Abstract
Routine postmortem examination and histologic evaluation of tissue sections demonstrated hepatic lipidosis (HL) in 2 adult captive porcupines with a history of sudden death. The male porcupine had a markedly enlarged pale liver that microscopically showed large unilocular vacuoles within hepatocellular cytoplasm. The periparturient female had similar but less marked hepatic lesions and an incidental pulmonary mycosis. These findings suggest HL as an important differential of spontaneous death in captive porcupines. It is hypothesized that in addition to the widely documented causes, HL in captive porcupines may be specifically associated with nutritional imbalances caused by the feeding of unsuitable commercial diets. The possible association of the condition with dietary and other factors in captive porcupines needs to be thoroughly investigated.
Keywords
Presently, not much is known about the various diseases that affect porcupines, and this is reflected by the limited number of disease descriptions in literature. 7,9,11,19 From the viewpoint of diagnosticians, zoo veterinarians, and wildlife veterinary practitioners, this poses a hindrance to the accurate diagnosis and treatment of ailments that affect porcupines. As such, there is a need to document disease syndromes in this animal species so as to provide reference data for those engaged in the diagnosis and treatment of porcupine diseases. Cutaneous acariasis, 19 toxoplasmosis, 11 baylisascariasis, 7 and hepatic capillariasis 9 are among the few porcupine diseases that have been documented in the literature. Recently, a case of hepatic lipidosis (HL) was reported in a 1-year-old captive North American porcupine raised in a Belgium zoo. 2 The authors of that report concluded that the interplay of nutritional factors and gastrointestinal parasitism was responsible for the death. Whether the HL incidence in captivity is comparable to that in wild porcupine populations is not presently known.
Porcupines in the wild are herbivorous animals that feed on grasses, leaves, flowers, and herbs during the spring and summer 1 and nuts, phloem, and cambium of coniferous and deciduous trees during winter. 10 In captivity, they are commonly fed commercial diets supplemented with a variety of vegetables and fruits. Research done in the late 1960s suggested that a daily caloric intake of 435 kcal was adequate for a 10-kg porcupine. 13 More recent experimental work has estimated a daily nitrogen (protein) requirement of 346.0–389.4 mg N/kg body weight in these animals. 6 Pregnant and periparturient mammals show a considerable increase in energy expenditure. In respect to porcupines, lactating females require about 17% more calories than nonlactating females. 5 Hepatic lipidosis and pregnancy toxemia have been reported in humans 8,17 does, cows, and ewes. 18 In women, hepatic dysfunction complicates about 3% of pregnancies 8 and tends to occur in the last trimester of gestation. Similarly, ewes on a low-energy diet in late gestation frequently develop pregnancy toxemia and HL. 18
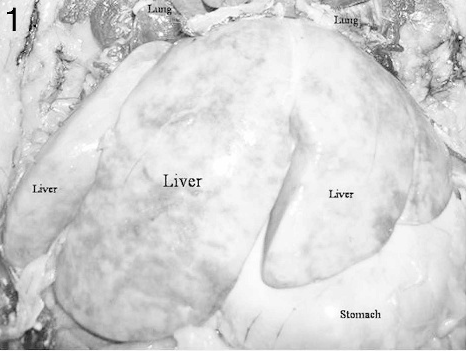
Liver from an adult male porcupine with severe hepatic lipidosis. Note the marked hepatomegaly, extreme pallor, and the rounded borders.
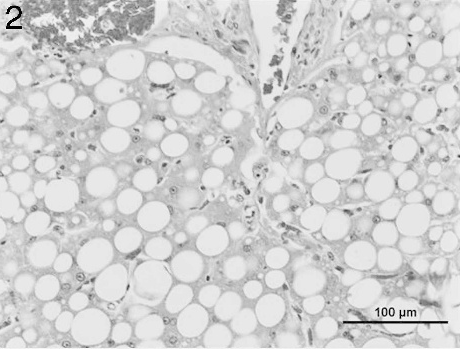
Liver from an adult male porcupine with severe hepatic lipidosis. Note the large unilocular cytoplasmic vacuoles. In most of the affected hepatocytes, the nucleus is squeezed into the displaced rim of cytoplasm. Hematoxylin and eosin. Bar = 100 μm.
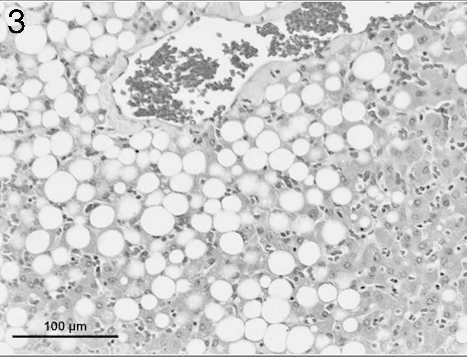
Liver from an adult female porcupine with severe hepatic lipidosis. Note the large unilocular cytoplasmic vacuoles. Hematoxylin and eosin. Bar =100 μm.
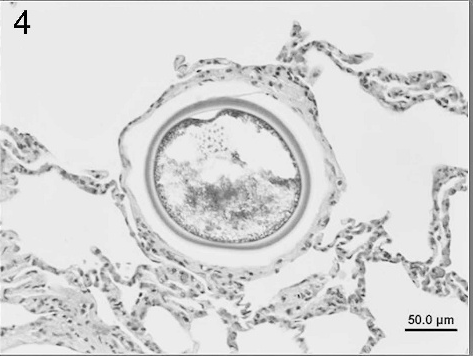
Lung from an adult female porcupine showing a large intraalveolar spherule consistent with Emmonsia crescens. Alveolar septae are infiltrated by a few inflammatory cells. Hematoxylin and eosin. Bar = 50 μm.
Hepatic fatty change, as HL is sometimes called, is characterized by intracytoplasmic accumulation of triglycerides within hepatocytes. This is usually associated with physiological factors 3 or pathological states. 12 The HL caused by nutritional and metabolic imbalances tends to be diffusely distributed, unlike the one caused by toxic or anoxic injury. 14 As is true for other mammalian species, we speculate that dietary and other factors, including a relatively sedentary lifestyle in captivity and pregnancy or lactation-related stress, are important in the pathogenesis of HL in captive porcupines. Comparative studies involving both wild and captive porcupines are needed to test this hypothesis. This report documents HL in 2 captive porcupines raised under similar management conditions and compares the 2 cases with another that was recently reported in the literature. The incidental finding of a pulmonary mycosis and a lone intraperitoneal nematode in the female porcupine are briefly discussed.
In July 2006, a dead captive male porcupine estimated to be about 4-years old was presented to the Veterinary Diagnostic Laboratory of North Dakota State University (VDL-NDSU) with a history of sudden death. The animal was wild caught in February 2005 and raised in captivity at the Red River Zoo in Fargo, North Dakota. According to the caretaker, the animal appeared clinically normal 3 hours before death. Subsequently, in May 2007, an adult female porcupine from the same zoo died spontaneously, and the carcass was sent for testing at the VDLNDSU. The animal was taken into captivity on April 9, 2007, and gave birth to a single male porcupette 19 days after capture. The dam was left to nurse her young for 2 days after which the neonate was withdrawn and hand-raised by the zookeepers. Questionnaire responses provided by the animal caretaker revealed that a nutritionist had recommended a commercial diet of browser pellet (high fiber/low protein) a supplemented with apples and carrots. Routine health practices at the facility included a yearly rabies vaccination and treatment with ivermectin for internal parasites. No routine blood work was reported.
A summary of biodata, dietary history, and test findings of 3 captive porcupines diagnosed with hepatic lipidosis.

Case reported by de Boscherre et al. 2006.
Indicates the 2 cases investigated in this report
The periparturient female porcupine of this report.
For both animals, a necropsy was done and a thorough macroscopic evaluation of all organ systems made. Fresh tissue samples including the lung, liver, small intestine, colon, kidney, and brain were taken for bacteriology and intestinal contents from the male porcupine tested by the fecal flotation technique. For aerobic culture, lung, liver, and intestine were inoculated on tryptose soy agar II 5% sheep blood b and incubated at 35°C for 12 hr under aerobic conditions. Representative tissue samples from both animals were fixed in 10% buffered formalin, processed routinely, and paraffin embedded. Five-micron sections were deparaffinized, stained with hematoxylin and eosin, c and examined by light microscopy. To further characterize the cytochemical properties of the intrapulmonary organism, the lung section from the female porcupine was stained with periodic acid-Schiff (PAS). c
At necropsy, the carcass of the male porcupine showed signs of obesity as implied by excessive amounts of subcutaneous and mesenteric fat deposits. The most remarkable gross finding was a markedly enlarged pale and friable liver that had rounded borders (Fig. 1). The gastric mucosa had scattered petechial hemorrhages. Rare mucosal ulcers measuring about 2 mm in diameter were seen in the colon. The rest of the organs and tissues appeared within normal limits. No parasitic eggs or ova were seen in intestinal contents tested by the fecal flotation procedure. The carcass of the female porcupine had a small crusty and partially healed wound on the caudal area of the abdomen by the linea alba. The right kidney was about 50% of the size of the left one. A white nematode macroscopically consistent with Setaria spp. was found in the perihepatic area of the peritoneal cavity. This parasite measured about 2.0 mm × 35.0 mm and was still alive when found. Apart from a few patchy areas of paleness, the liver appeared rather unremarkable. The uterus contained small quantities of very thick dirty brown material interpreted to be lochia.
Microscopic evaluation of histologic sections of the liver section from the male porcupine (Fig. 2) revealed severe hepatocellular fatty degeneration involving the entire hepatic lobules. The lesions were characterized by large, round cytoplasmic vacuoles that pushed the nuclei of the affected cells to the periphery. No significant changes were seen in other tissues. Compared to the male, the female porcupine liver (Fig. 3) showed less marked fatty degeneration characterized by large unilocular cytoplasmic vacuoles involving the hepatocytes. The lung had numerous randomly distributed intraalveolar fungal spherules that were rarely associated with a tissue reaction. Where inflammation was seen, it was composed of a mixture of low to moderate numbers of lymphohistiocytic cells along with few neutrophils and rare eosinophils. A thin fibrous tissue capsule was seen in the periphery of a minority of the fungal spherules. The spherules were 50–180 μm in diameter and had a thick eosinophilic staining wall measuring about 5 μm (Fig. 4). The interior of the spherules contained abundant amounts of basophilic staining granular material. Both the capsule and the intraspherular granular material stained positive with PAS. b Numerous blood vessels of the lung showed slight to moderate numbers of marginating leukocytes composed of low numbers of lymphocytes and rare neutrophils. A few airways showed a moderate increase in the number of peribronchiolar lymphocytes, and alveolar septae were infiltrated by very low numbers of lymphohistiocytic cells along with rare numbers of eosinophils. No remarkable changes were seen in sections of the adrenal gland, stomach, spleen, kidney, myocardium, cerebral cortex, cerebellum, and brain stem. Aerobic culture of samples from the male porcupine yielded a mixture of contaminants including Staphylococcus spp. and α Streptococcus spp. from the lung; Escherichia coli from the intestine; and no growth from the liver. Samples from the female porcupine also yielded a mixture of contaminants including Bacillus spp., α Streptococcus spp., and Pseudomonas spp. from the liver; Staphylococcus spp., α Streptococcus spp., and Pseudomonas spp. from the intestine; and no growth from the lung.
This communication reports HL in 2 captive porcupines that were being raised under the same zoo conditions. Considering lesion severity, it is concluded that the liver pathology contributed to the cause of death in the male porcupine. This animal showed classical gross and microscopic changes that were consistent with HL and comparable in severity and distribution (Fig. 2; Table 1) to those seen in the case reported by Boschere et al. 2 Interestingly, the female porcupine did not show characteristic gross hepatic changes despite a microscopically detectable HL of moderate intensity. The different grades of hepatic lesions in these two animals (Figs. 2, 3; Table 1) may be a function of diet and time spent in captivity. In addition, the female porcupine had just given birth, suggesting that increased lactation-associated energy expenditure coupled with possible stress due to withdrawal of her young may have contributed to the development of the hepatic lesions. It has been reported that lactating porcupines require about 17% more energy than nonlactating ones. 5
In the wild, porcupines are herbivorous but as indicated by the caretaker, the 2 animals were being fed a diet formulated for laboratory rodents. A case of HL was recently reported in a zoo-raised porcupine 2 and after consideration of the diet and test findings (Table 1), these authors suggested that nutritional imbalances including a deficit in energy resorption and increased mobilization of body fats were most likely responsible for the liver pathology seen. Spontaneous HL is a well-known disease entity in cats 4 and ponies 15 and has been attributed to obesity and nutritional stress. The male porcupine in the present report had excessive amounts of subcutaneous and mesenteric fat that most likely were a result of a high calorie diet. However, because porcupines in the wild are freely ranging animals, other factors like a relatively sedentary lifestyle under captivity, cannot be ruled out as contributory causal factors. On the other hand, the female porcupine did not show comparable amounts of body fat reserves. This finding suggests that, as occurs in other animal species, 4 a periparturient lactation-related energy drain coupled with nutritional stress may have provoked excessive mobilization of body fat. This is a plausible argument considering the recent history of pregnancy and parturition at a time of transitioning from a natural diet in the wild to a commercial feed in captivity.
Based on the characteristic size and shape of the adiaspores plus the thick wall and positive PAS staining, the numerous fungal spherules seen in the lung of the female porcupine were morphologically consistent with Emmonsia crescens but did not appear to cause an appreciable pathological response. Thirty years ago, Tobon et al. 20 reported E. crescens-induced pneumonia in a squirrel but not in the lone porcupine sampled in that study. Our results provide the first evidence that incidental E. crescens infections in porcupines are possible. Setaria spp. has not been reported in porcupines but is known to cause peritonitis and perihepatitis in wild North American ungulates. 16 Whether porcupines serve as a primary or aberrant host to this parasite needs to be clarified.
In conclusion, findings in this report underscore the apparent significance of HL as a disease entity in captive porcupines and for the first time document the condition in 2 animals exposed to the same environmental and management conditions. Additionally, presumed incidental pulmonary emmonsiosis and peritoneal setariasis are reported in 1 of the 2 porcupines; however, it remains unknown whether these 2 agents contributed to death. Evaluation of our data suggests that the universally known etiopathogenetic mechanisms may apply to HL in captive porcupines. Even though the numbers of porcupines reported with HL are still of questionable statistical significance, the present findings corroborated by the recently reported case 2 should indicate to diagnosticians that HL is an important differential in spontaneous deaths in captive porcupines. There is a need for animal nutritionists to empirically investigate appropriate diets for captive porcupines.
Footnotes
a.
Rodent Chow browser pellet (LabDiet® 5001), PMI® Nutrition International, Richmond, IN.
b.
Becton Dickinson, Sparks, MD.
c.
VWR International, West Chester, PA.
