Abstract
Non-commercial poultry flocks (referred to as “small flocks”) have become increasingly popular in Canada. Despite this popularity, little is known about the main causes of morbidity and mortality (health status) in these flocks. We assessed the baseline prevalence of infectious and non-infectious diseases among Ontario’s small poultry flocks by conducting a prospective surveillance study over a 2-y period. With the owner’s consent, for each bird (
Keywords
Introduction
The practice of growing poultry in peri-urban and urban areas (i.e., backyard poultry) has increased globally, thanks to the contribution of “organic farming” and “urban agriculture” movements.4,7 Despite their increased popularity, the health status of these flocks remains poorly documented. The few disease surveys regarding these flocks are mainly from the United States and Europe, and offer little information about the details of the postmortem findings, primarily focusing on pathogen prevalence. In these studies, infectious etiologies (viral, bacterial, parasitic, and fungal) have been reported to account for >60% of mortalities in autopsied birds, with Marek’s disease (6–26% of small flock poultry), mycoplasmosis (2–25%), and colibacillosis (7–16%) accounting for the most common causes of disease and death.3,6,9
With the goal of evaluating the health status of these flocks in Ontario, we conducted a prospective surveillance study of birds from small flocks that were submitted for postmortem examination to the Animal Health Laboratory (AHL) over a 2-y period (October 2015–September 2017). Results of our study are reported in 2 companion papers. The present work, the second paper of the series, illustrates the results derived from the postmortem analysis, focusing on the causes of morbidity and mortality (disease prevalence). The first paper of the series reports the results of the pre-set microbiology tests (pathogen prevalence). 2
Materials and methods
Details concerning the study design are reported in part 1 of the series. 2 Information on the bird species, age, sex, flock size, location, date of submission, and clinical history was collected as part of the postmortem submission. A routine postmortem examination was performed on all birds from each submission, including ancillary tests to determine the cause of morbidity or mortality. These tests could include histopathology, microbiology, virology, identification of internal and external parasites, and immunohistochemistry. The standard operating procedures routinely adopted by the AHL were used for all tests.
For molecular confirmation of Marek’s disease virus (6 chickens) and duck plague (
Interpretation and categorization of pathology data
A finalized report for each submission was issued by the AHL, including final diagnoses and results of ancillary tests for each bird in the submission, as well as the results of the pre-set tests derived from pooled samples (see part 1 of the series). 2 Findings of the finalized reports were entered into a spreadsheet (Excel 2016; Microsoft, Redmond, WA), formatted by individual bird and submission. All data entries were verified by a second member of the research team.
For each bird, final diagnoses listed in the pathology report were categorized as
Final diagnoses were further classified by body system (where the lesion occurred) and etiology (Table 1). Etiologies were broadly divided into infectious, non-infectious, idiopathic inflammation, and unknown. The idiopathic inflammation category was assigned to cases in which inflammatory lesions were observed, yet a specific etiology could not be determined. The unknown category was assigned if a pathologic process could not be clearly defined (lesions were not observed, or could not explain death or illness).
Number of primary and non-primary final diagnoses by body system and etiologic category, including the prevalence for each primary diagnosis, in small poultry flocks in Ontario, over a 2-y period.
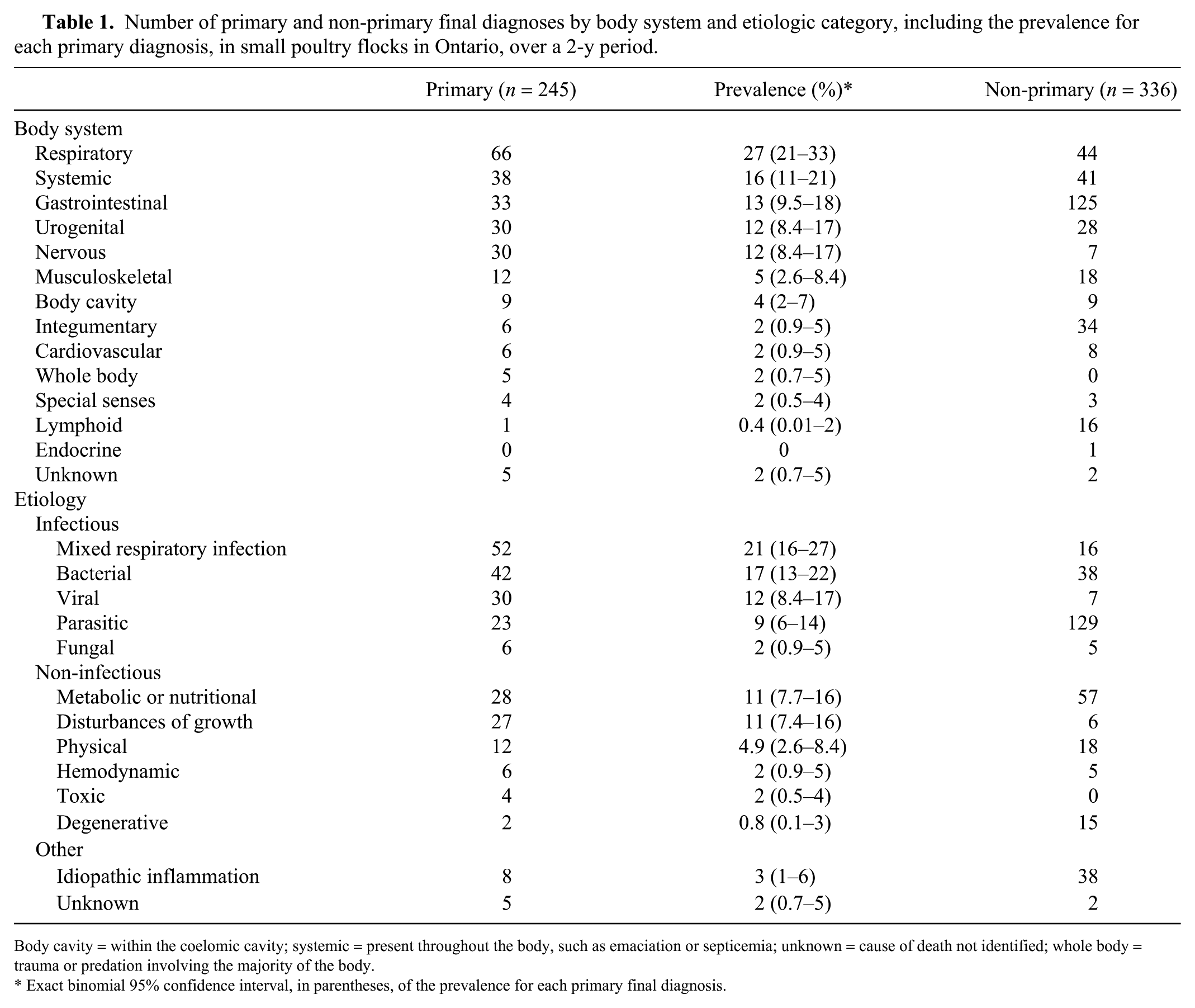
Body cavity = within the coelomic cavity; systemic = present throughout the body, such as emaciation or septicemia; unknown = cause of death not identified; whole body = trauma or predation involving the majority of the body.
Exact binomial 95% confidence interval, in parentheses, of the prevalence for each primary final diagnosis.
Classification of final diagnoses into etiologic categories was informed by the results of the ancillary and pre-set microbiology tests (see part 1 of the series), 2 when appropriate. Specifically, pathogens detected by the ancillary tests or pre-set tests were considered to be the causative agent of disease if lesions were consistent with infection, otherwise the pathogens were not considered to have caused disease. In submissions with multiple birds for which pooled samples were tested, for all birds with lesions consistent with infection, pathogens were considered to be the causative agent for that set of lesions.
Results
One hundred and sixty submissions were received, for a total of 245 individual birds. Birds received for postmortem included chickens (201 birds, 82%), turkeys (19, 8%), ducks (12, 5%), quail (6, 2%), peafowl (4, 2%), and pheasants (3, 1%). The age of birds ranged from 6 d to 7 y (median: 210 d; mean: 180 d). A total of 155 birds (63%) were recorded as female and 49 (20%) as male; the sex was not reported in 41 birds (17%) and entered as unknown.
Final diagnoses
Primary final diagnoses
Considering only the primary disease process for each bird, 245 primary final diagnoses were produced, one for each bird in the study (Tables 1, 2; Supplementary Table 1). The most common body systems affected were respiratory (27% of birds), systemic (16%), and gastrointestinal (13%). Overall, infectious causes of death represented 62% of cases (153 birds). When considered separately, the most common causes of death or illness in our study included mixed respiratory infections (21% of birds), bacterial diseases (17%), and viral diseases (12%).
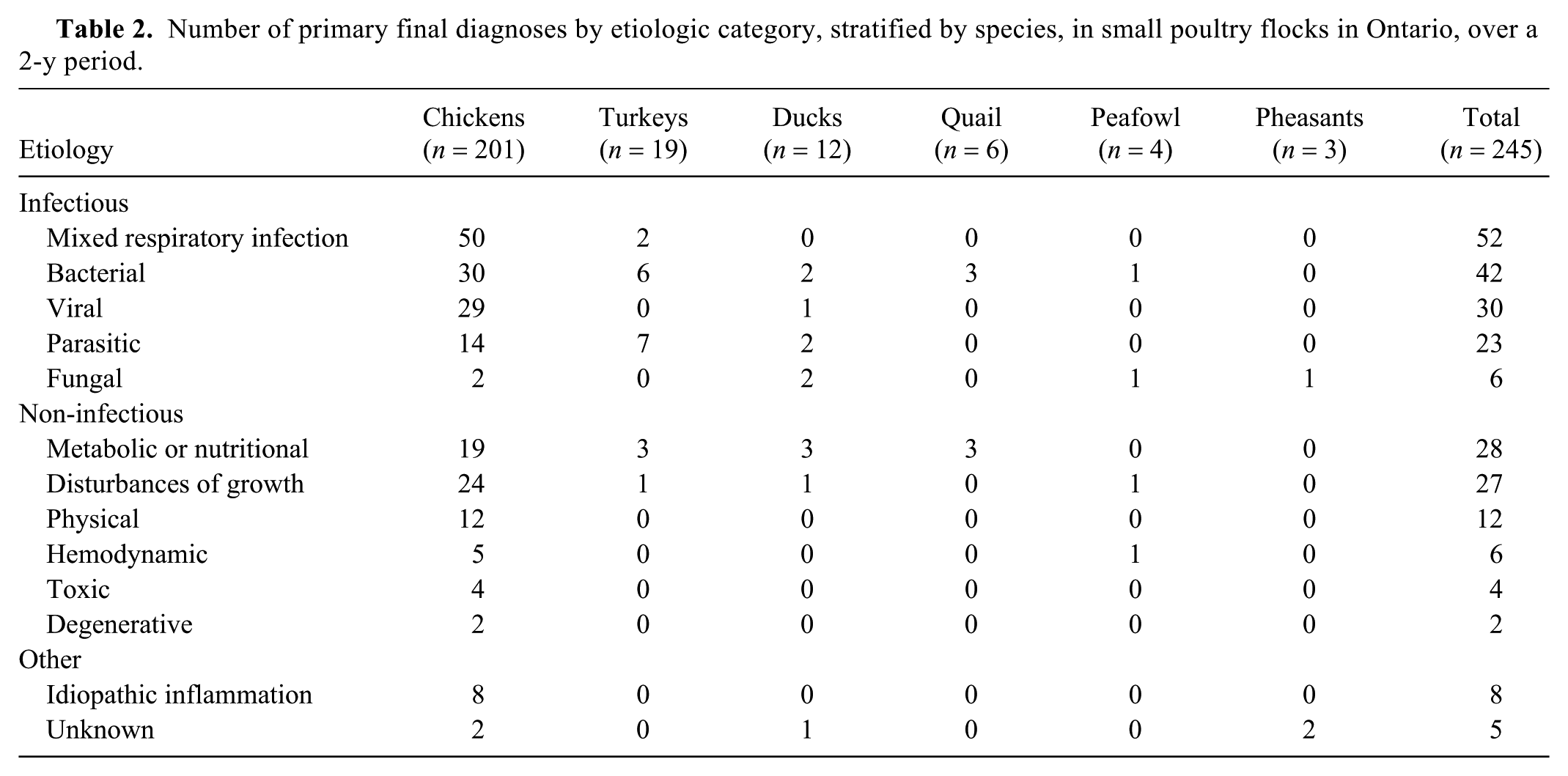
Number of primary final diagnoses by etiologic category, stratified by species, in small poultry flocks in Ontario, over a 2-y period.
Non-primary final diagnoses
Overall, 336 non-primary final diagnoses were identified, with an average of 1.4 non-primary diagnoses per bird (Table 1; Supplementary Table 1). The most common body systems affected were gastrointestinal (37% of non-primary diagnoses), respiratory (13%), and systemic (12%). The most common non-primary diagnoses were classified as parasitic (38% of non-primary diagnoses), metabolic or nutritional (17%), bacterial (11%), and idiopathic inflammation (11%).
Bacterial diseases
Primary bacterial diagnoses
Primary bacterial diseases were associated with the death of 42 birds (17% of primary diagnoses), including 30 chickens, 6 turkeys, 2 ducks, 3 quail, and 1 peafowl.
Pre-set tests for
Non-primary bacterial diagnoses
A total of 38 non-primary bacterial final diagnoses were determined in 32 birds (25 chickens, 3 turkeys, 3 ducks, and 1 peafowl). The most common bacterial agent,
Viral diseases
Primary viral diagnoses
Viral diseases were considered the primary cause of mortality in 30 of 245 birds (12% of primary diagnoses), including 29 chickens and 1 duck. Marek’s disease (MD) was the most commonly diagnosed primary viral disease (87% of primary viral diagnoses); it was identified in 26 chickens. A diagnosis of MD was based on the presence of lymphoid infiltrates within nerve trunks and visceral organs. Dubious cases (e.g., minimal lymphoid infiltrate of nerves) were tested by reverse-transcription, real-time PCR (RT-rtPCR) to assess the level of MD viral replication within tissues.
Based on the results of the pre-set tests,
2
infectious laryngotracheitis virus (ILTV; species
As part of the pre-set tests, infectious bronchitis virus (IBV; species
Duck viral enteritis was the primary disease diagnosed in a mature duck with esophagitis, enteritis, and hepatitis with intralesional intranuclear inclusion bodies and positive rtPCR results on the spleen and respiratory tissues (TAM-VMDL).
Non-primary viral diagnoses
A total of 7 non-primary viral diagnoses were determined in 7 birds (Table 1; Supplementary Table 1). Avian avulavirus 1 (AAvV-1; species
Mixed respiratory infection
Primary mixed respiratory infection
Mixed respiratory infection was considered the primary cause of mortality or illness in 52 birds (21% of primary diagnoses), including 50 chickens and 2 turkeys. A diagnosis of mixed respiratory infection was assigned when more than one respiratory pathogen was diagnosed as part of the pre-set (MG, MS, IBV, ILTV, AAvV-1) or ancillary microbiology tests requested by the pathologist (e.g., bacterial or fungal culture), in association with lesions (Supplementary Table 2). The 2 most common combinations of respiratory pathogens associated with primary lesions, as detected by pre-set tests, were MG, MS, and IBV (16 chickens) and MG, MS, IBV, and ILTV (8 chickens). Overall, bacteria most frequently isolated as part of primary mixed respiratory infections included
Non-primary mixed respiratory infection
Mixed respiratory infection was a non-primary diagnosis in 16 chickens, of which 2 were positive for IBV and AAvV-1 (Supplementary Table 2).
Parasitic and fungal diseases
Parasitic diseases were considered to be the primary cause of clinical signs or death in 23 birds (9% of primary diagnoses), the most common being coccidiosis (8 chickens), histomoniasis (5 turkeys), and acariasis (northern fowl mite, 5 chickens). As part of non-primary diseases, 129 parasitic infections (38% of non-primary diagnoses) were identified in 93 birds, including 48 caused by nematodes, 44 by protozoa (of which 23 were coccidia), and 27 by ectoparasites (14 with lice, 13 with mites).
Fungal infections were uncommonly the primary cause of disease. Two ducks and 2 game birds succumbed to respiratory aspergillosis, and in 2 chickens, crop candidiasis was considered the main cause of poor doing and emaciation. Crop mycosis caused by
Metabolic or nutritional diseases and toxicoses
A total of 28 birds (11% of primary diagnoses) died as a result of metabolic and/or nutritional diseases. Lesions associated with calcium, phosphorus, and vitamin D imbalances were the most common metabolic or nutritional condition (11 birds, 39% of primary metabolic diseases). Of these, 6 chickens, 1 duck, and 1 quail had thickening of the proximal tibiotarsal growth plates and enlarged parathyroid glands, suggesting rickets. Three hens had osteomalacia with thin brittle bone, characteristic of cage layer fatigue. Fifty-seven non-primary metabolic or nutritional diagnoses were observed in 50 birds, with the most common being emaciation and dehydration (15 and 8 birds, respectively), renal urate deposits (11 chickens and 1 quail, 13 diagnoses), and metabolic bone disease (9 chickens).
Ammonia toxicosis was suspected in 4 chickens from 1 submission with lesions of ulcerative and heterophilic keratoconjunctivitis, and anterior uveitis (primary cause of clinical signs). No other toxicoses were identified in the submitted birds (Table 1; Supplementary Table 1).
Disturbances of growth
Disturbances of growth were considered to be the primary cause of clinical signs or death in 27 birds (11% of primary diagnoses); of these, 26 died as the result of non-viral neoplastic disease. There were 20 cases of neoplasia of the urogenital tract in chickens (18 females and 2 males), including 16 cases of adenocarcinoma of the ovary and/or oviduct, 2 cases of nephroblastoma (both males), 1 case of ovarian sex cord-stromal tumor, and 1 oviduct leiomyoma. The remaining cases of neoplasia included pancreatic adenocarcinoma (2 hens), hematopoietic neoplasms not consistent with MD (1 male chicken, 1 male turkey, 1 male peafowl), and a peripheral nerve sheath tumor in the left shoulder of a male duck. In one euthanized hen, the left oviduct was obstructed by a large space-occupying cyst, which was the cause of respiratory distress and cessation of lay (non-neoplastic disturbance of growth).
Six non-primary disturbances of growth were observed in 5 birds, including 2 chickens with leiomyomas (considered to be incidental; 1 hen, 1 sex unknown), 1 hen with cysts of the ovary and oviduct, 1 hen with a heart valve malformation, and a female pheasant with colloid goiter (Table 1; Supplementary Table 1).
Physical, hemodynamic, and degenerative diseases
Predation, cannibalism, fractures, and other trauma-related injuries caused death in 8 chickens. An additional 3 chickens died as a result of being egg-bound, and 1 died as a result of crop impaction. Eighteen non-primary physical diagnoses were observed in 16 birds, with the most common being aspiration (5 birds), trauma (4 birds, 5 diagnoses), and egg-bound (4 birds).
Pulmonary hypertension was the primary cause of death in 3 chickens. Hemorrhagic liver syndrome was diagnosed in 2 chickens from the same submission. Intestinal hemorrhage, with no identifiable underlying cause, was the primary diagnosis in one peafowl. Non-primary diagnosis of portal vein thrombosis was present in one duck. Gastrointestinal hemorrhage and anemia were each present in 2 chickens that died of ovarian carcinomatosis, and mixed respiratory infection with marked intestinal parasitism, respectively.
Only 2 chickens were diagnosed with primary degenerative disease; 1 had myocardial dilation and 1 had myocardial fibrosis. Twelve birds had a total of 15 non-primary degenerative diagnoses, most of which (73%) consisted of lymphoid depletion in multiple lymphoid tissues, mainly bursa and spleen (Table 1; Supplementary Table 1).
Idiopathic and unknown causes of disease
When inflammation with no discernable cause was present, a diagnosis of idiopathic inflammation was assigned. A total of 8 birds (3% of primary diagnoses), all chickens, were considered to have died from idiopathic inflammation because the primary etiology could not be determined based on gross anatomic, histologic, and microbiologic testing. Non-primary inflammation of unknown cause was present in 38 different tissues, from 30 birds, most commonly affecting the urogenital tract (8 non-primary diagnoses) and respiratory system (7 non-primary diagnoses).
A primary cause of death or morbidity was not determined in 5 cases, including 2 chickens (found dead), 2 pheasants (found dead), and 1 duck (euthanized, submitted for health check).
Discussion
Overall, gross anatomic and histologic examinations of the birds allowed the case pathologist to determine a cause of death for the majority (95%) of birds, with only 5 cases having no final diagnosis and 8 having nonspecific inflammation as the primary cause of disease.
Respiratory disease caused by infection with multiple pathogens (i.e., mixed respiratory infection) was the most common cause of death or illness, accounting for 21% of the primary diagnoses. The presence of both bacterial and viral pathogens (and in rare instances, fungi) made identification of the inciting cause (i.e., primary agent) often impossible. Infectious agents, such as MG, MS, IBV, and ILTV can each cause respiratory disease on their own and can predispose birds to other respiratory infections. This is consistent with the relatively frequent isolation of other bacteria, such as
Voluntary testing of breeder flocks and hatcheries in the Ontario commercial poultry industry has made MG and MS extremely uncommon in the province. This is in stark contrast to the results of our study, where 20% and 23% of birds had respiratory lesions associated with detection of MG and MS, respectively. Sourcing birds and fertilized eggs from flocks with an unknown mycoplasma status rather than commercial hatcheries could account for the high prevalence that we observed. Once mycoplasma is introduced into the farm, it can remain indefinitely, only causing clinical signs during times of stress, such as the beginning of lay or transportation to poultry fairs. 10
Lymphoproliferative lesions consistent with MD were the main cause of death or clinical signs in 11% of birds, which was the highest rate of mortality associated with a single etiologic agent. However, the prevalence of MD-associated lesions in our study was lower than reported in the studies from California and Finland, which was as high as 22% and 27%, respectively.6,9 With MD as a common cause of mortality in small flocks, work is needed to increase awareness among small flock owners about the option of purchasing replacement birds that are vaccinated for this virus.
The most common sole bacterial pathogen causing disease or death in our study was
Neoplasia was the most common non-infectious cause of mortality in Ontario small flock birds, accounting for 11% of the submitted birds. This is similar to the survey conducted in California 1 yet higher than the study conducted in Finland, 9 which reported non-viral neoplasms in 9% and 2% of small flock birds, respectively. Of the 1,301 neoplasms associated with mortalities in birds from small flocks in California, 29% were adenocarcinoma, of which 73% and 17% derived from the ovary and oviduct, respectively. 1 In our study, 62% of the primary neoplastic processes were adenocarcinomas derived from the ovary or oviduct. This high prevalence might be explained by the overrepresentation of female birds in our study.
A relatively low percentage (<5%) of birds had signs of nutritional imbalance, which were suspected to be related to the quality of the feed ration offered to the flock, or possibly caused by excess supplemental feed (such as vegetable cuttings, scratch grains) that may limit the uptake of a balanced ration. Parasitic infection, both endoparasites and ectoparasites, were fairly common in the sampled birds; however, the submission of the parasites for formal identification was left to the discretion of the case pathologist, therefore some results were broadly classified. The wide variety of internal and external parasites is likely related to the practice of keeping small flocks in free-range settings and feeding rations that do not include coccidiostats, as shown by the questionnaires associated with our submissions (data not shown).
Supplemental Material
DS1_JVDI_10.1177_1040638719843575 – Supplemental material for A two-year prospective study of small poultry flocks in Ontario, Canada, part 2: causes of morbidity and mortality
Supplemental material, DS1_JVDI_10.1177_1040638719843575 for A two-year prospective study of small poultry flocks in Ontario, Canada, part 2: causes of morbidity and mortality by Nancy M. Brochu, Michele T. Guerin, Csaba Varga, Brandon N. Lillie, Marina L. Brash and Leonardo Susta in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the Animal Health Laboratory for the invaluable work in coordinating and processing all submissions and testing performed in this study, as well as Al Dam and Dr. Melanie Barham for their help in advertising the study. Summer students from the Ontario Veterinary College, Thisuri Eagalle, Kai Moore, and Elysha Smith, were invaluable in helping to organize the data. We also thank all of the veterinarians and small flock owners who submitted birds and participated in this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was funded by the Ontario Ministry of Agriculture, Food and Rural Affairs–University of Guelph Strategic Partnership (grant UofG 2015-2282), under the Disease Surveillance Plan, which was a joint federal-provincial Growing Forward 2 project. Financial support for Dr. Brochu’s stipend derived from the Ontario Veterinary College (OVC) DVSc fellowship fund. Summer students were supported through the OVC Andrea Leger Dunbar summer assistantship fund.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
