Abstract
A recently deceased juvenile male bottlenose dolphin (Tursiops truncatus) was found floating in the Gulf of Mexico, off Sand Key in Clearwater, Florida. At autopsy, we identified pneumonia and a focus of malacia in the right cerebrum. Cytologic evaluation of tissue imprints from the right cerebrum revealed fungal hyphae. Fungal cultures of the lung and brain yielded Aspergillus fumigatus, which was confirmed by amplification of a portion of the fungal nuclear ribosomal internal transcribed spacer 2 region sequence. Microscopic pulmonary lesions of bronchiolar epithelial cell syncytia with intracytoplasmic and intranuclear inclusions within bronchiolar epithelial cells were suggestive of Cetacean morbillivirus (CeMV) infection. The occurrence of CeMV infection was supported by positive immunohistochemical staining for morbillivirus antigen. CeMV detection was confirmed by amplification and sequencing a portion of the morbilliviral RNA-dependent RNA polymerase gene from lung tissue. This case provides CeMV sequence data available from the Gulf of Mexico and underscores the need for genomic sequencing across diverse host, temporospatial, and population (i.e., single animal vs. mass mortality events) scales to improve our understanding of these globally emerging pathogens.
Keywords
The most commonly reported mycotic infection in marine mammals is pulmonary aspergillosis. 21 Aspergillosis is caused by the genus Aspergillus, an ascomycete fungus with a worldwide distribution. Aspergillus spp. are environmentally abundant opportunistic fungal pathogens, with Aspergillus fumigatus the most common species infecting animals. 7 The respiratory tract is usually the primary site of infection. 16 Furthermore, fatal infections should raise suspicions of immunosuppression within the individual animal.2,16 Aspergillus fumigatus has been implicated in both pulmonary and central nervous system disease in cetaceans and may occur in both managed and free-ranging bottlenose dolphin (Tursiops truncatus) populations.8,9,20 Even though A. fumigatus infections alone can cause mortality, another important pathogen of cetaceans, Cetacean morbillivirus, may be found in a coinfection with aspergillosis.10,17,24
Members of genus Morbillivirus, in family Paramyxoviridae, include Canine distemper virus, Cetacean morbillivirus (CeMV), Feline morbillivirus, Measles virus (type species), Peste-des-petits ruminants virus, Phocine distemper virus, and Rinderpest virus.30,32 CeMV contains at least 4 northern hemisphere strains: pilot whale morbillivirus, porpoise morbillivirus, dolphin morbillivirus (DMV), and beaked whale morbillivirus,3,12,27,30,31 and at least 2 highly divergent southern hemisphere strains identified in Brazil and Western Australia in 2014.11,26
Clinical findings in cetaceans with CeMV infection include poor body condition, erosive and ulcerative skin lesions, mucosal erosions, tachypnea, abnormal respiratory patterns, tremors, self-mutilation, and abnormal behavior. 12 Dolphins infected with CeMV often strand and may occur as single 17 or mass mortality epizootic events. Mass mortality epizootics in bottlenose dolphins have occurred in 1987–1988 and 1993–1994 along the Atlantic 19 and Gulf 18 coasts of the United States, respectively, and in striped dolphins (Stenella coeruleoalba) in the Mediterranean in 1990–1992, and again in 2007. 5 In at least 3 cases in striped dolphins, necrohemorrhagic pneumonia and encephalitis associated with A. fumigatus were observed at postmortem examination. 10 The 1987–1988 mortality event contributed to the passage of Title IV of the Marine Mammal Protection Act, which created a system to respond to and investigate Unusual Mortality Events (UME) in marine mammals. One such UME involving DMV deaths in bottlenose dolphins was declared along the Atlantic coast of the United States in 2013–2014 (Fauquier D, et al. Update on the dolphin morbillivirus outbreak and the 2013–2014 Mid-Atlantic bottlenose dolphin (Tursiops truncatus) unusual mortality event. In: Proceedings of 2014 International Whaling Commission Scientific Committee annual meeting, 15–18 Sept 2014, Portoroz, Slovenia).
Our report describes the postmortem examination of a freshly deceased Gulf of Mexico bottlenose dolphin and diagnosis of a disseminated coinfection of A. fumigatus and dolphin morbillivirus based on culture, cytologic, histopathologic, immunohistochemical, and molecular findings. The body of a juvenile male bottlenose dolphin of unknown age was recovered from waters in the Gulf of Mexico, just off Sand Key near Clearwater, Florida in May 2012. The freshly dead animal was classified as a Code 2 23 and placed on ice in sternal position for transport to the University of Florida for postmortem examination. The body had mild autolysis. The 85-kg carcass had a standard length (SL) of 171 cm and a girth at the umbilicus of 84 cm. Body condition score was assessed as 2 of 5. The external blubber thickness was 8 mm on the dorsal cut surface. Based on the SL, wear on the teeth, and overall condition, the animal was judged to be a juvenile male.
The animal was thin, with minimal internal fat deposits. Serosanguineous effusions were noted surrounding the left lung (~100 mL) and within the pericardial sac (~20 mL). The partly collapsed lungs were edematous, with localized areas of emphysema under the red to dark-purple pleura; there were patchy, widely scattered areas of moderate to severe consolidation. White-to-tan caseous material was noted at the bifurcation of the trachea, where the mucosal lining was thickened and erythematous (Fig. 1A). All abdominal organs appeared grossly normal.
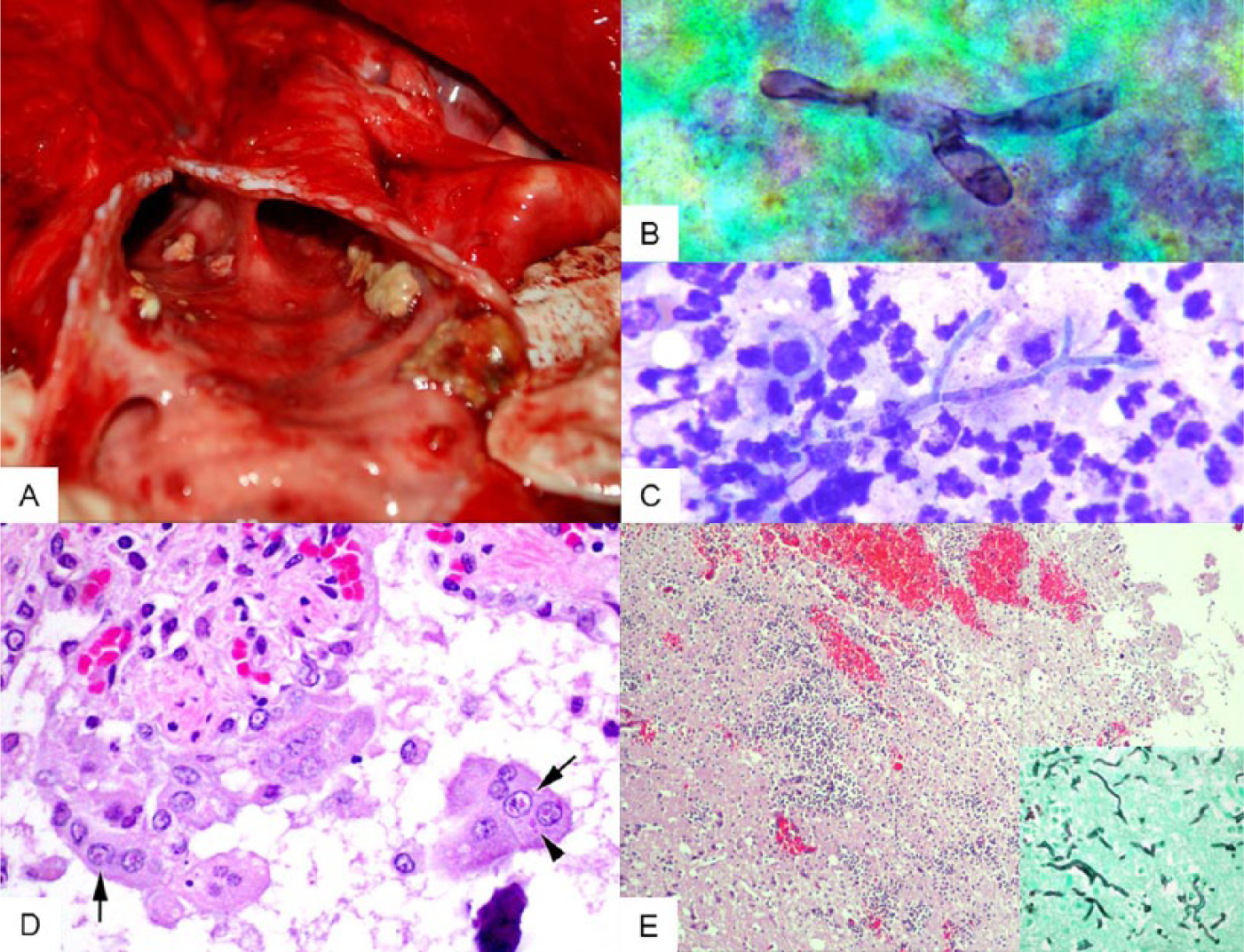
All tissues are from a juvenile male bottlenose dolphin (Tursiops truncatus).
The right cerebral hemisphere was swollen and erythematous, with viscous yellow material oozing from its caudal aspect. Impression smears and cultures were obtained from this site, and the entire brain was preserved in 10% neutral buffered formalin for histologic examination. The pterygoid sinuses had red, swollen mucosal surfaces that were partially covered by viscous yellow material.
Tissues were collected from multiple organs and frozen (−80°C and −20°C). 23 Samples from the brain, lungs, pterygoid sinuses, and thoracic fluid were processed for standard aerobic and anaerobic bacterial and fungal culture and identification. Impression smears of the right cerebrum, pleural and pericardial effusions, and of the white-to-tan caseous material from the tracheal bifurcation and fluid from the right pterygoid sinus were stained with Wright–Giemsa for cytologic evaluation. One slide of the impression smear of the brain was stained with Gomori methenamine silver (GMS) stain. Sections of lungs, urinary bladder, small intestine, kidneys, spleen, pancreas, dura, and brain were collected and preserved in 10% neutral-buffered formalin solution, processed routinely, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin; selected sections were stained with Brown and Brenn and GMS stains.
The impression smears of the right cerebrum revealed suppurative inflammation with occasional branching, septate fungal hyphae (Fig. 1B, 1C). No bacteria, parasites, or neoplastic cells were noted. The pleural fluid, reported as light red and slightly hazy, was consistent with a high-protein modified transudate with total solids (TS) of 0.42 g/L (4.2 g/dL) and a white blood cell estimate of <0.5 × 109/L (500/µL) consisting of 100% mononuclear phagocytes. The pericardial fluid was also a high-protein modified transudate and contained <0.5 × 109 white blood cells/L with 100% mononuclear phagocytes, but had slightly higher TS at 0.52 g/L (5.2 g/dL). Cytologic examination of the material identified at the bifurcation of the trachea revealed suppurative inflammation and numerous small bacilliform bacteria. Finally, material from the right pterygoid sinus exhibited suppurative sinusitis with mixed bacilli and respiratory epithelial cell hyperplasia.
Histologically, the airways contained macrophages, syncytial cells, and sloughed, necrotic epithelia. The bronchiolar epithelium was sometimes sloughed, attenuated, or disorganized. A number of epithelial cells and syncytial cells contained occasional eosinophilic nuclear and cytoplasmic inclusion bodies, with variable nuclear chromatin clearing (Fig. 1D). The adjacent interstitium contained few to moderate numbers of lymphocytes, plasma cells, and macrophages, with fewer neutrophils and eosinophils. A few small bronchioles contained luminal pyogranulomatous inflammation, frequently effacing the epithelium and infiltrating the surrounding interstitium and alveoli. Within the inflammatory foci centered on the small airways were small aggregates of cellular debris admixed with small to moderate amounts of mucin, and occasional fungal hyphae. The hyphae were ~6 µm in diameter, septate, and parallel-walled with acute-angle, dichotomous branching, consistent with members of genus Aspergillus. Hyphae within the lumina of the affected airways multifocally extended into the bronchiolar lamina propria and associated cartilage. Rarely, there were small to medium-sized airways filled with cellular debris and largely mineralized degenerate metazoans (presumptively lungworms) of 0.5–2 mm diameter.
Within both the gray and white matter in the cerebral cortex, thalamus, and cerebellum, there were multiple foci of inflammation, hemorrhage, and necrosis, with rarefaction of the parenchyma. The inflammatory infiltrate was composed of numerous neutrophils, a few macrophages, and low numbers of fungal hyphae similar to those described in the lung (Fig. 1E). Inflammation was sometimes centered on blood vessels, and affected vessels often had fibrinoid necrosis of the walls with transmural infiltration of neutrophils and low-to-moderate numbers of fungal hyphae.
Immunohistochemical staining for morbilliviral antigen was conducted in formalin-fixed, paraffin-embedded tissue sections of the lung. A monoclonal antibody to Canine distemper virus, known to be cross-reactive with other morbilliviruses, was used at a dilution of 1:1,000, according to a previously published protocol. 13 Positive immunohistochemical staining was observed prominently in bronchiolar epithelium and syncytial cells (Fig. 2A–C), as well as in fibroblasts (Fig. 2D). A variable intensity of immunohistochemical staining was observed in macrophages.
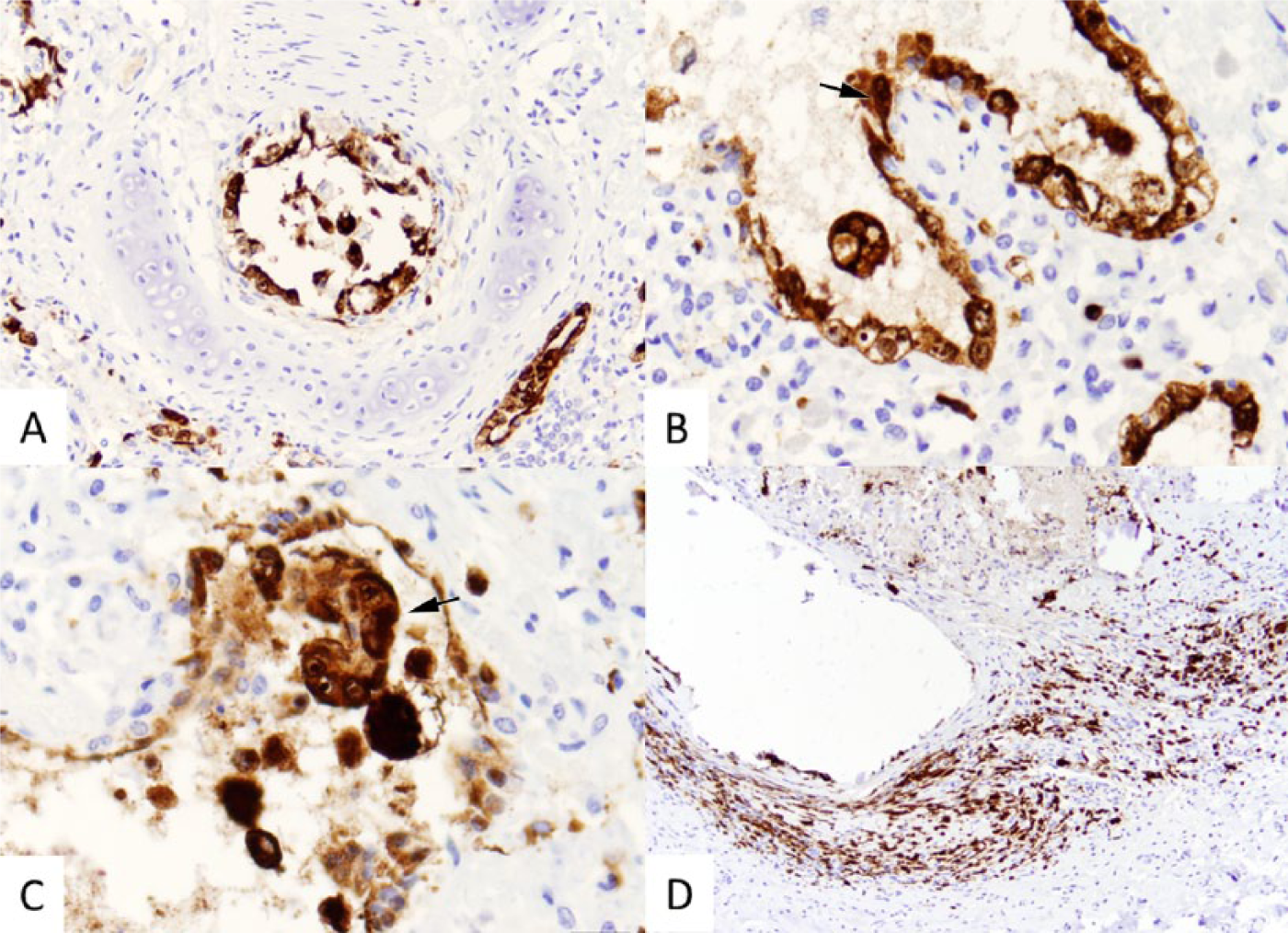
Lung from a juvenile male bottlenose dolphin (Tursiops truncatus), immunohistochemical (IHC) staining for morbillivirus antigen.
The DNA was extracted from frozen lung tissue using an automated extractor. a Amplification of the fungal nuclear ribosomal internal transcribed spacer 2 region (ITS2) was attempted on lung tissue DNA using primers ITS4 and ITS86, as described previously. 29 Total RNA was extracted from frozen lung tissue using the same extractor. a The RNA was subjected to a paramyxoviral reverse transcription polymerase chain reaction (PCR) assay that targets a portion of the viral RNA-dependent RNA polymerase using primers RES-MOR-HEN-F1 and RES-MOR-HEN-R, as described previously. 28 After electrophoresis in 1% agarose gels, bands were extracted and purified using a gel extraction kit. b Purified DNA samples were submitted to the University of Florida Interdisciplinary Center for Biotechnology Research (Gainesville, Florida) for Sanger sequencing in both directions on an automated sequencer. c Sequences were aligned, and primer sequences were removed. BLASTN searches (www.ncbi.nlm.nih.gov/blast/Blast.cgi) of the edited sequences were performed. 1
Aspergillus fumigatus was cultured out of the right pterygoid sinus, left lung, trachea, and brain, and the identity was confirmed by ITS2 PCR and sequencing. The sequence obtained from the paramyxovirus PCR was found to have a 99.1% sequence identity (554/559 nt) to a Mediterranean striped dolphin DMV isolate (reference sequence NC_005283). The Gulf DMV sequence was submitted to GenBank under accession KT964777. The sequence obtained from fungal PCR of lung was found to have 100% sequence identity (257/257 nt) to numerous A. fumigatus entries within GenBank (e.g., accession LN849893). The fungal sequence has been deposited in GenBank under accession KT625585. Vibrio alginolyticus was also isolated from the brain, left lung, right lung, trachea, and sinus; however, there was no histologic support for antemortem bacterial sepsis.
The fact that a single animal was affected and no mass mortality event occurred might be explained by the animal being immunocompromised, with the morbillivirus infection and/or the aspergillosis occurring secondary to this compromise.15,17 Morbilliviruses are recognized as causes of immune compromise in multiple species. DMV-infected dolphins often display lymph node necrosis, leading to lymphoid depletion and subsequent susceptibility to secondary infection. 12 Dolphins with serologic evidence of DMV showed decreased T lymphocyte proliferative responses. 6 In other mammals, morbilliviruses have also been shown to upregulate interleukin (IL)-4 and IL-10, causing a shift away from cellular immunity toward a humoral immune response. 14
The combination of fungal cultures from the brain, sinuses, and respiratory tract, together with the identification of lesions consistent with fungal etiology on cytology and histology, suggest that A. fumigatus was a contributing factor in the demise of this animal. One important virulence factor expressed by A. fumigatus is gliotoxin. Gliotoxin causes immunosuppression by inhibiting the transcription factor NF-κB protein complex, which regulates the immune response to cellular stimuli. 4 Although infectious disease is often studied as a single agent causing disease, this reductionist view has limited real world application; infection with multiple agents is often synergistic in causing disease. 25 Our study supports the idea that investigations should not cease at the identification of a single agent.
To our knowledge, this case is the first formal report of CeMV in the Gulf coast of Florida in nearly 2 decades and the only sequence data available for a CeMV strain from the region. Despite their significance as globally emerging pathogens of cetaceans, the molecular epidemiology of CeMVs is poorly understood. Only 1 complete genome, from a striped dolphin from the Mediterranean, is in GenBank at the time of this writing. 22 The fact that our Gulf CeMV strain exhibited >99% sequence identity to the Mediterranean CeMV that has not been detected and/or reported since 1993 could be significant. This northern hemisphere CeMV strain may have remained unchanged in the environment for decades, or this could be a reemergence of a potentially devastating disease for bottlenose dolphins. However, caution in interpreting these data is warranted until the full genome of this Gulf CeMV can be compared with other northern (e.g., Gulf, north Atlantic, Hawaiian, and Mediterranean) and southern (e.g., south Atlantic and Pacific) hemisphere CeMVs. Future studies investigating the genomic sequences of CeMV strains across diverse host, temporospatial, and population scales are needed to improve our understanding of these globally emerging pathogens.
Footnotes
Acknowledgements
We thank the members of the National Marine Fisheries Service Marine Mammal Health and Stranding Response Network (Southeast Region) and the Clearwater Marine Aquarium Stranding Team who recovered and transported the animal to the the University of Florida.
Authors’ contributions
SE Cassle and TB Waltzek contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. N Landrau-Giovannetti and A Leone contributed to acquisition and analysis of data. LL Farina, JFX Wellehan, NI Stacy, U Blas-Machado, JT Saliki, and MT Walsh contributed to acquisition, analysis, and interpretation of data. P Thompson, H Herring, and B Mase-Guthrie contributed to acquisition of data. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Maxwell 16 automated extractor, Promega Corp., Milwaukee, WI.
b.
QIAquick gel extraction kit, Qiagen Inc., Valencia, CA.
c.
ABI 3130 DNA sequencer, Life Technologies, Grand Island, NY.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by Grant No. NA14NMF46090276 awarded by the National Oceanic and Atmospheric Administration.
