Abstract
Two male juvenile central bearded dragons (Pogona vitticeps) were submitted for postmortem examination after dying at their respective homes. Dragon 1 had marked hemopericardium with restrictive epicarditis. The inner aspect of the distended pericardial sac was lined by a fibrinoheterophilic membrane. In addition, granulomas abutted the testes. Dragon 2 had acute hemopericardium and granulomatous arteritis of the great vessels exiting the heart. Histologically, both animals had granulomatous arteritis of the large arteries with intrahistiocytic gram-positive, slightly elongated, up to 2 μm long microorganisms that contained a vacuole. These microorganisms were also present in the paratesticular granulomas. On transmission electron microscopy, the microorganisms were identified as microsporidians given the presence of exospore, endospore, vacuole, nucleus, and a filament with 4–6 coils. The microsporidia were identified as Encephalitozoon pogonae based on sequencing of the internal transcribed spacer 1 of the ribosomal RNA genes. Microsporidia are agents of disease in bearded dragons. Intrapericardial arteritis of large arteries with hemopericardium or restrictive epicarditis is a fatal manifestation of this infection.
Microsporidia are atypical fungi that parasitize and cause disease in invertebrates, fish, amphibians, reptiles, and mammals. 17 Microsporidia reproduce within the host cell, including macrophages, in which they are detectable histologically.5,16 The spores are oblong and have a clear vacuole. They measure up to 3 μm long and ~1 μm in diameter, although some species have larger spores. 2 Mature spores are refractile, gram-positive, and have variable tinctorial properties when stained with silver stains, Giemsa stain, and when subjected to the PAS reaction. 5 On electron microscopy, the spores are characterized by an exospore, endospore, and coiled polar tubular filament.9,11 The number of windings of this filament varies depending on species and developmental stage. Fatal and non-fatal microsporidian infections have been reported in central bearded dragons (syn. inland bearded dragon; Pogona vitticeps).7,10,12–14 In 2016, a microsporidian detected in a bearded dragon was identified as the new species Encephalitozoon pogonae. 14
Two privately owned bearded dragons were submitted to the Minnesota Veterinary Diagnostic Laboratory (St. Paul, MN) for postmortem examination in June 2016 and March 2018 respectively. Both were intact juvenile males (6- and 10-mo-old, respectively). Dragon 1 had a 5-d course of anorexia and lethargy with terminal right head tilt. The animal was fed a daily diet of 10–15 superworms (commercially purchased), green turnips, and collard greens. The animal died 1 d after the owner noted that the beard of the animal had turned black. The owner reported that another bearded dragon in the household was healthy. Dragon 2, belonging to a different owner, was found dead without premonitory signs. Further information regarding the diet of this animal was not available.
Both animals were in a good nutritional state, weighing 145 g and 372 g, respectively. Grossly, the pericardial sac of dragon 1 was markedly dilated and filled with clotted and unclotted blood (Fig. 1). Fibrinous exudate adhered to and diffusely coated the inner aspect of the pericardial sac. Poorly defined white homogeneous glossy tissue was present at the base of the heart. The heart was maximally contracted. The coelomic cavity contained ~4 mL of clear amber watery fluid (hydrocoelom). An ~1 × 1.3 cm diameter conglomerate of ~0.5 cm diameter, smooth, beige nodules was present next to both testes. The dilated pericardial sac of dragon 2 was filled with unclotted blood. Multiple, beige, smooth, nodular masses, up to 1 cm diameter, expanded the walls of the large arteries exiting the ventricle (aortic arches and/or pulmonary artery). In both cases, the entire hearts with the pericardial sacs and samples of lung, liver, kidney, testes and epididymis, stomach, intestine, brain, and spleen, as well as the paratesticular masses in dragon 1, were fixed in 10% buffered formalin, routinely processed for histopathology, and stained with hematoxylin and eosin. Selected sections of heart (dragon 1 and 2) and testis (dragon 1) were stained with Gram stain, Gomori methenamine silver (GMS) stain, Giemsa stain, acid-fast stain, and subjected to a periodic acid–Schiff (PAS) reaction.
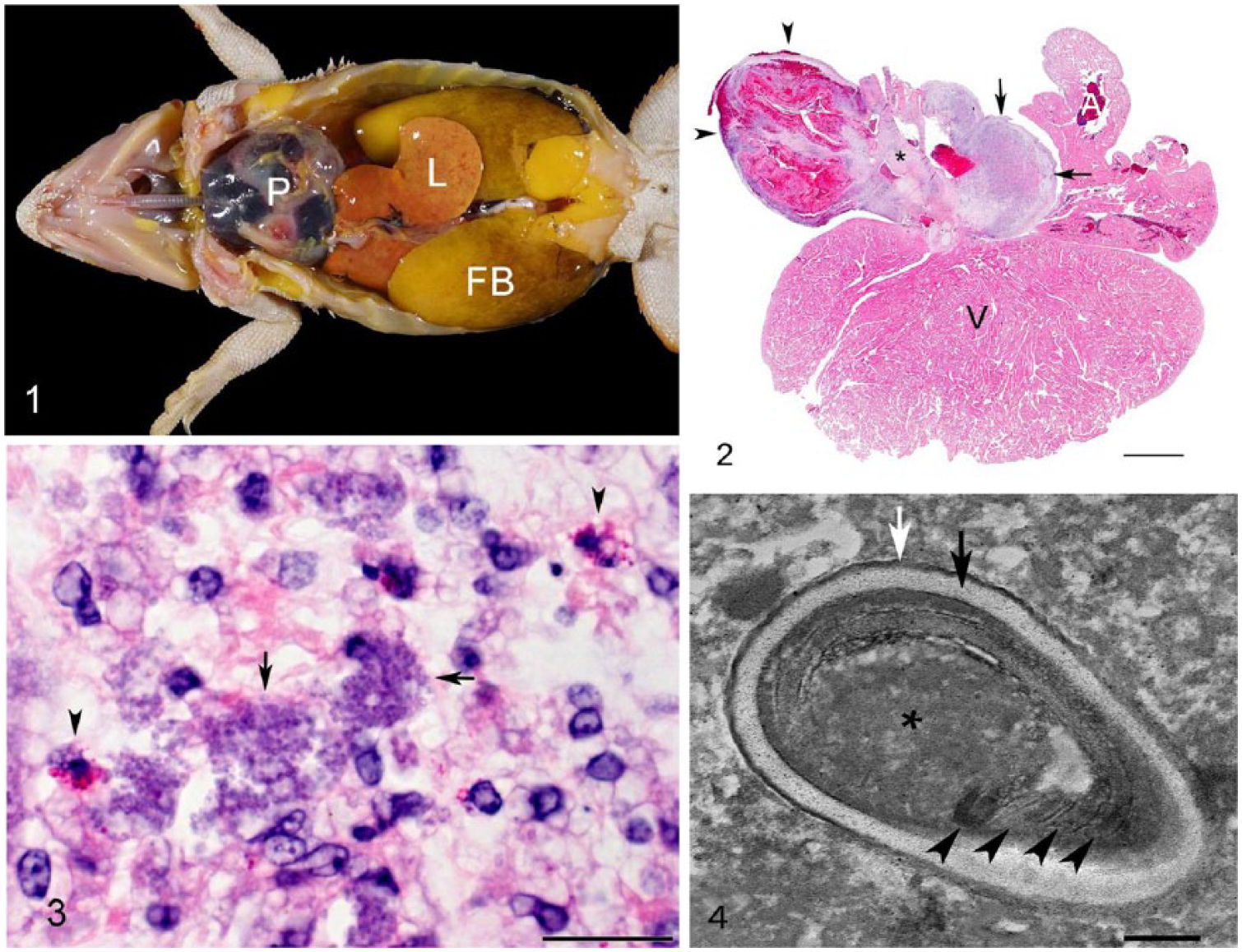
Intrapericardial Encephalitozoon pogonae–associated arteritis and hemopericardium in 2 juvenile central bearded dragons (Pogona vitticeps).
Histologically, both animals had severe granulomatous arteritis at the root of the great vessels exiting the ventricle (aortic arches and/or pulmonary artery). The arteritis was most pronounced on the adventitial aspect of the vessels, but in places was transmural. It was characterized by infiltration of abundant macrophages, some of which contained numerous, weakly basophilic-staining intracytoplasmic microorganisms (Fig. 2). These microorganisms were slightly elongated, up to 2 μm long, and contained a vacuole. The organisms were gram-positive and weakly PAS-positive but acid-fast–negative and negative with GMS stain (Fig. 3). Giemsa stain highlighted the wall and nucleus of the microorganisms. In dragon 1, the inflammatory process extended along the epicardial surface and mantled the entire circumference of the heart. In this animal, epithelioid macrophages, some of which contained microorganisms, were occasionally arranged around a core of eosinophilic necrotic debris admixed with extracellular microorganisms that had the same features as the intrahistiocytic microorganisms. The pericardial sac was thickened by fibrosis and was lined internally by a fibrinous membrane with degenerate granulocytes and erythrocytes. The paratesticular masses were composed of epithelioid macrophages with features similar to those described in the large arteries; abundant microorganisms were present. These nodules abutted the testes and epididymis but did not appear to invade these structures. The adrenal glands were not detected histologically. In dragon 2, a prominent outpouching of an affected large artery (likely an aortic arch) resembling an aneurysm was present. A blood coagulum adhered to the adventitial surface of this outpouching, indicating perforation. No microscopic lesions were identified in any other tissues examined in either case.
On transmission electron microscopy using formalin-fixed paratesticular masses from dragon 1, and formalin-fixed, paraffin-embedded (FFPE) heart tissue from dragon 2, the microorganisms were identified as microsporidians based on the presence of exospore, endospore, vacuole, nucleus, and a polar filament. The organisms averaged 1.83 μm long × 0.92 μm wide. The polar filaments had 4–6 coils and averaged 61 nm diameter (Fig. 4).
DNA was extracted from FFPE paratesticular masses from dragon 1 (DNeasy blood and tissue kit; Qiagen, Germantown, MD) according to the manufacturer’s instructions. The internal transcribed spacer 1 (ITS1) region of the ribosomal RNA genes was amplified by PCR using primer pair MSP3 and MSP4A as described previously. 8 The PCR products, including a negative and previously identified positive control, were run on 1.5% agarose gel with ethidium bromide and were visualized under ultraviolet light. Amplified products were excised and purified (QIAquick gel extraction kit; Qiagen), then submitted for Sanger sequencing in both directions (Genewiz, South Plainfield, NJ). Primer sequences were edited out prior to further analyses. The resultant 265-bp sequence was compared to those in GenBank (National Center for Biotechnology Information, Bethesda, MD), EMBL (Cambridge, UK), and Data Bank of Japan (Mishima, Shizuoka, Japan) databases using BLASTn. 1 The sequence was 100% homologous with E. pogonae (GenBank accession KR998311). The closest non–E. pogonae sequences were E. cuniculi (89–92% homology). Our sequence was submitted to GenBank under accession MH686240.
Microsporidiosis has been reported previously in bearded dragons. In 2 previous reports of bearded dragons infected with E. pogonae, the heart was mentioned as a site of infection but there were no references to arterial involvement or hemopericardium.13,14 Although vasculitis is a known finding in carnivores and monkeys with microsporidial infection, infection of larger arteries is rare. 18 Aortitis and arteritis of large caliber arteries, caused by E. cuniculi, has been described as the principal lesion leading to fatal cardiac insufficiency in a Goeldi monkey. 4 Both of our cases were characterized by a hitherto unreported, to our knowledge, manifestation of microsporidiosis in bearded dragons, namely granulomatous arteritis of the large arteries exiting the ventricle associated with severe hemorrhage into the pericardial sac. The exact origin of the hemorrhage was not apparent. The intrapericardial effusion had a slightly protracted course in dragon 1, as evidenced by a fibrinoheterophilic membrane lining the inner aspect of the pericardial sac. The microsporidial epicarditis present in this animal likely restricted diastolic dilation of the ventricle. The hydrocoelom is evidence of heart failure, although the good nutritional state of the animal suggests that the disease did not negatively affect the animal for a prolonged time. The course of the hemopericardium in dragon 2 was peracute without any notable inflammatory reaction in the pericardial sac. The intrapericardial hemorrhage likely was the result of perforation of a large artery based on the presence of the blood coagulum adherent to the adventitia at the site of one of the grossly apparent arterial granulomas. Based on previous reports,7,12–14 adrenal gland and paragonadal involvement, as noted in dragon 1, is quite common in bearded dragons with microsporidiosis. Aneurysmal vascular disease is a common problem in bearded dragons; further investigation of the role of E. pogonae in vascular damage is indicated. 15
The source of the microsporidian and the route of infection in both animals are uncertain. Bearded dragons are omnivores, and vegetables and insects usually comprise the majority of their captive diet. Insects harbor many known microsporidian organisms. 17 The possibility of transmission of microsporidians from their insect prey to bearded dragons has been examined, 7 but the examined feeder crickets were negative for microsporidia. Fecal contamination of fruits and vegetables has been found to be a source of microsporidia. 3 Vertical transmission of Encephalitozoon species has been documented in rabbits infected with E. cuniculi. 6 Further epidemiologic investigations are indicated.
Footnotes
Acknowledgements
We thank the histology staff of the Minnesota Veterinary Diagnostic Laboratory for their excellent preparations.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
