupine, housed at the Zoo de Granby (Quebec, Canada), was found acutely lethargic, in sternal recumbency, and dyspneic. The animal was housed in a heated building with another Cape porcupine and had access to an outside enclosure. The indoor pen had a concrete floor covered with wood shavings and various enrichment items. The outside enclosure had a soil substrate and was shared with numerous meerkats (
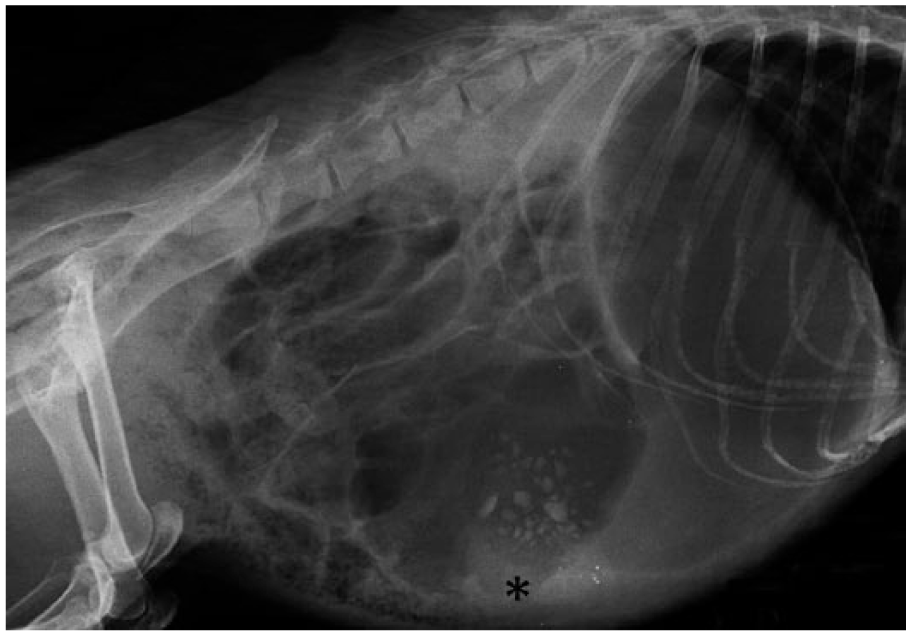
Marked and diffuse dilation of the gastrointestinal tract on a right lateral radiographic view in a Cape porcupine. A heterogeneous mass effect, believed to be caused by ingesta in the stomach, is also present in the cranioventral abdomen (asterisk).
The following day, the porcupine was in lateral recumbency with severe dyspnea and gaseous abdominal distension. The marked distension of the digestive tract and the suspicion of an abnormal abdominal structure motivated the need for an exploratory laparotomy to rule out causes such as intussusception, intestinal torsion, gastrointestinal foreign bodies, and digestive infiltrative disease. Anesthesia was induced as described previously. Following endotracheal intubation, the level of respiratory effort decreased dramatically. Given the suspicion of upper airway obstruction leading to aerophagia, endoscopic examination of the upper respiratory and digestive tracts was attempted prior to laparotomy. Flexible endoscopic examination of the oral cavity was unremarkable. The endoscopic evaluation of the nasal cavities was not possible because the diameter of the nares was too small to permit the entry of a 6.6-mm flexible endoscope or a 4-mm rigid endoscope. Orogastric intubation was performed to reduce gastric dilation prior to the exploratory laparotomy, and drained ~200 mL of green-tinged fluid from the stomach throughout the procedure. Preparation for a laparotomy was difficult because the skin was extremely delicate, and removal of the quills resulted in 2 small skin lacerations that were repaired. The quills were stabilized at their base connection with the skin and extracted vertically to avoid additional tears. A standard approach for a midline laparotomy was performed and revealed gaseous distension of intestines, marked enlargement of mesenteric lymph nodes, and significant thickening and multinodular appearance of ~15 cm of the jejunal wall. Biopsies of the jejunal wall and mesenteric lymph node were performed because there was suspicion of an infiltrative disease process. The heterogeneous mass identified on radiography was not located and was assumed to be artefactual or material within the gastrointestinal tract. Recovery was unusually long, and the porcupine regurgitated upon extubation. The regurgitated material was suctioned out of the oral cavity, but some ingesta were likely aspirated. Medical treatment was continued as initially prescribed; however, the porcupine was found dead the following day and was submitted for autopsy.
The most remarkable gross anatomic finding was marked and diffuse necrosis of the right and left nasoturbinates, maxilloturbinates, ethmoid turbinates, and olfactory nerves. Numerous, green plaques were noted on the mucosa of the left nasal cavity. One large plaque (15 × 7 mm), present in the medial meatus, invaded the wall of the left maxillary sinus (Fig. 2).The tracheal mucosa was diffusely erythematous. One fragment of straw was found in the primary left bronchus. Mandibular and mesenteric lymph nodes were hypertrophic. Tissues were fixed in 10% buffered formalin, embedded in paraffin, sectioned at 3 µm, stained with hematoxylin, phloxine, and saffron (HPS), and examined by light microscopy.
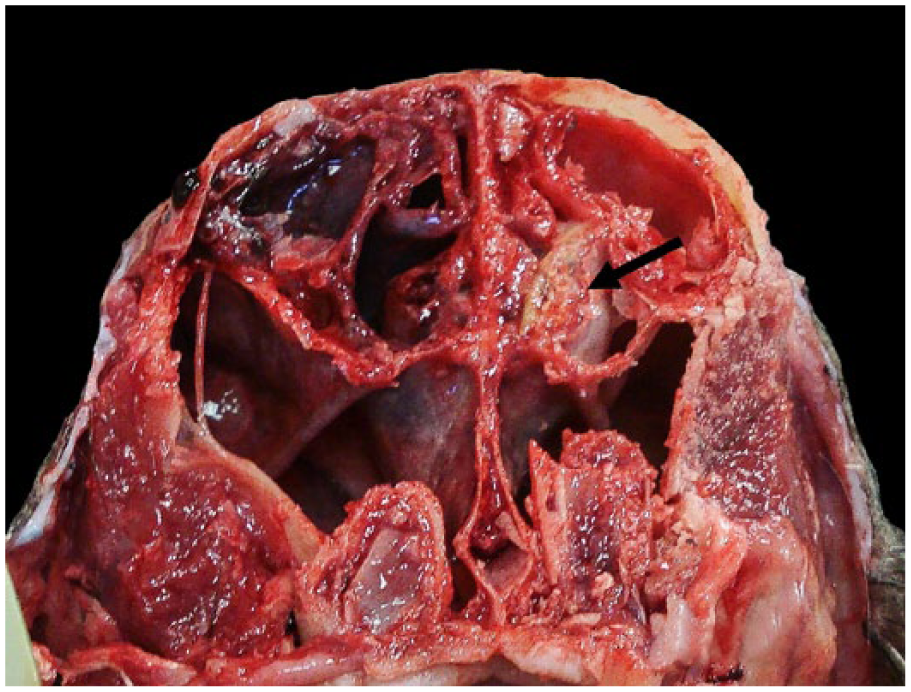
Marked destruction of the nasal turbinates in a Cape porcupine with congestion of the mucosa of the right nasal cavity and a fungal plaque in the left nasal cavity (arrow).
Histologic examination of the nasal turbinates revealed focally extensive ulceration of the mucosa associated with diffuse fibrosis and marked destruction of the turbinate bones. Examination of one of the plaques seen in the nasal cavities at gross examination showed a large aggregate of fibrin containing degenerate epithelial and inflammatory cells. Numerous branching and septate fungal hyphae were invading this fibrinous material (Fig. 3). Numerous conidia, often organized around vesicles, formed conidiophores showing characteristic morphology of
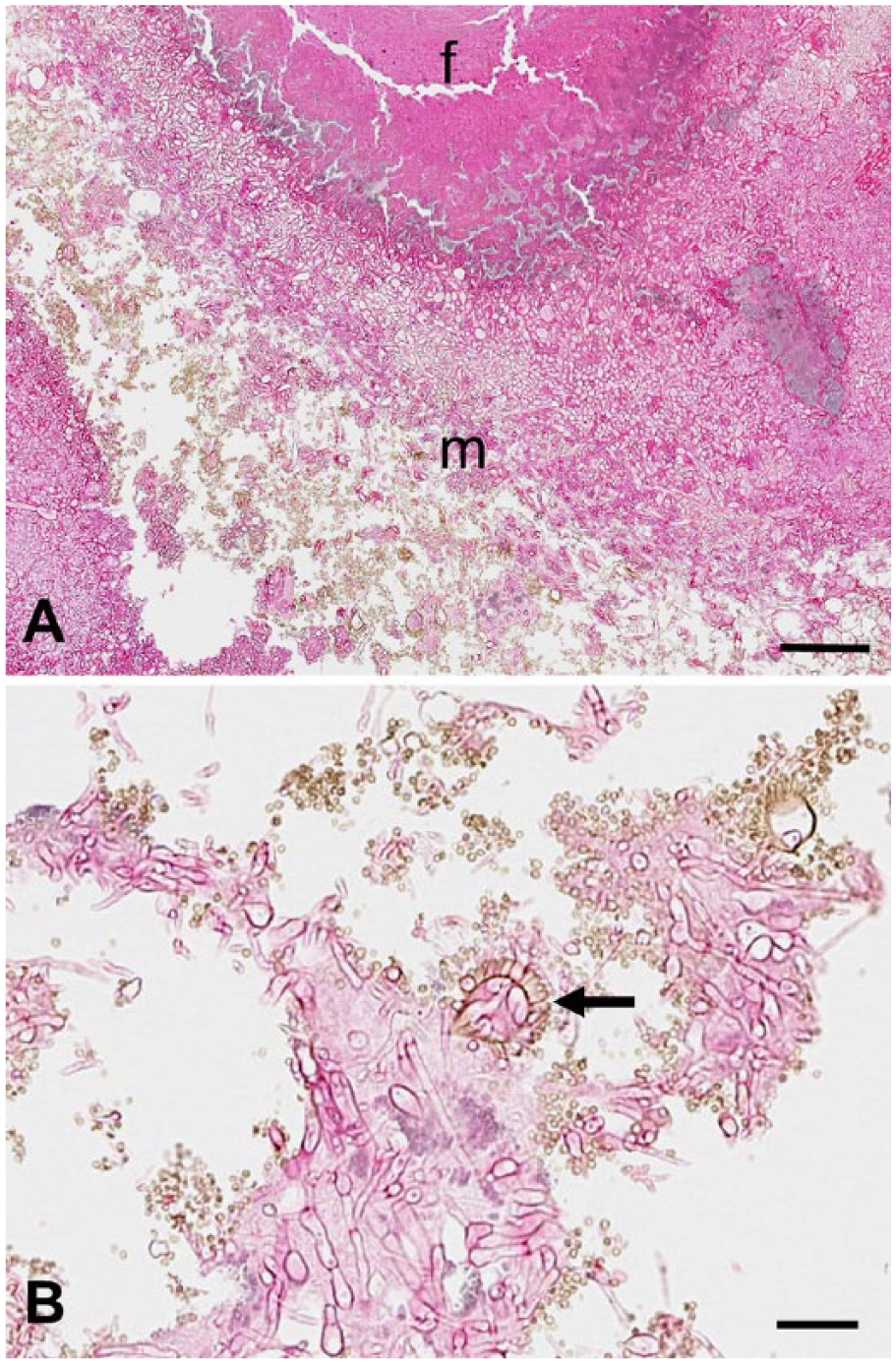
The final diagnosis was chronic ulcerative fibrinous fungal (
Although a few cases of pulmonary or disseminated fungal diseases have been reported in porcupine species,1,3,9 fungal rhinitis has not been reported previously in a large rodent species, to our knowledge. A few case series of environmentally linked fungal rhinitis have been reported in laboratory rats.11,12 Additionally, rabbits and rodents have been frequently used as models of fungal rhinosinusitis for humans.6,15 Mammalian fungal rhinosinusitis is most often caused by
Although infections are commonly noted in immunocompromised patients,5,8
Clinical signs in canine SNA commonly include chronic purulent nasal discharge, nasal pain, nasal planum ulceration or depigmentation, occasional sneezing, epistaxis, decreased appetite, depression, facial deformity, and epiphora.
5
Sneezing and nasal scratching, audible sniffing noises, inspiratory stridor, brown-black debris around the nostrils, and hemorrhages were observed in laboratory rats with
Despite years of research, establishing a diagnosis of canine SNA is still difficult. A combination of imaging (computed tomography [CT] or radiography), rhinoscopy or sinuscopy, biopsy with histopathology, cytology, fungal cultures, and serology is recommended to achieve a definitive diagnosis. 5 In our case, no skull abnormality was detected with conventional radiography, and access to a CT unit was not available. Furthermore, the acute presentation limited additional diagnostic procedures. In addition, postmortem examination confirmed that anterograde rhinoscopy could not be performed in this species of porcupine with the available endoscopic equipment given the narrowness of the nares. Even if the correct diagnosis had been established, appropriate treatment in the porcupine would have been a challenge; prolonged topical treatment via a catheter placed in the affected sinus is the recommended therapy in dogs 5 and would not have been accomplished easily in a porcupine.
Footnotes
Acknowledgements
We thank the technicians of the Animal Health Service and the dedicated keeper staff at the Zoo de Granby for their contributions to this case.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
