Abstract
A 5-y-old female bottlenose dolphin (Tursiops truncatus) from an aquarium in Japan had clinical signs of anorexia, vomiting, and bradykinesia. Enrofloxacin and lactated Ringer solution were administered for treatment of bacterial infection and for rehydration. Elevations of creatine kinase and aspartate aminotransferase activities were detected on day 4 of treatment, indicating that rhabdomyolysis had developed on day 3. On day 5, serum creatinine and urea concentrations increased and remained high throughout the remaining treatment; the dolphin died on day 16. Postmortem examination revealed massive necrosis of the longissimus dorsi muscles. Histologic examination revealed extensive necrosis of skeletal myofibers, multifocal renal tubular necrosis with intratubular casts and crystals, and suppurative bronchopneumonia. The renal casts labeled positively with anti-myoglobin antibody; expression of aquaporin-1 was decreased in renal tubules compared to normal kidney tissue. To our knowledge, this description of clinicopathologic findings of rhabdomyolysis leading to acute kidney injury with concomitant crystalline nephropathy has not been reported previously in a bottlenose dolphin.
Rhabdomyolysis is characterized by breakdown of skeletal muscle fibers, resulting in the release of intracellular components into the blood. Rhabdomyolysis can lead to severe systemic complications such as hypotension, disseminated intravascular coagulopathy, electrolyte imbalances, cardiac arrhythmia, myoglobinuria, and acute kidney injury (AKI). 3 AKI resulting from rhabdomyolysis has been documented in humans and in a wide range of terrestrial animals; AKI occurred in >45% of human patients diagnosed with rhabdomyolysis.6,18,20 Among cetaceans, capture myopathy has been reported to include the histopathologic changes of degeneration and contraction band necrosis of cardiomyocytes and segmental necrosis of skeletal muscle.4,14 Similarly, live stranding of cetaceans causes severe physiologic stress that can lead to myonecrosis and associated myoglobinuric nephrosis.2,15 We describe here the histopathologic changes and immunohistochemical features of rhabdomyolysis associated with myoglobinuric nephrosis and crystalline nephropathy in a bottlenose dolphin that developed AKI during medication.
A 5-y-old female bottlenose dolphin (Tursiops truncatus) born and raised in an aquarium in Japan had initial clinical signs of anorexia, vomiting, and bradykinesia. Vaginal mucus discharge was observed on day 2 and used for bacterial culture; reproductive system infection was suspected. The staff veterinarian initially provided supportive treatment, including antibiotics, antiemetics, and digestive supplements. Blood was collected daily from dorsal fin or tail fluke blood vessels for hematology and biochemical analysis (XE-5000, Sysmex; Labospect 008, Hitachi). On day 3, the dolphin exhibited signs of shock (labored breathing, balance instability) ~9 min after medication while water was refilling the medical pool. Respiratory distress was then observed, along with dark-red urine, and myoglobinuria was suspected.
Bacterial culture and antimicrobial susceptibility tests (resistant to ampicillin, amoxicillin, sultamicillin, cefazolin, kanamycin, ofloxacin, tosufloxacin, levofloxacin) confirmed infection with a multidrug-resistant Escherichia coli. Amoxicillin–clavulanate (5 mg/kg, PO, q8h, days 1–3), ampicillin sulbactam (50 mg/kg, IV, q24h, day 4), enrofloxacin (5 mg/kg, IM, q24h, days 4–7), metronidazole (5 mg/kg, PO, q12h, days 8–11), imipenem (0.5–1 mg/kg, IM, q12h, days 10–14), and doxycycline (2 mg/kg, PO, q12h, days 15, 16) were administered. In addition to these antimicrobial agents, fluid therapy including lactated Ringer solution and 5% glucose solution, and supportive treatments including administration of furosemide, were conducted for AKI treatment from day 4. The dolphin died after 16 d of treatment.
Serum aspartate aminotransferase (AST) and creatine kinase (CK) activities, and urea and creatinine concentrations, increased gradually from day 1 to day 3. Marked increases in most of these parameters were observed from day 4 onwards. Most notably, CK rose to more than 7 times the baseline concentration. The serum creatinine concentration increased to >1.5 times baseline with progressively increased urea (average concentration ≥2.6 times baseline) throughout the treatment from day 5 to day 13. On day 14, all of these values became extremely elevated compared to reported RIs for the bottlenose dolphin (Table 1). 12 In addition, serum electrolyte abnormalities resulting from AKI, and leukocytosis, continued until the dolphin died.
Abnormal serum biochemistry values in a bottlenose dolphin with rhabdomyolysis, myoglobinuric nephrosis, and acute kidney injury.
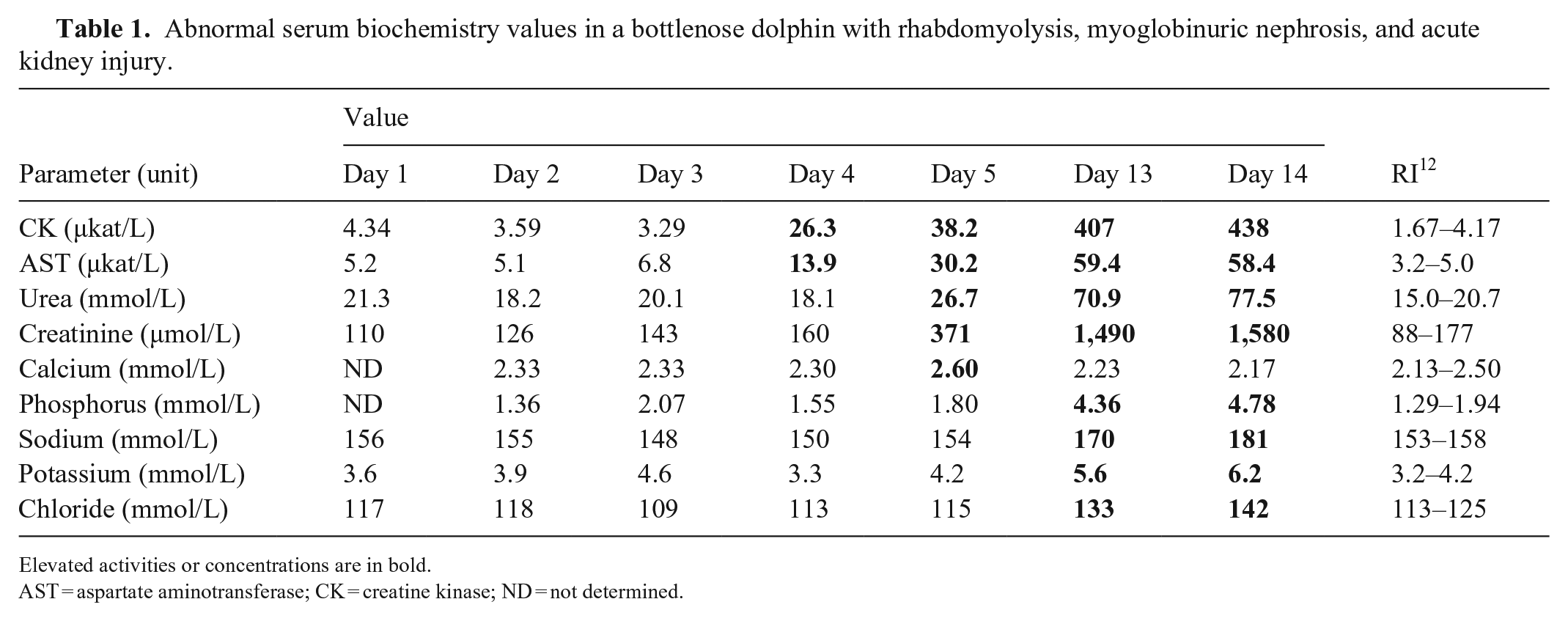
Elevated activities or concentrations are in bold.
AST = aspartate aminotransferase; CK = creatine kinase; ND = not determined.
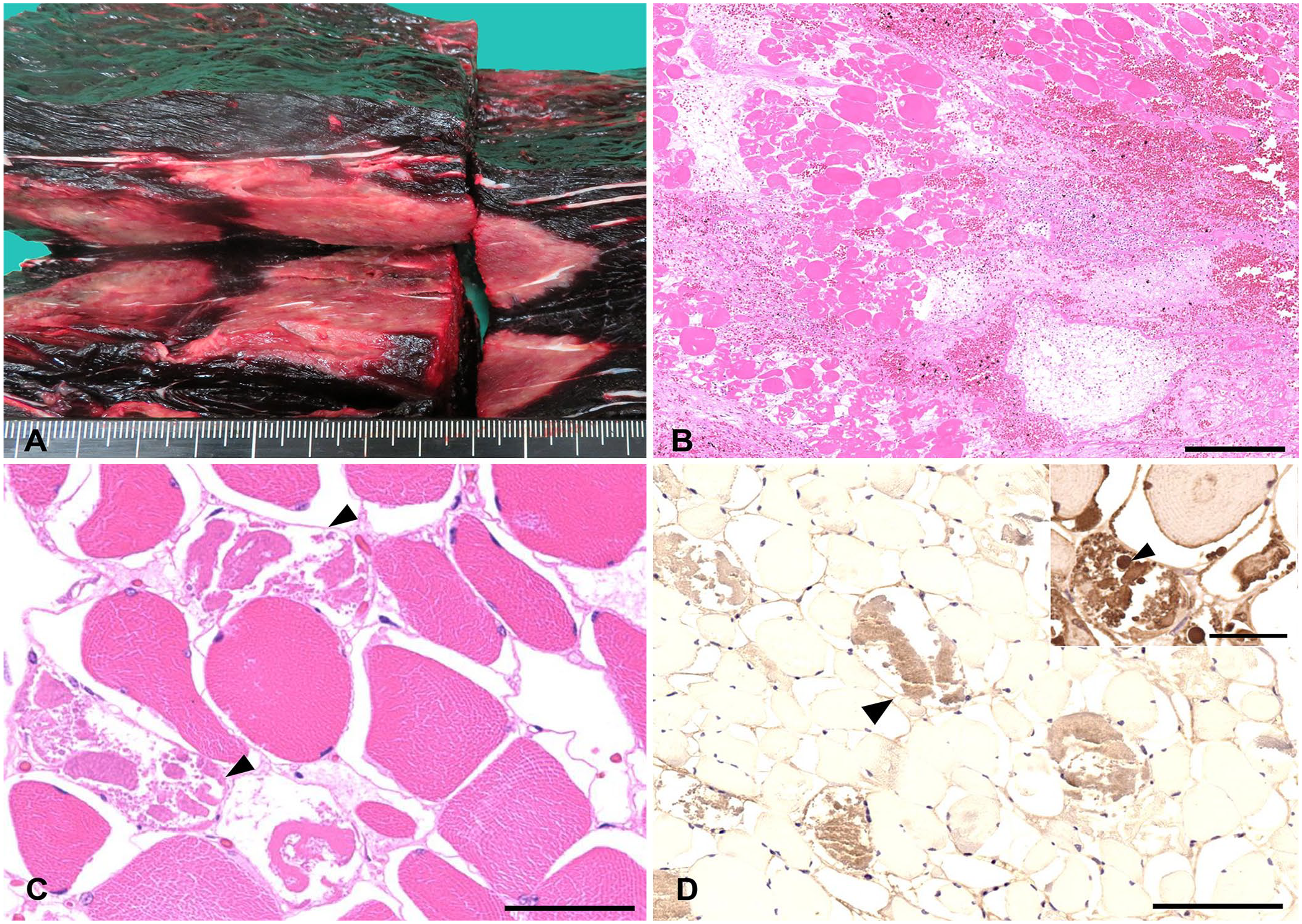
The dolphin died and was refrigerated; an autopsy was performed within 16 h after death. Erosive wounds were present at the dorsal fin, left pectoral fin, and tail fluke. Extensive multifocal hemorrhagic necrosis was detected in several muscles, including the sternomastoideus muscle and the longissimus dorsi muscles, with pale-pink discoloration of the cut surface of non-hemorrhagic muscle (Fig. 1A). The lungs were moderately edematous and failed to collapse; petechiae were present in the ventral part of the left lung with a large amount of serosanguineous fluid in the thoracic cavity. Lung parenchyma was rubbery and dark-red on cut surface, but no fluid was present in the bronchial lumens. Serosanguineous fluid (~500 mL) was observed in the abdominal cavity. Petechiae were present on the splenic capsule and on the thickened gastric mucosa. No macroscopic lesions were observed in other organs.
Rhabdomyolysis in a bottlenose dolphin.
Tissues samples were fixed in 10% neutral-buffered formalin, processed routinely, and sections stained with H&E; kidney sections were stained with von Kossa, periodic acid–Schiff (PAS), and Masson trichrome. Immunohistochemistry (IHC) was performed on kidney sections using anti-human myoglobin antibody, anti-human aquaporin 1 (AQP1) antibody, anti-human fibrinogen, and anti-human heat shock protein 60 (HSP60; Table 2). Antibodies used were modified from a previous publication 14 or were based on the homology of amino acid sequences between humans and dolphins. After antigen retrieval, all slides were immersed in methanol with 0.3% H2O2 for 30 min to block endogenous peroxidase, then in Blocking 1 solution (Nacalai Tesque) or 1.0% skim milk solution for 30 min to block nonspecific binding. The primary antibody was applied for 1 h at 37°C under high-moisture conditions. Secondary antibody reactions were performed (Histofine simple stain MAX-PO; Nichirei Bioscience). Healthy dolphin muscle and kidney tissues were included as negative controls. Immunoreactivity was visualized using 3,3′-diaminobenzidine substrate (MilliporeSigma) and hematoxylin counterstain.
The antibody and immunohistochemistry methodology used in a bottlenose dolphin.

NA = not available.
Microscopically, hemorrhagic myonecrosis was prominent. Multifocal myofiber degeneration and necrosis was surrounded by moderate amounts of edema fluid (Fig. 1B). Affected myofibers had lost cross-striations, nuclei were pyknotic or karyorrhectic, and cytoplasm was pale and fragmented (Fig. 1C). Immunohistochemically, necrotic myofibers exhibited granular intracytoplasmic immunoreactivity with anti-fibrinogen antibody (Fig. 1D) compared to healthy myofibers. The intracellular myoglobin globules expressed dark-brown homogeneous positivity within fragmented myofibers (Fig. 1D, inset). Occasionally, necrotic myofibers were mineralized.
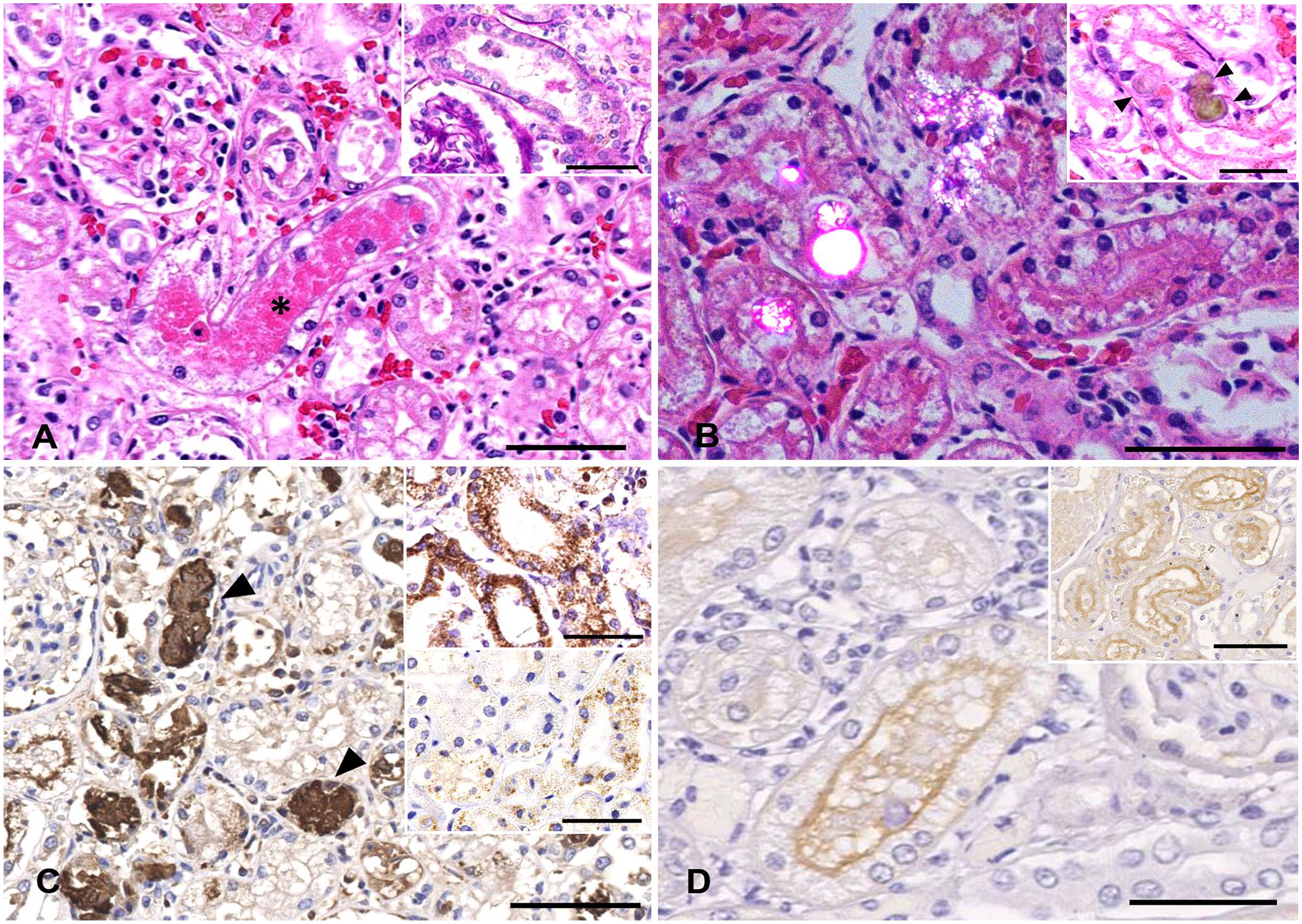
Renal tubular degeneration and necrosis was widespread and marked throughout the cortex and medulla; epithelial cells were markedly degenerate, swollen with vacuolated cytoplasm and loss of nuclei. Tubular lumens were dilated and contained eosinophilic granular casts (Fig. 2A), which were labeled intensely by anti-myoglobin antibody. The brush borders of proximal convoluted tubules were lost (Fig. 2A, inset). Multifocally, intratubular crystals were observed (~20 crystals/low power field [10×]), appearing strongly birefringent under polarized light (Fig. 2B). Various sizes of homogeneous, opaque, green-to-brown crystals were seen, with some irregularly shaped, some round-to-oval, and some in lamellar bundles with a stellate structure. Crystals were detected multifocally in renal tubular epithelial cells (endocytosis of crystals) and in lumens of degenerate proximal and distal convoluted tubules (Fig. 2B, inset). Some epithelial cells were swollen with karyorrhectic nuclei, and there was sloughing of cells into tubular lumens. The crystals did not react to von Kossa, PAS, or Masson trichrome stains.
Rhabdomyolysis and myoglobinuric nephrosis causing acute kidney injury in a bottlenose dolphin.
In other histologic findings, lung sections were characterized by severe suppurative bronchopneumonia; airways and alveolar spaces were filled by neutrophils, lymphocytes, macrophages, and cellular debris. A small number of cocci were present in macrophages. We found no evidence of endometritis or vaginitis.
Immunohistochemically, renal myoglobin globules were granular and brown, and various-sized globules were intracytoplasmic and in tubular lumens (Fig. 2C). The degenerate proximal tubules in the cortex expressed HSP60 intensely in the cytoplasm (Fig. 2C, inset, upper) compared to proximal tubules of a normal kidney, which expressed only mild immunostaining (Fig. 2C, inset, lower). Mild expression of HSP60 was observed in medullary collecting ducts; there was no differential expression in the renal medulla between the affected and control kidneys. AQP1-positive cells had decreased expression and weak intensity in the kidney of the dolphin with AKI, particularly in the cortex (Fig. 2D), compared to kidney tissue from a normal dolphin (Fig. 2D, inset).
The intracellular immunostaining of fibrinogen in necrotic myofibers is consistent with a previous study in cetaceans,14,15 indicating that fibrinogen is an important marker for ischemic muscle injury. Elevated serum activities of CK and AST, which are released when myofibers are disrupted, are markers of rhabdomyolysis in domestic animals and rat models.5,8 The elevations of CK and AST activities on the morning of day 4 suggest that rhabdomyolysis had occurred on day 3. High serum concentrations of creatinine and urea were observed on days 5–13, indicating that the dolphin had developed AKI after muscle injury. The mechanisms underlying AKI secondary to rhabdomyolysis remains controversial. The most severe complication of rhabdomyolysis is extensive exposure to myoglobin components. The renal myoglobin casts observed with IHC analysis may have contributed to renal tubular necrosis. Myoglobin casts can trigger AKI via renal vasoconstriction, tubular obstruction by intratubular casts, and direct toxicity of myoglobin to tubular epithelial cells. 25
HSP60, an intracellular protein involved in cellular homeostasis, has been used as a marker of cellular injury in induced acute tubular injury in rats. 13 A similar pattern of results was obtained in cytoplasmic expression in affected tubules in the renal cortex, including proximal and distal convoluted tubules, and the intensity might be related to the severity of degeneration. Expression of HSP60 was mild in both proximal and distal convoluted tubules in the cortex of the control kidney. The marked immunostaining of this intracellular protein in tubule cells in our study is consistent with AKI.
AQP1, a member of a family of membrane water channels, plays an essential role in the concentration of urine. 19 The expression of AQP1 was decreased in tubular epithelial cells throughout our dolphin AKI kidney, especially in the cortex, compared to the non-AKI kidney. Renal AQP1 expression has been reported to be decreased in experimental rat models of AKI.1,17
The composition of the renal crystals in our case is unclear; 2 possible sources are considered. First, calcium oxalate crystals were suspected. These have been reported in cetaceans with neutral-to-acidic urine conditions. 16 These crystals are known to take various shapes, 9 appearing as colorless to yellow-brown 21 birefringent crystals. Renal oxalosis delayed recovery in AKI human patients in one study and was postulated to have exacerbated the severity of the lesion. 26 Second, our dolphin had been treated with amoxicillin–clavulanic acid and enrofloxacin before azotemia occurred. Amoxicillin has been reported to induce crystalline nephropathy in human patients treated with high intravenous doses under acidic urine conditions. 11 However, enrofloxacin administered together with lactated Ringer solution is thought to induce alkaline urine. The major metabolite of enrofloxacin is ciprofloxacin, the plasma concentration of which varies among animal species depending on absorption ability and administration route. 23 Ciprofloxacin has been reported to generate variably shaped crystals in alkaline urine, including needle-shaped stellate forms, fans, stars, sheaves, butterflies, and unusual shapes with lamellar structures.10,22 Lactated Ringer solution can induce alkaline urine in patients with doxylamine-induced rhabdomyolysis 7 ; hence, the lactated Ringer solution administered in our case might have induced alkaline urine and exacerbated ciprofloxacin precipitation. However, urine pH was not evaluated in our study. Based on our morphologic and polarized light observations, both calcium oxalate and enrofloxacin metabolite crystals remain as possibilities.
The causes of rhabdomyolysis in our case are debatable. Ischemic injury and trauma to muscle are suspected as simultaneous causes. The dolphin had a shock reaction, which would have impaired blood circulation and reduced peripheral blood volume by vasoconstriction. Shock may exacerbate ischemic muscle injury and reduce renal blood flow. In addition, cetacean species possess higher myoglobin content in skeletal muscle compared to terrestrial animals, which might be a factor contributing to myoglobinuric nephrosis. 24 The clinical presentation, gross findings, and histopathologic and immunohistochemical features imply that myoglobinuria and crystalluria led to AKI in our dolphin.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
