Abstract
In Ontario, within the past few years, there has been a marked increase in the number of non-commercial poultry flocks (referred to as “small flocks”). Small poultry flocks may act as a reservoir of avian and zoonotic pathogens, given the flocks’ limited access to veterinary services, inadequate biosecurity practices, and increased risk of contact with wild birds. Despite these potential risks, there is a scarcity of data concerning the prevalence of poultry and zoonotic pathogens among these flocks. To assess the baseline prevalence of bacterial and viral infectious pathogens, prospective surveillance of small flock postmortem submissions to the Animal Health Laboratory was conducted over a 2-y period. With the owner’s consent, a postmortem examination and pre-set tests for infectious agents were conducted. A total of 160 submissions, mainly chickens (84%), were received. Among bacterial pathogens,
Keywords
Introduction
In countries with an industrial poultry sector, such as Canada, there has been a substantial increase in the number of non-commercial poultry flocks (i.e., small flocks) raised in urban and peri-urban areas (Canadian Broadcasting Corporation, http://www.cbc.ca/news/the-backyard-chicken-debate-1.916153). In Canada, poultry production (chicken and turkey industries) is regulated by a quota system. In the province of Ontario, non-commercial poultry operations may have up to 299 broilers, 99 layers, and 49 turkeys. Non-commercial poultry owners can register their flock with a provincial marketing board (such as the Chicken Farmers of Ontario [CFO]) at the time of purchase of birds from broker dealers or hatcheries; in 2016, >16,000 non-commercial poultry growers registered their flocks (CFO, https://www.familyfoodgrower.ca/). However, a definitive number of non-commercial flocks within the province is not available and is likely higher than this, given that registration is voluntary and not done by owners who keep birds in urban areas where the practice is illegal (Yonge Street, www.yongestreetmedia.ca/features/backyardchickens05082013.aspx; Global News Radio 640 Toronto, https://globalnews.ca/news/3782510/toronto-city-council-approves-pilot-project-allowing-backyard-chickens).
In Canada and the United States, surveys have shown that relaxed biosecurity practices, insufficient veterinary supervision, and lack of prophylactic measures (e.g., vaccination) are common in non-commercial poultry flocks.4,5,12,20,23,32 A survey directed to Ontario’s small flock owners registered with the CFO in 2010–2011, demonstrated that nearly half of evaluated flocks had an increased risk of contact with wild birds.
4
Wild birds are considered potential reservoirs of avian pathogens, and increased likelihood of contact could facilitate spillover of several infectious agents, including avian influenza virus (AIV; species
Despite these concerns, the contribution of small flocks to the spread of high-consequence infectious agents, such as AIV and AAvV-1, has been small when compared to commercial flocks.1,33 For instance, during the H5N2 highly pathogenic AIV outbreak in Ontario in 2015, no small flocks tested positive for the virus (Canadian Food Inspection Agency [CFIA], http://www.inspection.gc.ca/animals/terrestrial-animals/diseases/reportable/ai/eng/1323990856863/1323991018946), and during the H7N3 highly pathogenic AIV outbreaks in British Columbia in 2004, only 2 small flocks tested positive, in contrast to 28 positive commercial premises. 29
Non-commercial flocks also represent a concern for public health by increasing the risk of human exposure to zoonotic and foodborne pathogens, such as
Despite the ever-increasing popularity of small poultry flocks, to our knowledge, there is limited published data documenting the baseline prevalence of poultry and zoonotic pathogens among these flocks in Canada, and more specifically, Ontario. 19 To establish the baseline prevalence of poultry pathogens and diseases among these flocks in Ontario, we conducted a prospective surveillance study of small flock postmortem submissions to the Animal Health Laboratory (AHL) over a 2-y period (October 2015–September 2017). An extensive array of pre-set microbiology tests and postmortem examinations were carried out for each submission. Results of our study are presented in 2 companion papers. In this first paper in the series, we report the results derived from the pre-set microbiology tests (pathogen prevalence). In the second paper of the series, we report detailed results of the postmortem investigation (disease prevalence). 3
Materials and methods
Advertisement and inclusion criteria
From October 1, 2015 to September 29, 2017, Ontario small poultry flock owners were encouraged to submit their birds for postmortem examination through a licensed veterinarian to the Guelph or Kemptville AHL (University of Guelph) locations. Submissions could be dropped off directly or delivered to these laboratories through a prepaid courier service. Owners were charged a nominal fee to include all testing.
The study was advertised through professional veterinary organizations (Ontario Association of Poultry Veterinarians, Ontario Veterinary Medical Association), industry groups (CFO, Poultry Industry Council, Ontario Broiler Hatching Egg and Chick Commission), poultry research networks (Poultry Health Research Network at the University of Guelph), blogs, social media, and periodicals specifically addressing poultry fanciers (The Exhibitor). The study was also advertised at cooperatives, hatcheries, and poultry fairs because these are common sites for the purchase of new birds.
To be included in our study, both the flock and flock owner must reside within the province of Ontario, the owner was required to complete a husbandry and biosecurity questionnaire, and the flock must be within the numerical limits imposed by the quota system and not have a primarily commercial aim (i.e., non-quota, non-commercial). In addition, each owner was required to sign a consent form as per University of Guelph requirements (Research Ethics Board REB-16-12-657). Included in the study were broilers, layers, and turkeys from non-quota flocks (i.e., flocks composed of a maximum of 299 broiler chickens, 99 layer chickens, or 49 turkeys). For other poultry species not regulated by the quota system (game birds and waterfowl), a maximum flock size of 300 birds was adopted, because it was considered representative of flocks not raised for commercial purposes. Birds in the Columbiformes order were excluded from the study. A submission was defined as having a maximum of 5 sick and/or dead birds (fresh dead or frozen) of 1 species from the same flock (i.e., different species were divided into different submissions, even if from the same owner). As per AHL policy, each submission was made by a referring veterinarian.
Postmortem examination and pre-set microbiology tests
Two sets of data were derived from each submission; one focusing on pathogen prevalence at the submission level, and the other on diseases and lesions at the individual bird level (Supplementary Fig. 1). The pre-set array of microbiology tests for infectious agents was conducted on pooled samples from all of the birds in each submission, regardless of history or pathology findings (Table 1). Therefore, negative or positive test results for a pathogen are reported on a submission basis, rather than on a bird basis. This was done to detect infection of the flock, rather than of single birds. A postmortem examination was also performed on each bird; the methodology and results for that part of the study are reported in the second paper of the series. 3 All tests were conducted in accordance with the standard operating procedures adopted by the AHL, an American Association of Veterinary Laboratory Diagnosticians–accredited diagnostic facility that functions as the provincial animal health laboratory for Ontario.
Pathogen prevalence at the submission level, and details of pre-set microbiology tests employed in a small poultry flock surveillance study in Ontario, over a 2-y period.
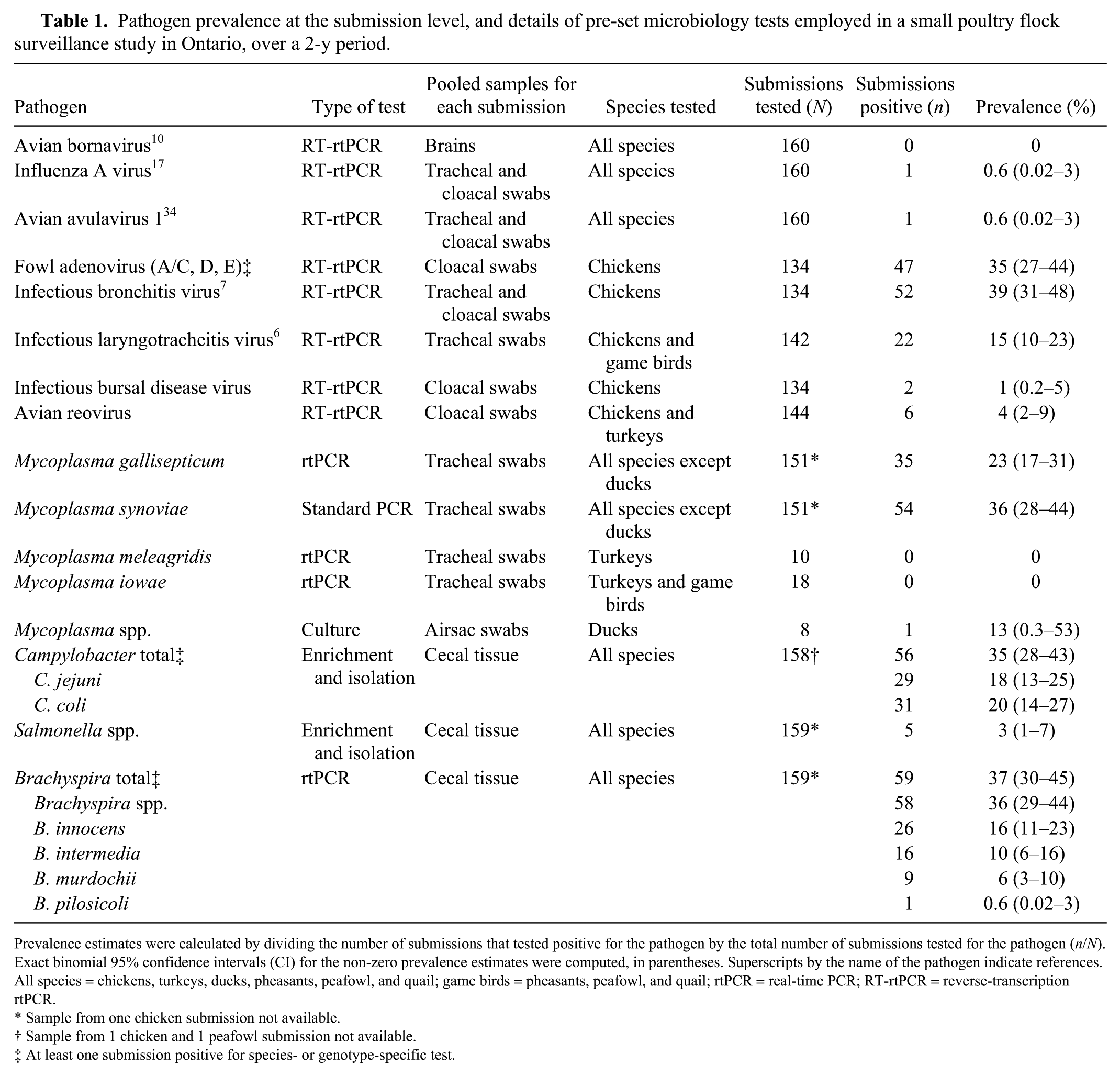
Prevalence estimates were calculated by dividing the number of submissions that tested positive for the pathogen by the total number of submissions tested for the pathogen (
Sample from one chicken submission not available.
Sample from 1 chicken and 1 peafowl submission not available.
At least one submission positive for species- or genotype-specific test.
All
Database formatting and data analysis
Data derived from the pre-set microbiology tests were entered into a spreadsheet (Excel 2016; Microsoft, Redmond, WA) formatted by submissions, and results were entered as 0 (negative) or 1 (positive). If a specific test was not performed on a submission (e.g., reverse-transcription, real-time PCR [RT-rtPCR] for IBV was not performed for waterfowl, game birds, or turkeys), results were entered as not applicable. Inconclusive RT-rtPCR results, as determined in the final report, were considered as negative.
For each pathogen, prevalence estimates were calculated by dividing the number of submissions that tested positive for the pathogen by the total number of submissions tested for the pathogen (Excel 2016; Microsoft). Exact binomial 95% confidence intervals for the non-zero prevalence estimates were computed (Stata v.14.1; StataCorp, College Station, TX). Venn diagrams (jvenn, http://jvenn.toulouse.inra.fr/app/index.html) were used to illustrate relations among sets (e.g., co-detection of respiratory pathogens) and subsets (e.g., co-detection of different species of
Results
Types of submissions received
Over the 2-y study period, we received 160 submissions of 1–5 birds (median: 1; mean: 1.3), for a total of 245 individual birds. Chickens accounted for 84% of submissions (134 submissions), followed by turkeys (10 submissions), game birds (8 submissions: 4 peafowl, 2 quail, and 2 pheasants), and ducks (8 submissions). Submissions originated from flocks of 1–299 birds (median: 25; mean: 26), mainly from southern and eastern Ontario (Fig. 1).
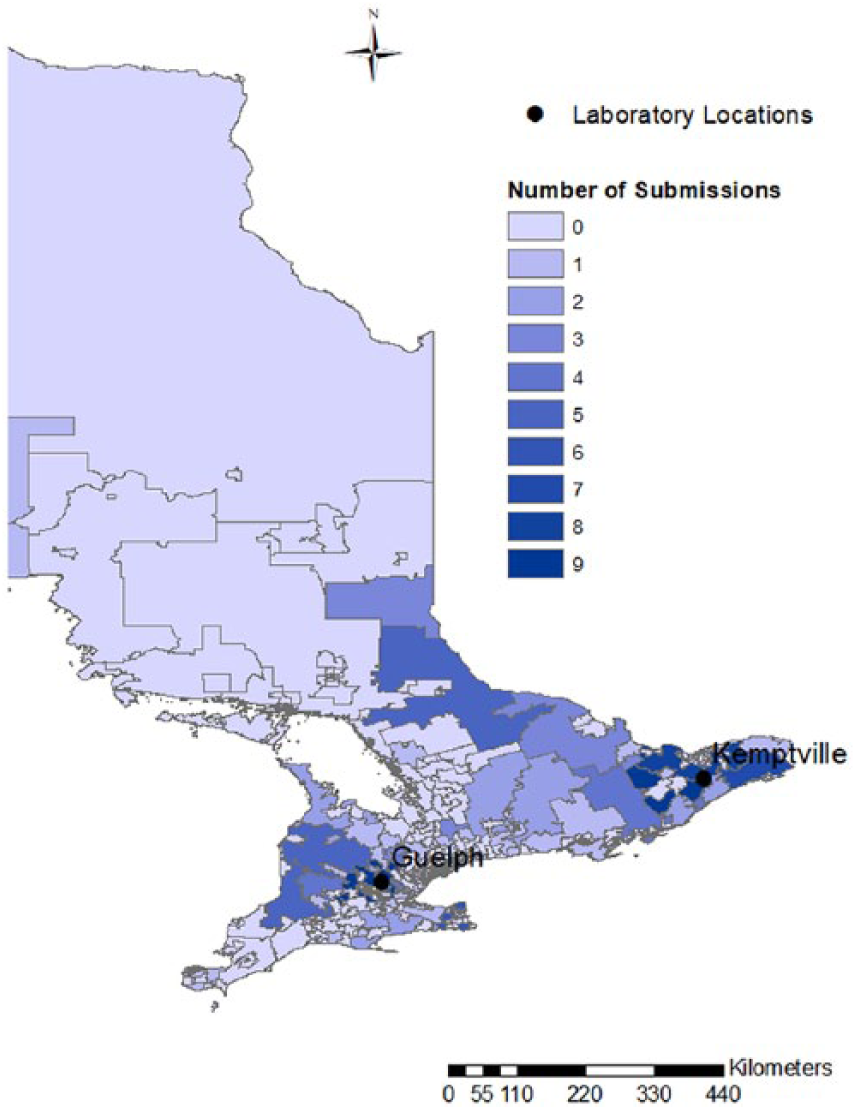
Choropleth map of Ontario, Canada, subdivided by forward sortation areas (first 3 characters of the postal code). The number of submissions from each area is represented using a heat map, with lighter colors denoting low numbers and darker colors denoting high numbers. The locations of the 2 Animal Health Laboratories are identified.
Pathogen detection by submissions
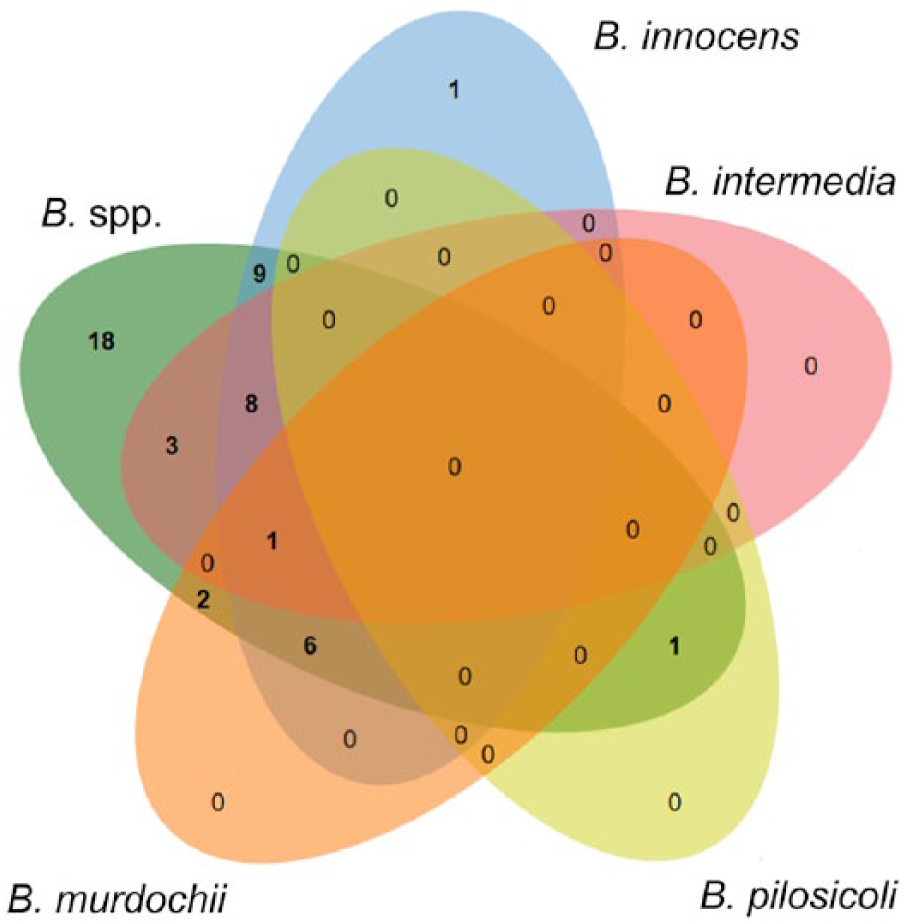
Salmonella spp., Campylobacter spp., and Brachyspira spp
Number of positive submissions and total number of submissions tested (in parentheses), by species, in a small poultry flock surveillance study in Ontario, over a 2-y period.
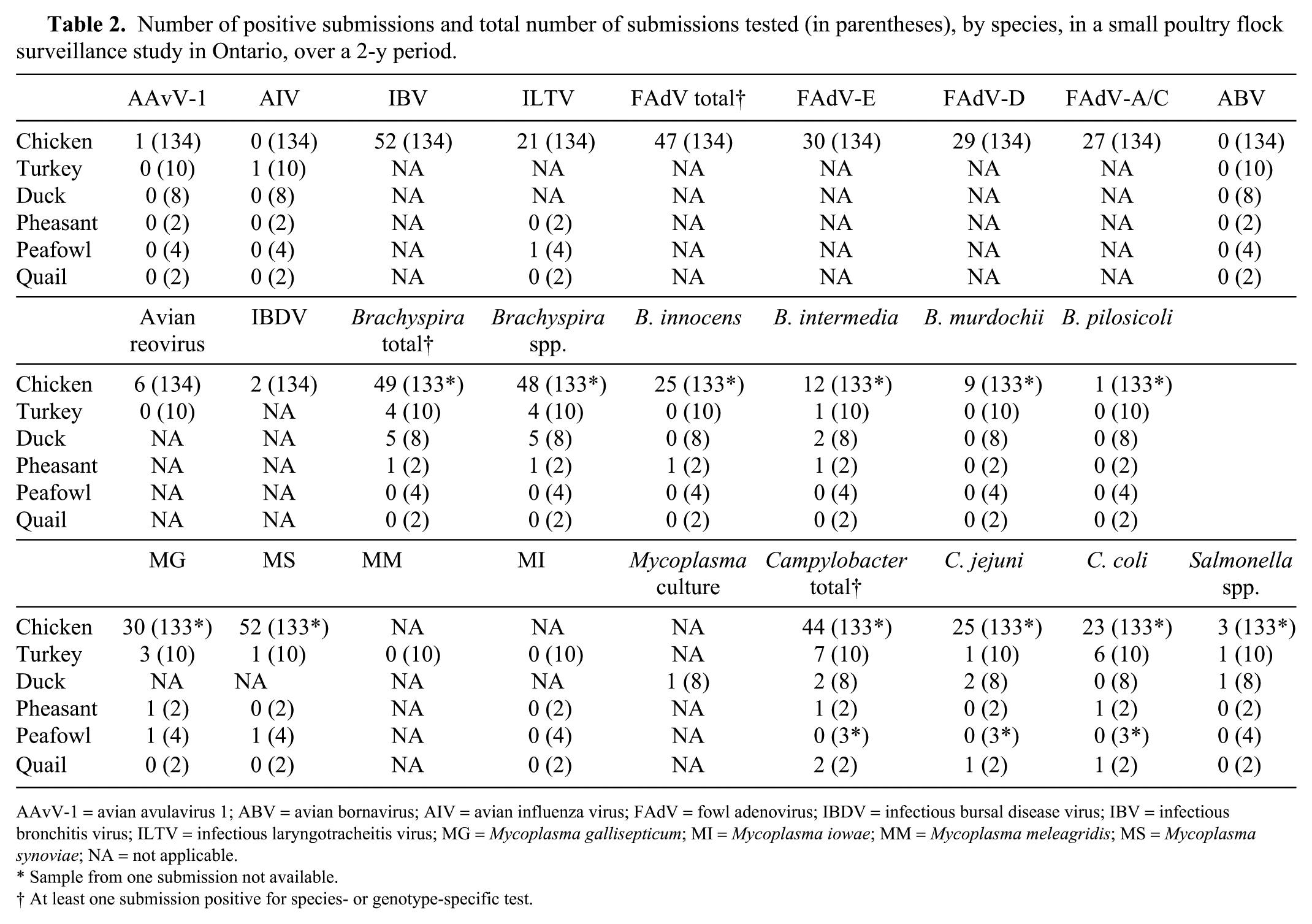
AAvV-1 = avian avulavirus 1; ABV = avian bornavirus; AIV = avian influenza virus; FAdV = fowl adenovirus; IBDV = infectious bursal disease virus; IBV = infectious bronchitis virus; ILTV = infectious laryngotracheitis virus; MG =
Sample from one submission not available.
At least one submission positive for species- or genotype-specific test.
Real-time PCR for
Venn diagram illustrating relations among species in
Fowl adenovirus, avian reovirus, infectious bursal disease virus, and avian bornavirus
FAdV was detected in 35% of 134 chicken submissions (Tables 1, 2). A total of 30, 29, and 27 submissions were positive for FAdV genotype E, D, and A/C, respectively; many were positive for more than 1 genotype (Fig. 3). Avian reovirus was detected in 4% of 144 chicken and turkey submissions; all of the positives were chicken submissions. Infectious bursal disease virus (IBDV) was detected in 1% of 134 chicken submissions. Avian bornavirus was not detected in any submissions.
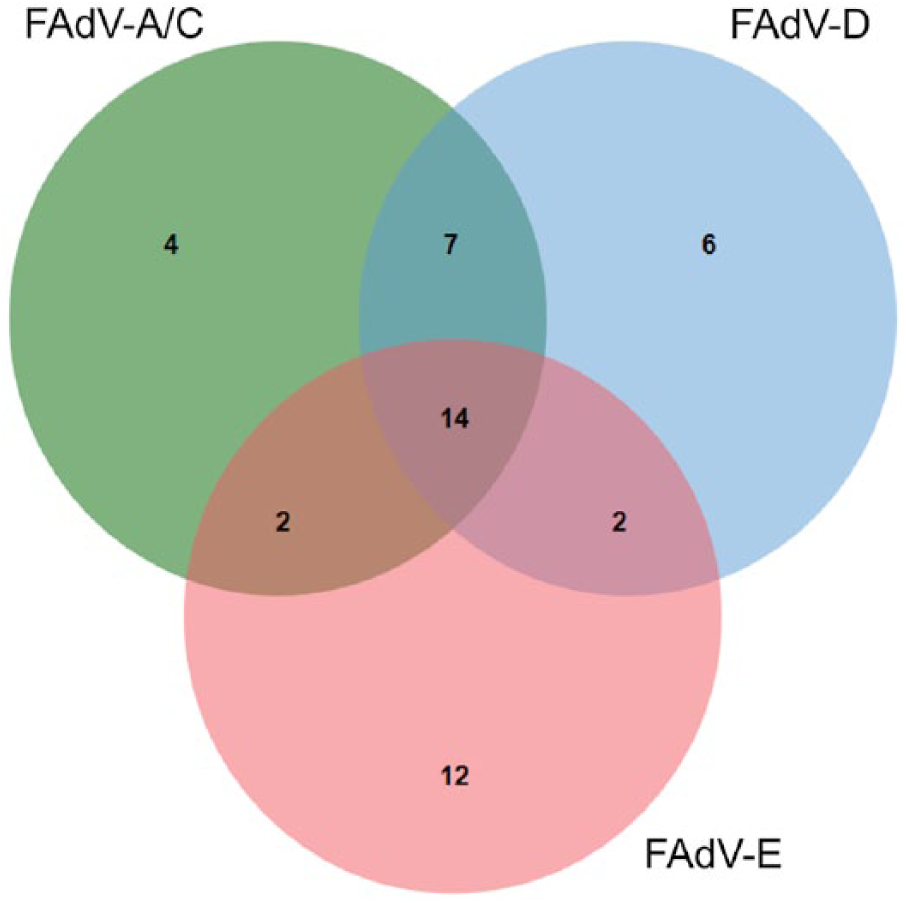
Venn diagram illustrating relations among genotypes in fowl adenovirus (FAdV)-positive chicken submissions in a small poultry flock surveillance study in Ontario. The presence of FAdV was detected using reverse-transcription, real-time PCR on pooled cloacal swabs. Genotypes A/C, D, and E were differentiated based on multiplex assays designed to detect the FAdV genotypes found most commonly in Canadian chickens.13,28
Mycoplasma spp
Infectious bronchitis virus
IBV was detected in 39% of 134 chicken submissions (Tables 1, 2). Specifically, in 26 submissions, IBV was detected in both tracheal and cloacal swabs, in 1 submission it was detected in the tracheal swab only, and in 25 submissions it was detected in the cloacal swab only.
Of the 52 positive submissions, partial sequencing of the
Infectious laryngotracheitis virus
ILTV was detected in 15% of 142 chicken and game bird submissions, of which 21 were from chickens and 1 from a peafowl (Tables 1, 2). Partial sequencing detected a vaccine strain in 12 of 22 positive submissions (including the peafowl submission) and a wild-type strain with high similarity to the Niagara strain reported in the 2004 Ontario outbreak 27 in 9 submissions. The ILTV strain of one submission differed from both the vaccine strain and the Niagara strain, and did not match strains in GenBank.
Avian avulavirus and avian influenza virus
Non-virulent AAvV-1 was detected from a 2.5-wk-old chicken submission (2 birds); AIV was detected from one 2-wk-old turkey poult (Tables 1, 2). Both samples were sent to the NML for further characterization and were determined to be a lentogenic vaccine strain (AAvV-1) and a wild-bird origin low pathogenicity AIV (H10N8).
Co-detection of respiratory pathogens
The frequency of co-detection of MG, MS, IBV, and ILTV was evaluated for chicken submissions (Fig. 4). A total of 44 (33%) of 133 tested chicken submissions were positive for more than one of these respiratory pathogens. The most common combinations included MG, MS, and IBV (9%); MS and IBV (8%); and MG and MS (4%). For other species, 1 peafowl submission was positive for both MS and ILTV, and 1 turkey submission was positive for both MG and MS.
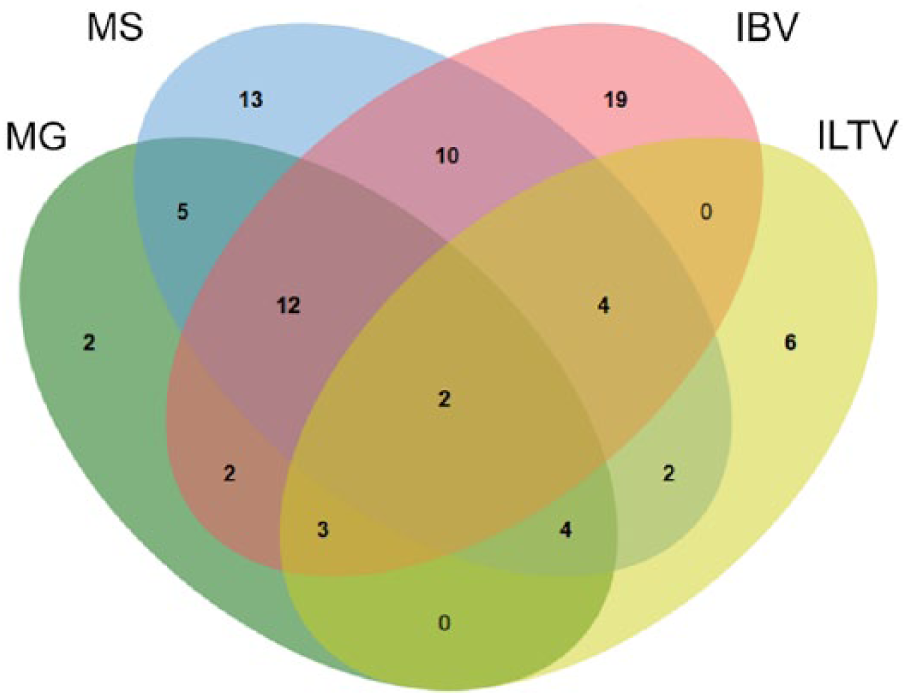
Venn diagram illustrating relations among 4 respiratory pathogens (MG =
Prevalence of pathogens by poultry species
Among chicken submissions, the most commonly detected pathogens were MS and IBV (39% each),
Discussion
A 2014–2015 survey of healthy birds sampled at Ontario provincial abattoirs reported a significantly lower prevalence of
The 35% overall prevalence of
Certain
Estimated prevalence of FAdV, IBDV, and avian reovirus in Ontario’s commercial broiler flocks, based on slaughter samples, has been reported as 96%, 49%, and 91%, respectively.13,26 The prevalence of the same pathogens for chicken submissions in our survey was markedly lower, with only 35% positive for at least one genotype of FAdV, 1% positive for IBDV, and 4% positive for avian reovirus. However, it should be noted that both serology and pathogen detection methods were used in the estimation of prevalence in the studies involving commercial flocks. Further, the chickens in our study were primarily layers or dual-purpose birds. These methodologic and population differences may explain some of the discrepancy between studies. Overall, none of the tested enteric pathogens (
The strains of IBV that we detected in Ontario small flocks between 2015 and 2017 are similar to those reported within Canada between 2000 and 2013. 24 Concurrent with the sampling period of our small flock study, the AHL detected an increase in both submissions and positive test results for IBV in all commercial chicken commodities in Ontario (Ontario Animal Health Network Poultry Expert Network, https://oahn.ca/wp-content/uploads/2016/12/Dec-2016-OAHN-special-report-on-IBV-FINAL.pdf). During this time, the Delmarva (DMV) strain emerged as the predominant strain in commercial poultry, whereas only 3 of the small flocks in our study tested positive for the DMV strain. These 3 positive flocks were sampled during October 2016 and July 2017, after the DMV strain was identified in the commercial industry in September 2016, suggesting spread from the commercial sector to small flocks.
Strains of ILTV from our study included both wild-type and Niagara outbreak–like strains, both of which have been described in Ontario’s commercial poultry industries. 27 The presence of a vaccine-like strain in 12 submissions suggests that vaccine strains might be circulating among small flocks, possibly as a direct consequence of ILTV vaccination of these flocks (likely a rare event), or routine vaccination in commercial layers and breeders. 27 The detection of the Niagara-like strain in 9 submissions suggests that the strain from the 2004 commercial outbreak has now become endemic in the province. 27
The seroprevalence of various pathogens, such as those detected in our samples, has also been reported in various studies of small poultry flocks from the United States, Belgium, and Finland conducted between 1988 and 2016.11,15,21,22,25,30 In those studies, the prevalence of AAvV-1, AIV, IBV, IBDV, ILTV, MG, and MS was 0–78%, 0–23%, 47–91%, 20–74%, 12–77%, 13–73%, and 56–96%, respectively. One of the studies from California
11
found that birds purchased from hatcheries certified under the National Poultry Improvement Plan, a U.S. program similar to the Ontario Hatchery and Supply Flock Policy to ensure pathogen-free replacement chicks, had a lower antibody prevalence for AAvV-1, MG, and MS compared to flocks with birds from other sources. The Belgian study
15
also reported a 100% flock seroprevalence for
The potential for pathogen exposure and spread from small flocks to commercial poultry flocks has long been a point of contention.11,15,23,25 An outbreak of any of the 4 federally reportable poultry diseases in Canada (Newcastle disease, notifiable avian influenza, pullorum disease, fowl typhoid; CFIA, http://www.inspection.gc.ca/animals/terrestrial-animals/diseases/reportable/eng/1303768471142/1303768544412) could have a dramatic impact on the commercial poultry industry. With only one positive test result for non-notifiable AIV and one for vaccine strain AAvV-1, and no
Given that this was a passive surveillance project based on voluntary submissions, the 160 flocks tested in our study may not be representative of all small flocks within the province. On the other hand, an active surveillance program was not logistically or financially feasible in a province with a population of >13 million people spread over a million square kilometers (Statistics Canada, http://www12.statcan.gc.ca/census-recensement/2016/as-sa/fogs-spg/Facts-pr-eng.cfm?Lang=Eng&GK=PR&GC=35&TOPIC=1), and where a complete registry of small flocks is not available. The majority of submissions originated from areas relatively close to the AHL laboratories, possibly indicating a higher population density in metropolitan areas near the AHL facilities, or a lower willingness of submitters to ship postmortem samples over long distances. The $25 processing fee required for each submission, albeit heavily subsidized, could have decreased the number of submissions depending on an individual owner’s perceived value of the bird(s). In addition, small flock owners who did not have a veterinarian–client–patient relationship already in place might not have been able to submit their birds. Finally, some small flock owners might have been reticent to submit birds, either because of the ambiguity of municipal regulations or from fear of investigation by government authorities if a reportable disease was detected.
Supplemental Material
DS1_JVDI_10.1177_1040638719843577 – Supplemental material for A two-year prospective study of small poultry flocks in Ontario, Canada, part 1: prevalence of viral and bacterial pathogens
Supplemental material, DS1_JVDI_10.1177_1040638719843577 for A two-year prospective study of small poultry flocks in Ontario, Canada, part 1: prevalence of viral and bacterial pathogens by Nancy M. Brochu, Michele T. Guerin, Csaba Varga, Brandon N. Lillie, Marina L. Brash and Leonardo Susta in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the Animal Health Laboratory for the invaluable work in coordinating and processing all submissions and testing performed in this study, as well as Al Dam and Dr. Melanie Barham for their help in advertising the study. Summer students from the Ontario Veterinary College, Thisuri Eagalle, Kai Moore, and Elysha Smith, were invaluable in helping to organize the data. We also thank all of the veterinarians and small flock owners who submitted birds and participated in this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was funded by the Ontario Ministry of Agriculture, Food and Rural Affairs–University of Guelph Strategic Partnership (grant UofG 2015-2282), under the Disease Surveillance Plan, which was a joint federal-provincial Growing Forward 2 project. Financial support for Dr. Brochu’s stipend derived from the Ontario Veterinary College (OVC) DVSc fellowship fund. Summer students were supported through the OVC Andrea Leger Dunbar summer assistantship fund.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
