Abstract
Urinalysis is a rapid, simple, inexpensive, and reliable test that documents urine abnormalities reflecting various types of renal, hormonal, or metabolic diseases. Urinalysis could assist proper monitoring of the health of dolphins under human care; however, normal baseline values for dolphin urinalysis have not been reported, to our knowledge. We sampled urine from 193 common bottlenose dolphins (Tursiops truncatus), living under human care in 24 Caribbean dolphinariums, by voluntary free-catch and analyzed the urine for chemical and microscopic variables using multi-agent dry reagent chemistry dipstick test strips, dedicated pH reagent strips, and unstained sediment slides. Most urine was clear, pale yellow to dark yellow, and had a fishy odor. Dipstick glucose, bilirubin, ketones, and nitrites were negative in all dolphins. The urine pH was acidic (
Keywords
Introduction
The common bottlenose dolphin (Tursiops truncatus) is distributed worldwide in tropical and temperate waters. 18 It is one of the most studied marine mammals in the wild and under human care.7,22 Although it is the most common species used for public display in zoos and aquaria, and its veterinary care and clinical diagnosis are well documented,5,28 published reference values for urine parameters are limited except for an assessment of renin and aldosterone in urine 20 and values presented in a conference proceeding (Deming A, et al. Urinalysis in free-ranging bottlenose dolphins (Tursiops truncatus) from Sarasota Bay, Florida. Proc 43rd Ann Conf Int Assoc Aquatic Animal Med; 2012; Atlanta, GA. Available from: https://www.vin.com/apputil/content/defaultadv1.aspx?id=5377975&pid=11354). Urinalysis is commonly used in veterinary medicine as a clinical tool to monitor renal function and conditions.8,11,16,24,27 However, it is an underused, useful tool in marine mammal veterinary medicine that would allow assessment of early signs of renal and metabolic diseases that could occur without clinical signs.10,23 We report herein baseline urine values for normal chemical and microscopic variables for common bottlenose dolphins living under human care.
Materials and methods
Sample collection
Urine samples were collected at 24 aquaria throughout Mexico, the Caribbean, and one in Florida between January and July 2017. All dolphins were of Gulf of Mexico or Caribbean origin or born under human care from U.S., Mexican, Cuban, or Jamaican parents. Facilities in which dolphins were maintained were either tanks with artificial seawater (n = 8,
The samples were collected non-invasively by free catch through trained medical behaviors 26 prior to the dolphin’s first feeding, which consisted of capelin (Mallotus villosus), Atlantic herring (Clupea harengus), and supplements (Mazuri Vita-Zu mammal tablet or Bedoyecta multivitamins capsules, Bayer Equilibrium calcium, Bayer Equilibrium Ages, Culturelle probiotics, Siliprotec Natural tablets, and vitamins E, B complex, and B12) on an individual basis depending on the dolphin’s health and growth history. Dolphins at 7 sites were hydrated using 1–2 kg of gelatin/d; dolphins at 18 sites were hydrated when needed with 1–2 L of fresh water through trained voluntary intubation. The urine collection behavior included the following trained steps: call to station, rollover, maintenance of belly above the water level, drying of the genital slit, and massage above pubic area, which after a few minutes produced urination (Fig. 1). Because in some cases the sampling took several minutes, the trained behavior for urine sample included allowing the dolphin to roll from dorsal recumbency to a partial lateral recumbency position for a breath without compromising the cleaned and dried genital area.

Operant conditioning procedure for the medical behavior of urination used in this study. Steps included:
The 24 dolphin facilities in the study housed ~278 individuals at the time of sampling. Of these, dolphins included in our study were those >1 y-old, trained for voluntary urination, and previously assessed as healthy through physical examination, totaling 193 dolphins. Of these, 54.4% were females and 45.6% were males, 2–40 y old (
Samples were collected in a sterile polypropylene 120-mL container (Urine collection container; Globe Scientific, Mahwah, NJ) or with a 20-mL sterile plastic syringe. The collection container was labeled with the dolphin’s unique identifier, and the date and time of collection. Samples were stored in a hand-carry cooler with ice until transported back to the laboratory. At the laboratory, samples were stored in a refrigerator at 4–5°C until analyzed. Samples were analyzed 15–240 min after collection, with a mean of 57 min (± 28 min).
Urinalysis
The urinalysis consisted of 3 successive steps as described previously 29 : gross examination, chemical examination, and microscopic examination. Results were recorded on a marine mammal urinalysis form (Supplementary Data 1) and transferred to an Excel spreadsheet (Microsoft, Redmond, WA) by one author at the end of each sampling and analysis day. Possible transcription errors were checked between the urinalysis form and the spreadsheet prior to result analysis.
Gross examination
The volume of urine collected from each dolphin was measured with a Nalgene polymethylpentene plastic 50-mL graduated cylinder (Thermo Fisher Scientific, Waltham, MA). Physical characteristics of each sample were described, including color (colorless, pale yellow, yellow, dark yellow, dark brown–yellow, straw, ochre, red-brown, or red), appearance (clear, hazy, or slightly cloudy), and odor (none, urinoid [strong urine smell], fruity-sweet [sweet smell], fruity-fishy [mix of sweet and fish smell], fishy [fish smell], pungent [sharp or strong smell], sulfuric [rotten egg smell], aromatic [fragrant smell], fecal [smelling of feces], or ammoniacal [long-standing ammonia smell]). Given that smell to perceive and classify urine odors is subjective, evaluators practiced during clinical veterinary work with domestic species and other marine species (dolphins, sea lions, and manatees), and on one occasion by training at a human clinical laboratory.
Chemical examination
After gross examination, urine was transferred to a 15-mL conical sterile polypropylene Falcon centrifuge tube (Greiner Bio-One, Monroe, NC) for chemical examination using dry reagent dipstick strips. This was performed using 2 different urinalysis reagent strips (AimStrip 10-SG, Germain Laboratories, San Antonio, TX for glucose, bilirubin, ketones, nitrite, protein, blood, leukocytes, specificity gravity [SG], pH, and urobilinogen; URS-2M, CLIAwaived, San Diego, CA for creatinine and microalbumin). The reagent dipstick was introduced completely into the urine in the tube and removed immediately, the excess urine dried by placing the edge of the strip on a paper towel, and the results of color change compared to the color chart values on the urine strip bottle. We recorded the presence (±, +, ++, +++, ++++) or absence (negative) of glucose, bilirubin, ketone, nitrite, protein, blood, and leukocytes, and the value of SG, pH, and urobilinogen. Creatinine and microalbumin were measured in nominal values of 10, 30, 80, or 150 mg/L for microalbumin, and 0.1, 0.5, 1.0, 2.0, or 3.0 g/L for creatinine. Given that the values for pH and SG in the AimStrip-10 were found to be inaccurate during the first few samples analyzed, pH was also analyzed using a pH reagent strip (pH test strips; HealthyWiser, Santa Monica, CA), and SG was measured with a clinical veterinary refractometer (ATC REC-300ATC; Tekcoplus, Hong Kong) calibrated with distilled water.
Microscopic examination
The urine sample was centrifuged for 2.5 min at 1,163 × g in the urine tube (Clay Adams compact II centrifuge; BD Diagnostics, Franklin Lakes, NJ). The supernatant was discarded, and the pellet containing insoluble material was gently mixed with the remaining liquid (~0.5–1 mL) by pipetting. A small drop of resuspended sediment was placed on a glass microscope slide and coverslipped. The sediment was examined microscopically with an absolute microscope magnification of 100× and 400× in a minimum of 10 fields for white blood cells (WBC), red blood cells (RBC), epithelial cells (transitional, squamous, or tubular), casts (hyaline, granular, waxy, epithelial, or fatty), crystals (calcium oxalate dihydrate, uric acid, triple phosphates, calcium phosphate, amorphous phosphates, and urates), bacteria, yeast or fungi, spermatozoa, and parasites or parasite ova. The average number of elements (WBC, RBC, epithelial cells, etc.) in ten 400× fields was recorded and categorized as none present, rare (≤25% of HPF), few (26–49% of HPF), some (50–74% of HPF), or many (≥75% of HPF) present.
Statistical analysis
Results were analyzed with descriptive statistics and RStudio (https://www.rstudio.com/). Mean (
Results
Of the 215 animals from which the free-catch urination behavior was requested, 193 (89.8%) provided a urine sample within 5–10 min, which varied in volume 2–40 mL (14 ± 10).
Gross examination
Most urine samples were pale yellow or green-yellow, followed by yellow and pale green–yellow (Table 1). With the exception of 5 cases, urine was clear. The urine odor varied between fishy and fruity-fishy.
Free-catch urine gross findings in 193 common bottlenose dolphins under human care.

Chemical examination
All parameters except protein had predominantly negative results (Table 2). For protein, 32.9% of samples were positive, 15 were trace (0.0015 g/L), 31 were 1+ (0.003 g/L), and 17 were 2+ (1.0 g/L). The average refractometer SG (1.035 ± 0.008) did not differ by sex (p = 0.999) and was also not significantly different by age group (p = 0.989): 2–7 y-old (1.037 ± 0.008), 8–14 y-old (1.034 ± 0.008), 15–20 y-old (1.032 ± 0.007), and 21–40 y-old (1.036 ± 0.010). Urine SG measured by refractometer was not significantly different between habitats (p = 0.969): in tanks with artificial sea water (1.033 ± 0.008), in mangrove-surrounded enclosures (1.037 ± 0.009), or at sea pens (1.035 ± 0.008). Urinalysis reagent strip for SG was statistically lower 1.023 ± 0.007 than the refractometer (p < 0.050); however, this was no doubt an artifact because the reagent strip only measures between 1.000 and 1.030, and our findings indicate that dolphin urine is more concentrated (>1.030).
Free-catch urinalysis biochemical findings in 193 common bottlenose dolphins under human care.
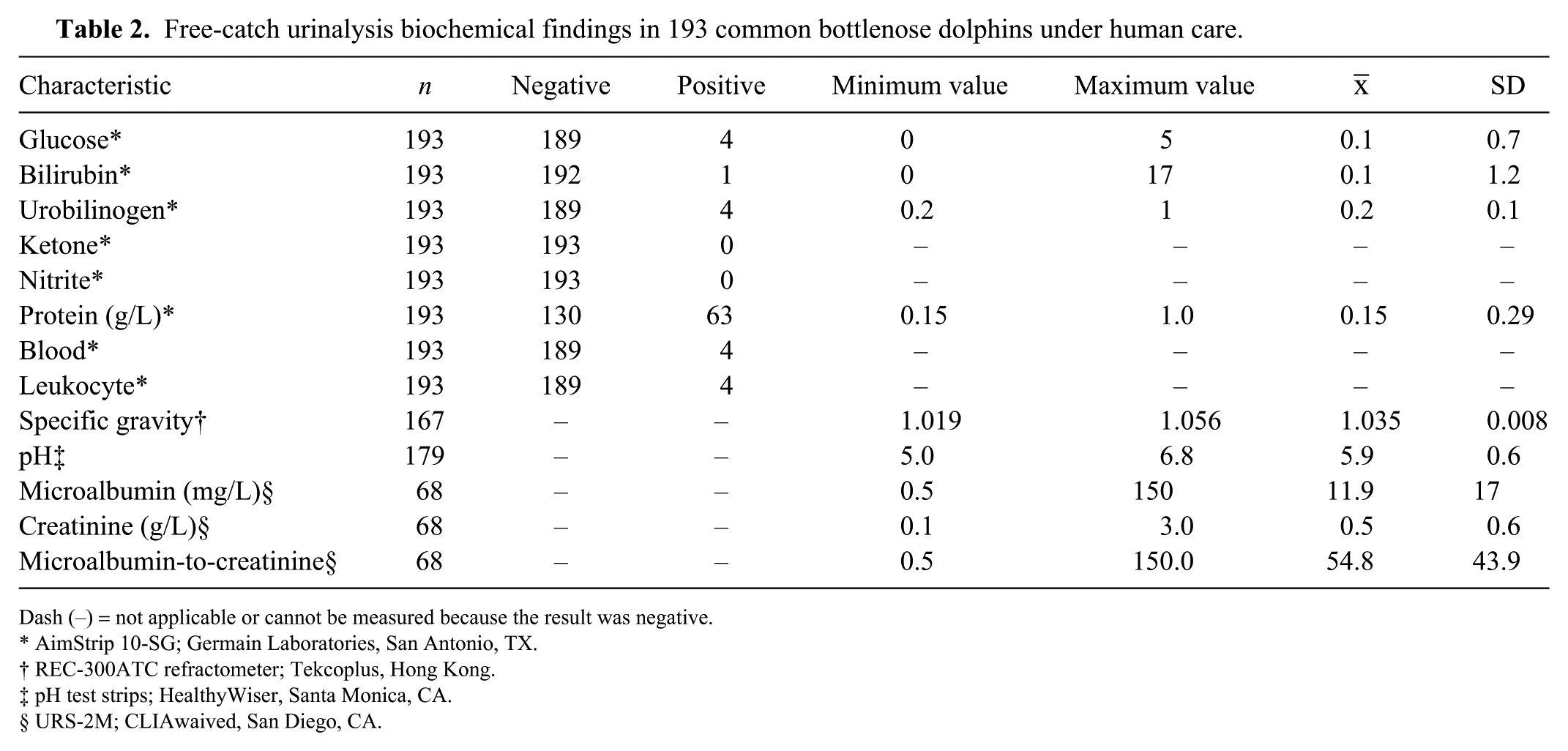
Dash (–) = not applicable or cannot be measured because the result was negative.
AimStrip 10-SG; Germain Laboratories, San Antonio, TX.
REC-300ATC refractometer; Tekcoplus, Hong Kong.
pH test strips; HealthyWiser, Santa Monica, CA.
URS-2M; CLIAwaived, San Diego, CA.
Urinalysis dipstick reagent strip pH had a higher average (6.2 ± 0.2) and was significantly different than with the pH-specific strip (p = 0.043), with the latter taken as the true value in our study. The average pH did not differ (p = 0.988) by sex, and was not significantly different by age group (p = 0.349): 2–7 y-old (5.8 ± 0.6), 8–14 y-old (5.9 ± 0.5), 15–20 y-old (6.1 ± 0.4), and 21–40 y-old (5.8 ± 0.6). Urine pH was significantly different between habitats evaluated (p = 0.047): in tanks with artificial seawater (5.9 ± 0.5), in mangrove-surrounded enclosures (5.7 ± 0.7), and at sea pens (6.0 ± 0.6).
The microalbumin-to-creatinine range was 20–100 mg/g in most of the samples (79.4%). There was no correlation between samples positive for microalbumin and those positive for protein.
Microscopic examination
WBCs and RBCs were present only in low numbers (Tables 3, 4). Epithelial cells were observed in half of the samples (55.7%), and all were squamous. Crystals were observed in 36.6% of the samples. The most common crystals were calcium oxalate dihydrate (48.2%) and amorphous urates (42.4%), with a few cases of struvite (4.7%), calcium oxalate monohydrate (3.5%), and cysteine (1.2%) crystals. Bacteria and casts were rarely found; all casts were hyaline. Sperm were only seen in one male sampled. Parasite ova and fungal elements were not found in any of the 193 urine samples.
Free-catch urinalysis microscopic sediment findings (per 400× field) in 193 common bottlenose dolphins under human care.
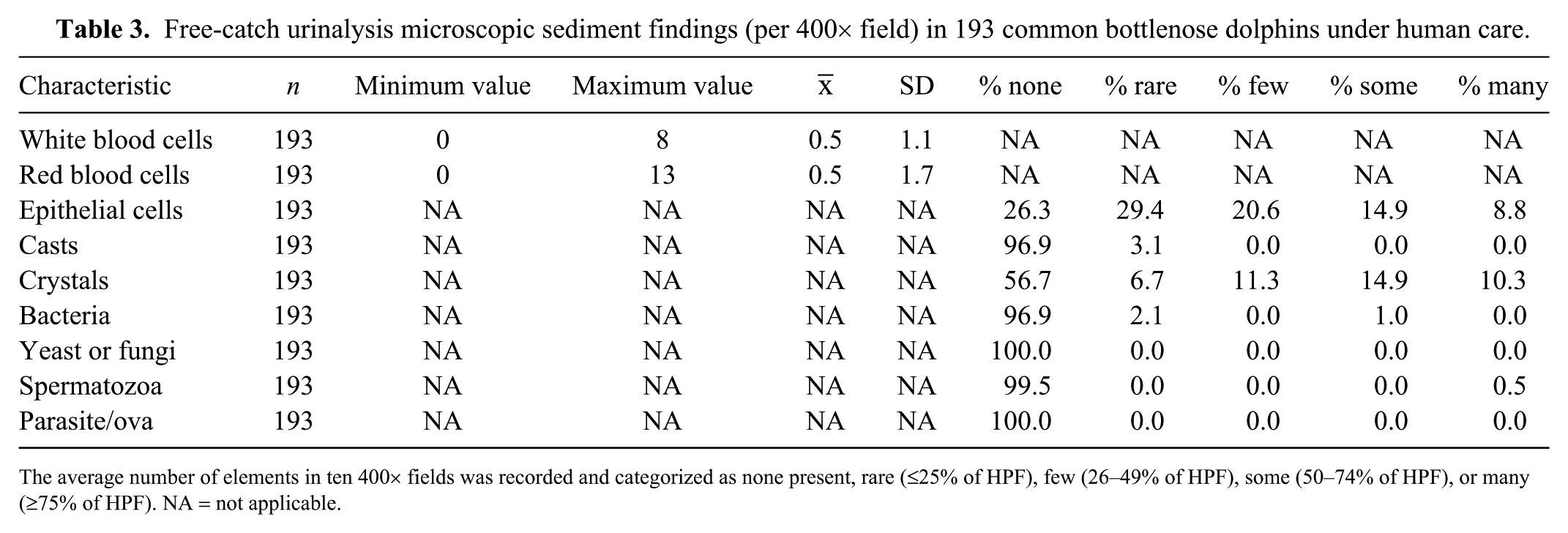
The average number of elements in ten 400× fields was recorded and categorized as none present, rare (≤25% of HPF), few (26–49% of HPF), some (50–74% of HPF), or many (≥75% of HPF). NA = not applicable.
Urinalysis reference values established in 193 common bottlenose dolphins under human care.

Discussion
The use of trained medical behaviors for obtaining samples for clinical diagnosis has been useful in the husbandry, management, and care of zoo and aquarium animals. 26 These behaviors initially focused on blood collection, but the technique has been expanded to include imaging, sperm collection, and treatments, among others. The collection of urine under operant conditioning, as in our study, appears to be an effective, non-invasive, non-stressful method that could be included in the physical examination of dolphins under human care to help early diagnosis of medical conditions, reducing the need for cystocentesis or urinary catheterization.
In most animals, urine is normally clear, and normal urine color is typically yellow or amber depending primarily on the concentration of urochromes and urobilin. The color intensity is influenced by the volume of urine collected, SG, and concentration of the urine. 3 Urine color can change because of a disease process or dietary supplementation, including food or medications.8,23 Although 60% of urines in our study were pale to dark yellow, 40% of the samples were pale to dark green–yellow. This green coloration may be caused by biliverdin, a tetrapyrrolic bile pigment, present in the blue-green blood of many species of marine fish, including the ones being fed to the dolphins in our study. 13
The turbidity of urine varies between and within wildlife and domestic species, but is generally clear, although urine may be turbid in horses and cattle. 3 The appearance of urine in healthy animals may change with the presence of pigments, crystals, hemoglobin, white blood cells, or lipids. However, cloudy urine is not necessarily indicative of disease.8,23 Sediment examination is required to evaluate cellular elements for potential impact on turbidity. 11 In the dolphins studied, almost all had clear urine, except a few cases in which urine sediments and spermatozoa made the sample hazy or slightly cloudy.
Urine odor depends on urine concentration and diet. Bacterial infections may create strong odors as a result of pyuria, or strong ammonia odor when bacteria produce urease. 3 Dolphin urine had a strong fish odor, at times combined with a fruity smell, directly related to the animals’ diet (nonpathogenic trimethylaminuria) and dietary supplements, 1 even though samples were taken prior to the dolphin’s first morning feeding and supplementation.
The results obtained for glucose, bilirubin, urobilinogen, ketone, nitrite blood, and leukocyte with the dipstick were negative in most of the samples. However, proteinuria was observed in 28.9% of the dolphins sampled without signs of proteinuria-related disease (e.g., glomerulonephritis, diabetes, urinary infection, congestive heart failure).
The SG of urine of the dolphins in our study did not vary significantly based on habitat salinity. However, SG is influenced not only by water consumption and electrolyte balance but also by many other parameters such as dietary protein source and quantity.16,23 Given that dolphins are piscivores with a high protein intake, and their freshwater source comes from their prey, their SG is expected to be high (1.035 ± 0.008) in comparison to other mammals. Dolphins, like other marine mammals, have the ability to concentrate urine because of their reniculate kidneys. 11 Our results are similar to those reported for other artiodactyls (dolphins are cetaceans members of the order Cetartiodactyla), including mule deer (1.036 ± 0.006), 12 and sheep, goats, and cattle (1.035 ± 0.009). 23
We measured dolphin urine pH with a specific pH strip, rather than from the multi-reagent urinalysis dipstick. Results were shown to be significantly higher when using the urinalysis dipstick than when using the specific pH strip. In addition, the range used in the urinalysis dipstick is 5.0–8.5 (compared to a wider range in the pH-specific strip of 4.5–9.0). Urine pH is an estimate of the body’s acid-base status, which in dolphins is partially affected by the salinity of the habitat, but is highly variable and greatly affected by diet, bacterial infections, storage time, and urinary retention. 23 Carnivores, in our case piscivores, have diets high in protein that produces acid-to-neutral urine, contrary to herbivores, which produce alkaline urine.8,11 Changes in pH towards alkalinity in carnivores and piscivores may indicate urinary tract disease, obstruction, or renal tubular disease. 23 Urine pH may also be associated with the formation of crystals and nephrolithiasis.32,33 Variation in urine pH between the different habitats (marine vs. estuarine) may be the result of accidental ingestion of water of different salinities and its effect on the renal system, as observed in the urine pH of our dolphins in marine versus estuarine enclosures. Variation in pH may also arise from using different pH reading strips, as our mean urinary pH taken with the urinalysis dipstick was 6.2 ± 0.3, whereas it was 5.9 ± 0.6 when measured with the pH-specific reagent strip. The pH-specific indicator strips may be more reliable given that their scale varies by 0.25 units in comparison with the urinalysis dipstick with a scale graduation of 0.5 units, and would be more precise if they were equally accurate. The pH range obtained in our dolphins is consistent with previously reported values for free-ranging common bottlenose dolphins, killer whales under human care, and other marine mammals (Renner MS. Urinary indices in the captive killer whale (Orcinus orca). Proc 33rd Annual Conf Int Assoc Aquatic Animal Med, 2002; Albufeira, Portugal. Available from: https://www.vin.com/apputil/content/defaultadv1.aspx?id=3864796&pid=11257&print=1; Deming A, et al. 2012). 5
Microalbuminuria is an indicator of glomerular damage. 23 In some cases, trace proteinuria by dipstick test may be an indicator of microalbuminuria, 19 but in the dolphins we observed with proteinuria, the microalbumin was found to be within normal RIs. Creatinine in urine is influenced by dietary factors such as protein intake and the muscle mass of the animal. 9 The RIs of creatinine in the urine of our dolphins (0.1–0.5 g/L) is consistent with those for cattle (0.54–1.75 g/L), sheep (0.04–0.22 g/L), goats (0.45–0.77 g/L), 6 and harbor seals (0.17–0.74 g/L). 30
Although the microalbumin-to-creatinine ratio has been strongly correlated with total protein excretion, 17 this was not the case in the dolphins we studied. Normal values for microalbumin-to-creatinine in humans and dogs are <30 mg/g2,19,25,35; in contrast, normal values in common bottlenose dolphins in our study were 20–100 mg/g.
Urine of bottlenose dolphins in our study contained 0–2 WBCs/HPF and 0–2 RBCs/HPF. Urines with >2 WBC or 2 RBC per HPF could be associated with the presence of renal stones or genital tract contamination (Deming A, et al. 2012). Squamous epithelial cells as found in our dolphin samples are more common in urine samples taken by free-catch, 8 and do not appear to be clinically significant and may be attributable to normal shedding within the urogenital tract.11,34
Casts were not found or rarely found in the urine of the dolphins we studied, which is considered to be normal in most animal species. 8 Calcium oxalate and struvite crystals have been reported in bottlenose dolphins and other cetaceans.21,32,33 The crystals in our samples were predominantly calcium oxalate, which occurs in neutral to acid urine.8,23 However, the healthy dolphins sampled that had a previous medical history of urolithiasis did not have crystals in the analyzed sediment, although crystalluria and uroliths are not always present concurrently. 31 Crystalluria may occur in dolphins because of their high dietary protein and supersaturation of urea.
In our dolphin urine samples, the rare presence of bacteria in a few of the samples (3.1%) was attributed to contamination. Similarly, spermatozoa may be observed on rare occasions, 8 and in our case, in one sexually active and alpha male dolphin in a particular facility. Parasites, their ova, and fungi or yeast are rarely, if at all, observed in urine samples unless there is some underlying disease. 8 Dolphin urine samples in our study did not harbor parasites, ova, fungi, or yeast.
Supplemental Material
DS1_JVDI_10.1177_1040638719839110 – Supplemental material for Baseline urinalysis values in common bottlenose dolphins under human care in the Caribbean
Supplemental material, DS1_JVDI_10.1177_1040638719839110 for Baseline urinalysis values in common bottlenose dolphins under human care in the Caribbean by Natalia Jiménez-Zucchet, Tamara Alejandro-Zayas, Christian A. Alvarado-Macedo, María Renée Arreola-Illescas, Lissette Benítez-Araiza, Lilian Bustamante-Tello, Danilo Cruz-Martínes, Nayeli Falcón-Robles, Luz Garduño-González, María Concepción López-Romahn, Ana Michelle Martínez-Taylor, Adriana Mingramm-Murillo, Carlos Ortíz, Antonio L. Rivera-Guzmán, Rocío Sabater-Durán, Angélica Sánchez-Jiménez, Roberto Sánchez-Okrucky, Lydia Staggs, Raúl Torres-Salcedo, María Vences-Fernández, Bert Rivera-Marchand and Antonio A. Mignucci-Giannoni in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Jonathan Cowart and Irma Méndez for manuscript review. The assistance from colleagues and students from PRMCC in the field and laboratory, especially Lorna Alvarado is greatly appreciated. Thanks to the trainers of Dolphin Discovery’s Anguilla, Costa Maya, Cove Cayman, Cozumel, Cayman, Dolphin Cove Montego Bay, Dolphin Cove Ocho Rios, Dreams, Gulf World, Isla Mujeres, Moon Palace, Playa del Carmen, Punta Cana, Riviera Maya, Saint Kitts, Six Flags, Tortola, and Tulum-Akumal, and Delphinus’ Acuario Interactivo, Puerto Morelos, Punta Cancun, Riviera Maya, Xcaret, and Xel-ha, for assistance in urine sample collection.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was conducted with financial support from the Undergraduate Research Program of the Inter American University of Puerto Rico, and the Puerto Rico Manatee Conservation Center.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
