Abstract
A metastatic gastric neuroendocrine carcinoma in a 2.5-year-old inland bearded dragon (Pogona vitticeps) with a chronic history of anorexia, weight loss, depression, and acute melena is described. Histologic examination of the gastric mass revealed a densely cellular tumor arranged in nests and occasional rosettes of hyperchromatic cells with oval to spindle-shaped nuclei and minimal cytoplasm; the tumor was supported by a moderate fibrovascular stroma. Similar cells invaded through the gastric mucosa, and there were multiple hepatic metastases. The neoplastic cells were weakly immunopositive for neuron-specific enolase and moderately positive for somatostatin but were negative for chromogranin AB and gastrin. Ultrastructural studies revealed scattered neurosecretory granules in the neoplastic cells, confirming the diagnosis of a neuroendocrine carcinoma.
Neuroendocrine (NE) cells are derived embryologically from the gut and are widely distributed in various mammalian tissues, such as the tracheobronchial tree, liver, pancreas, and genitourinary system. 15 In human medicine, NE carcinomas have been found in a wide range of organs, with the gastrointestinal 2 and pulmonary tracts 12 being the most common sites. In domestic animals, NE carcinomas have been occasionally reported in the intes-tines, 6 liver, 18 lungs, 11 esophagus, 17 skin, 19 and nasal cavity, 16 specifically from dogs and cats.
Neuroendocrine carcinomas typically have the histo-pathologic pattern of sheets and nests of small- to medium-sized cells separated by a delicate fibrovascular stroma, which are characteristics that help the pathologist recognize these neoplasms as being of endocrine origin. Neuroendo-crine differentiation is further defined by the presence of ultrastructural dense-core neurosecretory granules and immunohistochemical expression of a large array of NE markers, but is most consistently defined by neuron-specific enolase (NSE), synaptophysin and/or chromogranin. The presence of neurosecretory granules is related to the degree of tumor cell differentiation. 27
Neoplasia appears to be rarely described in bearded dragons, and only a few reports exist in the veterinary literature documenting lymphoid and myelogenous leukemia most commonly in this species. 25 However, because NE cells have been located within the gastrointestinal mucosal epithelium as well as the islet parenchyma of reptiles, bearded dragons are susceptible to this kind of neoplasm. 20,21
A 2.5-year-old, female inland bearded dragon (Pogona vitticeps) that had been acquired by the owner 1 year previously was mildly thin (217 g) and had a scarred left eyelid and missing right eye due to prior mate aggression. During the first 9 months of ownership, the bearded dragon gained 84 g due to growth and achieved a good body condition (301 g). The diet consisted of romaine lettuce, or occasionally other dark green leafy vegetables, and 3 superworms (Zophobas morio) daily. The female was housed with a male cage mate. Housing consisted of a 91 cm × 61 cm × 61 cm enclosure within a 29°C room, including a basking area with an ultraviolet B light. a A 60-W light bulb attached above the screened area at the top of the cage b provided a warmer area of approximately 30°C. Additionally, a water bowl large enough for an entire bearded dragon to be immersed was present. Gradually, the female bearded dragon became partially anorexic, lost weight, and became lethargic. At a weight of 265 g, she was presented for examination at Small Animal Clinical Sciences, College of Veterinary Medicine, University of Tennessee (UTCVM), and heterophilic leukocytosis with a left shift, azurophilia, and monocytosis were present. In addition, a mild, nonregenerative anemia was documented (Table 1). No abnormalities were palpated or auscultated on physical examination. Enrofloxacin c (5 mg/kg, subcuta-neously every 24 hr), itraconazole d (5 mg/kg, orally every 24 hr), and 0.9% saline (30 mg/kg, subcutaneously every 24 hr) were given until death. Fourteen days later, the bearded dragon developed acute melena and was euthanized under isoflurane gas anesthesia with pentobarbital given intravenously in the tail vein.
Leukogram and packed cell volume from a 2.5-year-old inland bearded dragon (Pogona vitticeps) presenting with anorexia, lethargy, and weight loss. *
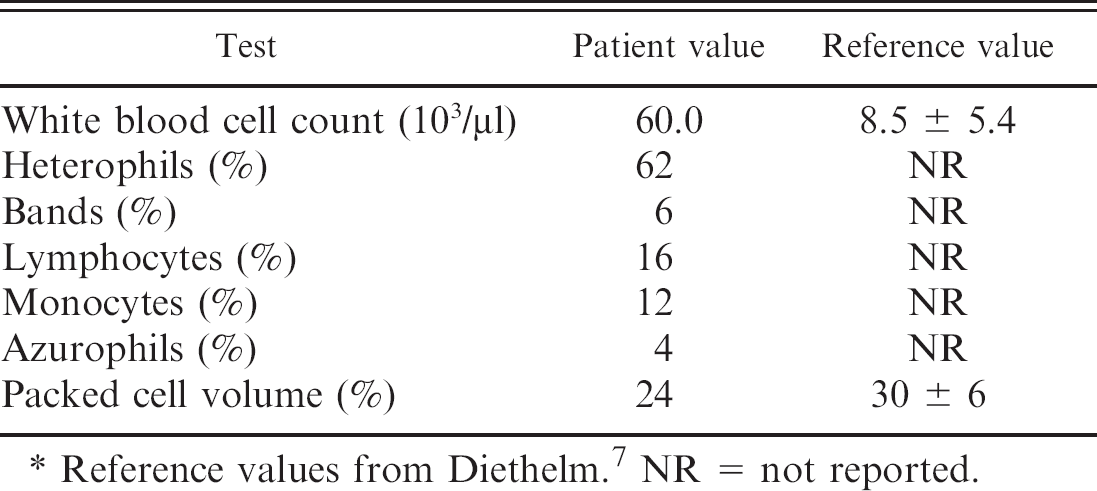
Reference values from Diethelm. 7 NR = not reported.
The bearded dragon was submitted to UTCVM Department of Pathobiology for postmortem examination. The bearded dragon was in thin body condition with little subcutaneous fat, and the coelomic fat pads were markedly atrophied. A single 5 mm × 5 mm × 4 mm, firm, white, transmural, ulcerated, nodular mass was present in the proximal portion of the stomach. The interior of the mass was white and of a homogenous consistency on cut section. The stomach contents were brown-black and of coffee ground consistency, indicative of melena, with an estimated volume of 0.5–1 ml. There were no contents in the small intestine. The liver was diffusely yellow and floated in formalin. Moderate, multifocal, white-tan, 1–5-mm diameter, firm nodules were present throughout the hepatic parenchyma (Fig. 1). Multiple tissues, including the stomach mass, liver, spleen, pancreas, heart, lung, ovary, and brain, were fixed in 10% neutral-buffered formalin, processed routinely, embedded in paraffin, sectioned, and stained with hematoxylin and eosin.
Histologically, the gastric mass (Fig. 2) was characterized by a poorly demarcated, unencapsulated, densely cellular expansile and infiltrative mass composed of round to polygonal cells arranged in sheets and nests and occasionally forming rosettes. The cells had scant, eosino-philic, moderately vacuolated cytoplasm with indistinct borders, and round to oval nuclei with moderately vesiculated chromatin. There were occasional prominent nucleoli and up to 6 mitotic figures per high-power field (40x). There was moderate to marked anisocytosis, anisokaryosis, multifocal karyomegaly, and a variable nuclear–cytoplasmic ratio. The neoplasm was supported by a moderate, fibrovascular stroma. The neoplastic cells invaded from the submucosa to the mucosal surface, creating focal ulceration and fibrosis. The tumor cells invaded along the submucosa, and islands of neoplastic cells were present in endothelial-lined vessels (Fig. 3). The multifocal masses in the liver consisted of neoplastic cells with a similar pattern of arrangement and morphologic characteristics as those described for the gastric mass. Within the ovary was a focal area of marked inflammation composed of macrophages, lymphocytes, and plasma cells, as well as epithelioid macrophages, and numerous multi-nucleated giant cells with multifocal mineralization; yolk peritonitis was an incidental finding.
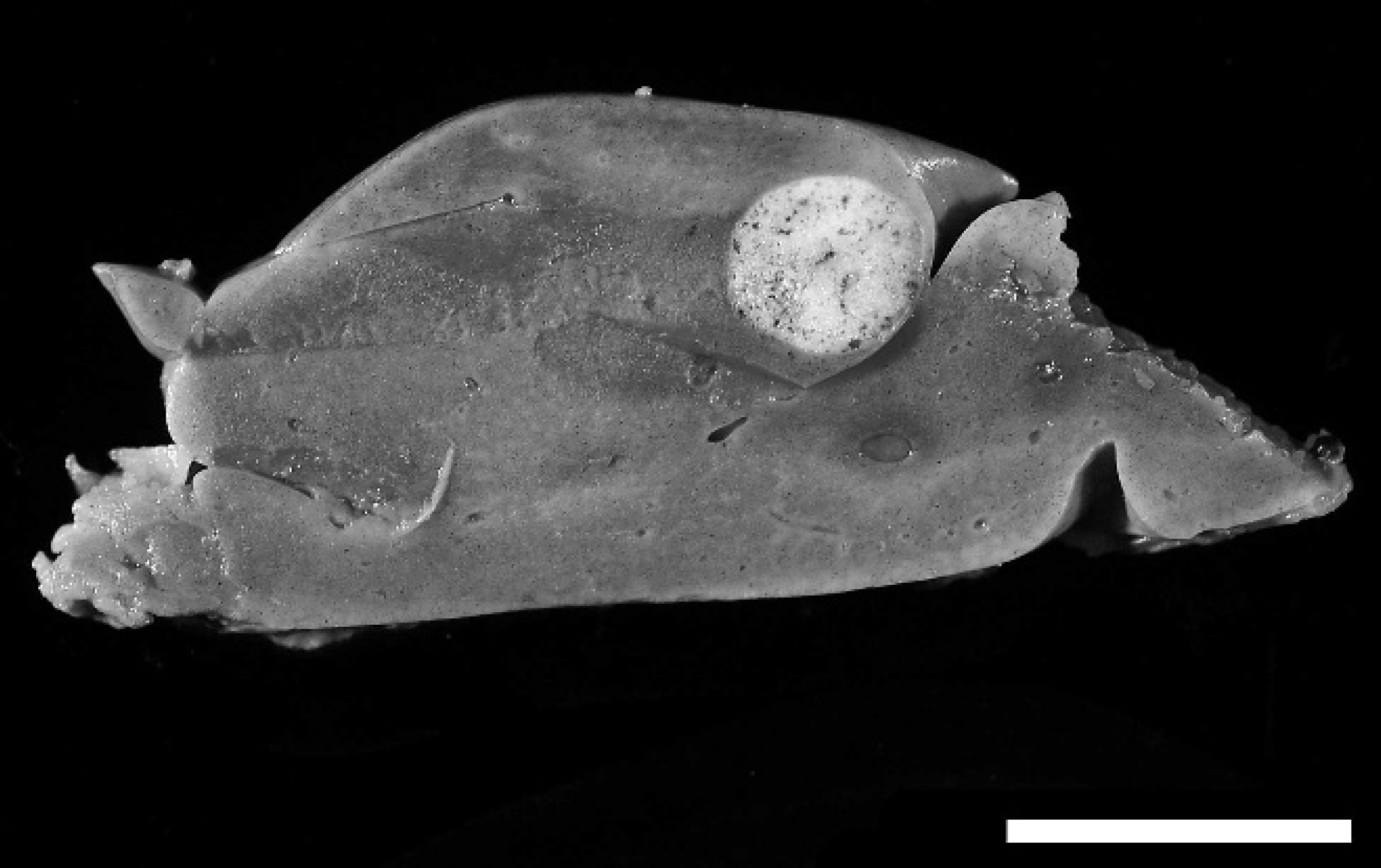
Liver; inland bearded dragon (Pogona vitticeps). Cross-section of liver with a focal, tan nodular mass consistent with metastatic tumor. The liver was also diffusely yellow, indicative of hepatic lipidosis. Bar = 1 cm.
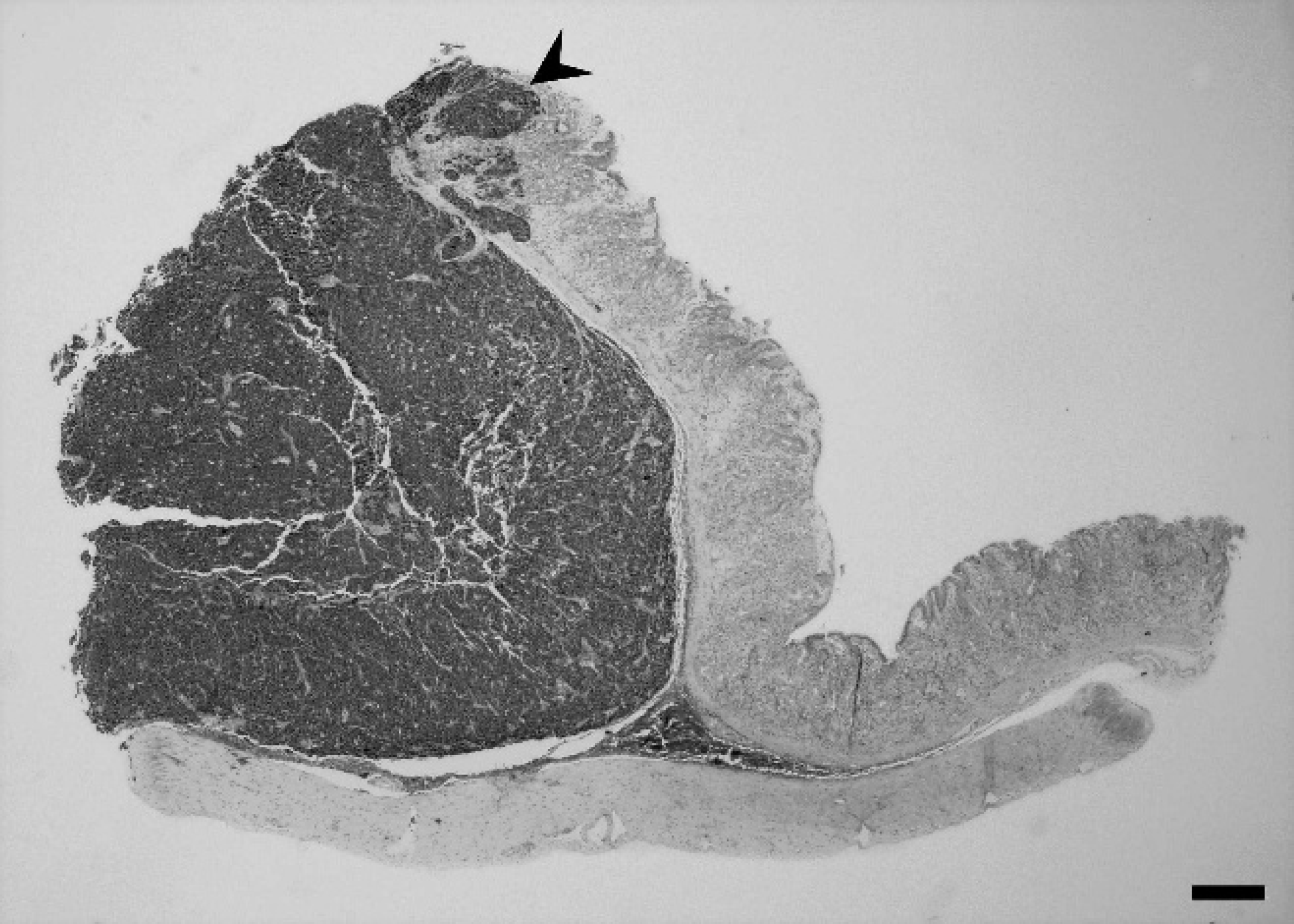
Stomach; inland bearded dragon (Pogona vitticeps). Gastric neuroendocrine carcinoma with neoplastic cells organized in small lobules supported by delicate fibrovascular stroma. Local invasion into the mucosa with subsequent ulceration is noted (arrowhead). Hematoxylin and eosin. Bar = 600 μm.
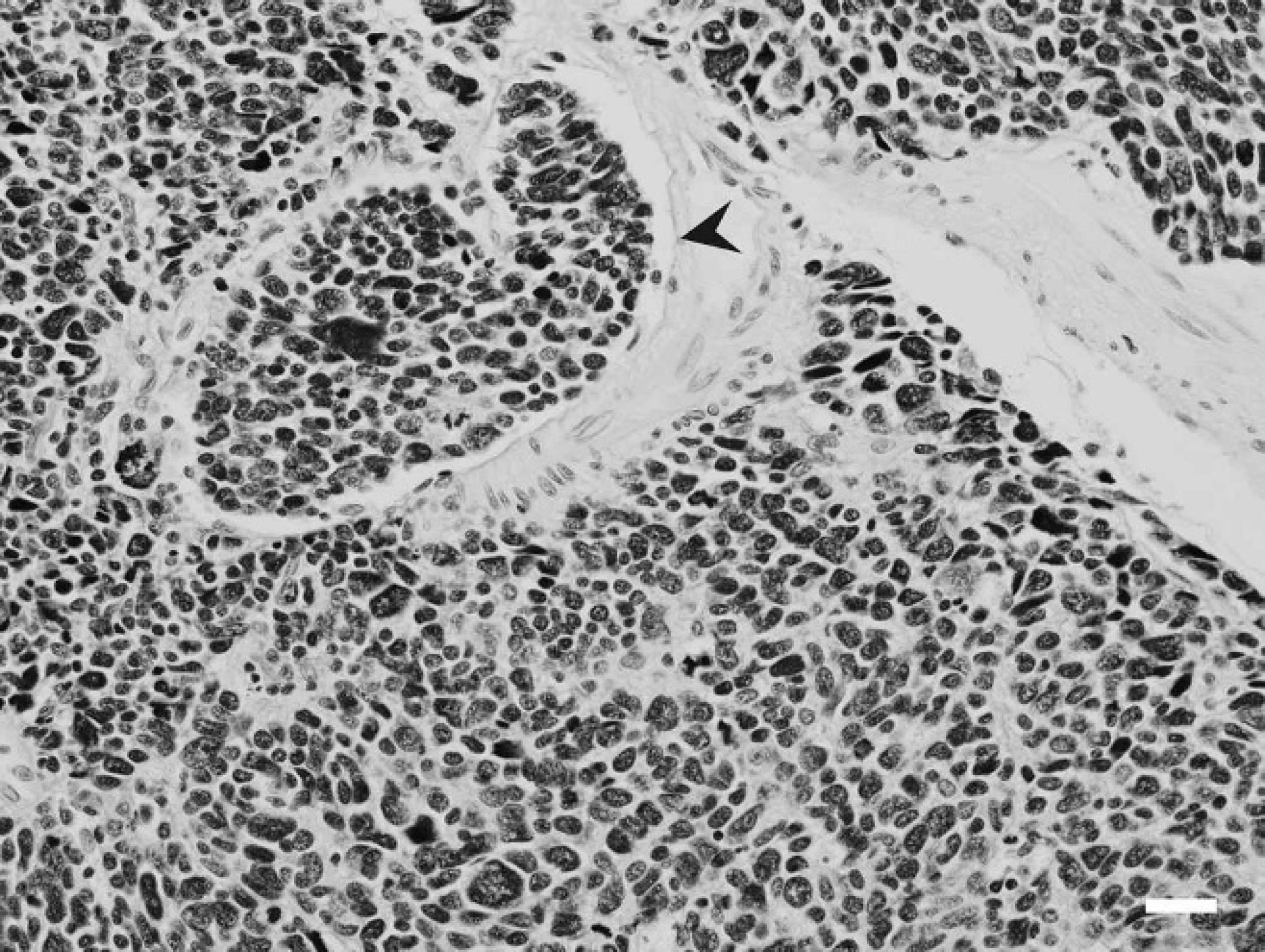
Stomach; inland bearded dragon (Pogona vitticeps). Gastric neuroendocrine carcinoma with intravascular invasion (arrowhead). Hematoxylin and eosin. Bar = 35 μm.

Liver; inland bearded dragon (Pogona vitticeps). Metastatic neuroendocrine carcinoma with moderate stippled cytoplasmic staining (arrowhead). Antisomatostatin stain. Bar = 70 μm. Inset shows positive somatostatin expression in neuroen-docrine cells of the stomach.
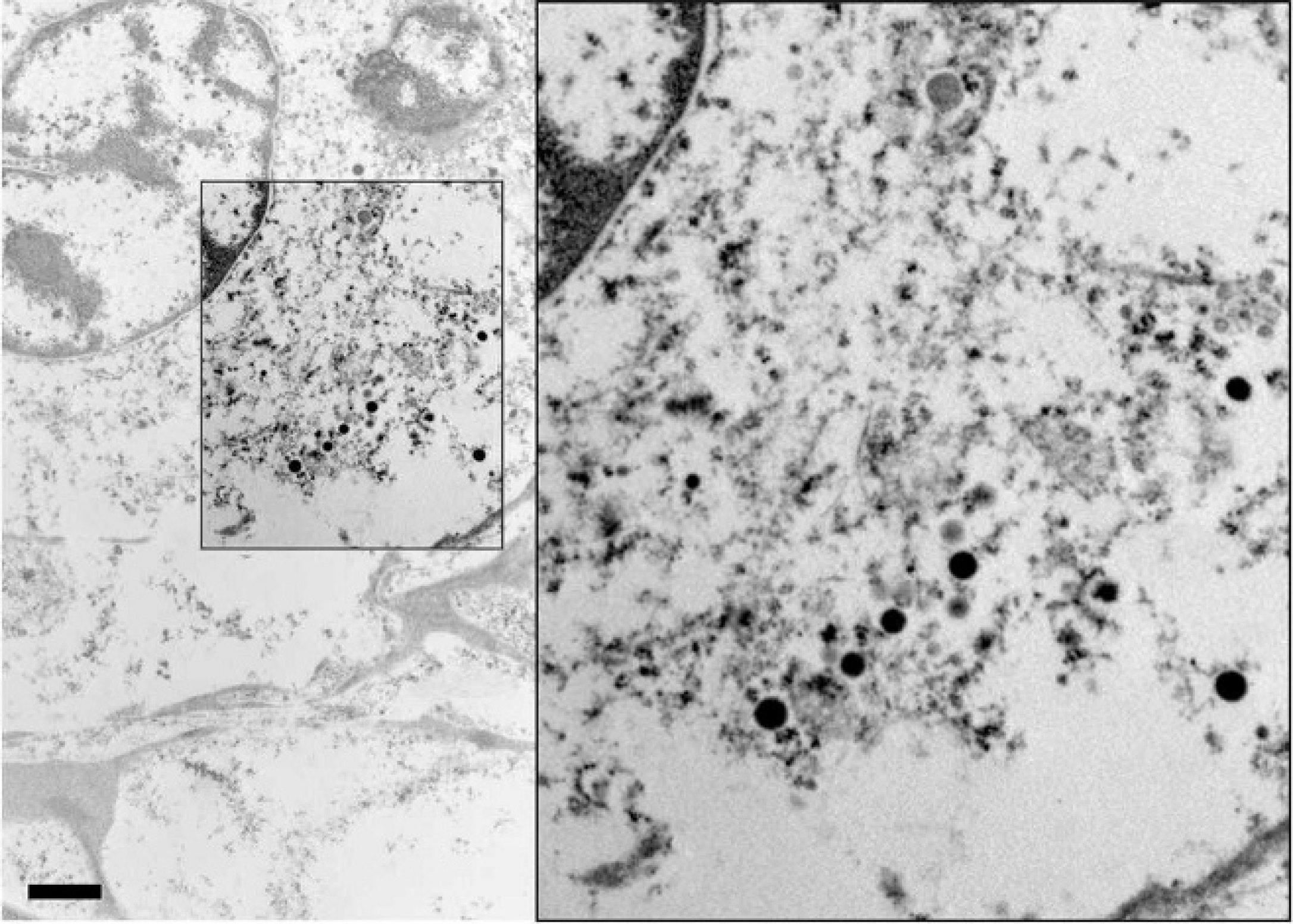
Inland bearded dragon (Pogona vitticeps). Metastatic tumor cells were poorly differentiated, and neurosecretory granules were sparse. When present, the neurosecretory granules were electron dense and approximately 200 nm in diameter (inset). Transmission electron microscopy. Bar = 1.14 μm. Inset: 2x magnification.
The histologic appearance of the neoplastic cells was consistent with a NE carcinoma, and because this tumor type had not been previously described in bearded dragons, further confirmatory procedures were initiated. The use of Churukian–Schenk stain to identify the neurosecretory granules has been previously shown to be the most accurate light microscopic special stain for this purpose. 23 Surprisingly, Churukian–Schenk stain proved to be inconclusive in the current case, perhaps because of poor cross-reaction with reptilian tissue, but more likely because of the poorly differentiated nature of the tumor (i.e., fewer granules).
Immunohistochemical staining for endocrine markers was performed by the Diagnostic Center for Population and Animal Health, Michigan State University (Lansing, MI). Immunohistochemistry (IHC) was performed by the UTCVM Immunohistochemistry Laboratory to evaluate expression of NSE and somatostatin. Slides were initially deparaffinized and rehydrated through graded alcohols to water. Antigen retrieval for NSE was performed on slides by first heating in citrate buffer e (pH 9.0) for 25 min at 95°C in a steamer and subsequently leaving the slides to cool for 20 min. For somatostatin, antigen retrieval was performed on slides by heating in ethylenediamine tetra-acetic acid e (pH 9) for 25 min, followed by cooling. After antigen retrieval, all slides were processed using a computer-controlled, automated stainer. f Slides were first soaked in Tris-buffered saline (pH 7.6) for 5 min and then incubated in hydrogen peroxide 3% for 5 min. A 5-min nonserum protein block d was used. The anti-NSE primary antibody g (1:25,000) and anti-somatostatin antibody h (1:25) were applied for 30 min at room temperature. A horseradish peroxidase–labeled polymer i was applied to all slides and incubated for 30 min. All slides were then rinsed, and 3,39-diaminobenzidine chromogen was applied to slides for 10 min. Last, slides were rinsed, counterstained with Harris hematoxylin, dehydrated, and cover slipped. Negative control slides were included with primary antiserum replaced by Universal Negative Control+ mouse serum, e where appropriate.
Bearded dragon stomach served as a positive control tissue for somatostatin, which was evident in individual cells lining the gastric crypts. Bearded dragon pancreas served as a positive control tissue for NSE, which was evident as strong diffuse expression in the islet cells.
For examination by electron microscopy, metastatic tumor tissue from the liver was fixed in formalin as above. Samples were washed 3 times for 10 min in water, and then postfixed for 60 min in 2% osmium tetroxide. Samples were again washed in water, and then dehydrated in a graded ethanol series at 15-min intervals. After final dehydration, samples were gradually infiltrated with epoxy resin j during a 24-hr period. After changing to fresh epoxy, samples were placed in a 68°C oven for 24 hr. Thin sections of tissue were cut with an ultramicrotome, k mounted on copper grids, and then stained with uranyl acetate and lead citrate before examination with a transmission electron microscope l operating at 75 keV.
Neoplastic cells were immunonegative for chromogranin AB and gastrin and positive for somatostatin. In-house IHC showed minimal immunopositivity for NSE, and approximately 40% of the neoplastic cells had moderately stippled cytoplasmic staining for somatostatin (Fig. 4). Ultrastructurally, the metastatic tumor cells contained small numbers of cytoplasmic secretory granules measuring approximately 200 nm in diameter (Fig. 5). A diagnosis of a somatostatin-producing NE carcinoma (somatostatinoma) was made based on histologic appearance, the immunohistochemical phenotype, and the ultrastructural demonstration of neuro-secretory granules within the neoplastic cells.
Although once thought of as uncommon, neoplasia is now encountered more often in reptile medicine. 24 The prevalence of neoplasia is reported to be highest in snakes, followed by lizards, chelonians, and crocodilians, but published reports in bearded dragons are rare. 9,24 Endocrine tumors in reptiles are very rare but have been reported in iguanids (1 islet cell tumor and adrenocortical carci-noma). 8 Although at the time of initial diagnosis of the current case, there were no reports of NE carcinomas in the available veterinary literature, a recent abstract and subsequent publication documents a series of gastric somatostatinomas, 21 and it is known that bearded dragons are susceptible to myelogenous leukemia. 25
In instances when previously unreported neoplasms are identified in exotic species, pathologists are often presented with a diagnostic challenge. Although histologic pattern and cell morphology can point diagnosticians in a certain direction, numerous other ancillary tests, such as special light microscopic stains, IHC, and electron microscopy, can help confirm initial suspicions about the origin of neoplastic tissues. These techniques were particularly helpful in the current case. Because of the poorly differentiated nature of the neoplastic cells, the silver stains and ultrastructural analysis did not closely mimic what might be expected in normal NE tissues; namely, positive staining and numerous intracytoplasmic neurosecretory granules.
Although rarely described in domestic animals, 13 somato-statinomas are described in human medicine and produce a predictable clinical syndrome. 14 Different endocrine cell types have been identified and characterized in the human gastric mucosa. In addition to the enterochromaffin-like cell producing histamine, the enterochromaffin cell producing serotonin, and the G cell producing gastrin, there is a D cell that produces somatostatin. 26 In humans, somatostatin has been shown to suppress pancreatic secretion of insulin, 1 glucagons, 10 and pancreatic polypeptides. 22 Similarly, in the intestinal tract, somatostatin inhibits gastrin release, gastric acid secretion, gastric motility, intestinal motility, intestinal absorption, and gallbladder contraction. 3–5 This leads to a particular syndrome characterized by diabetes mellitus, hypochlorhydria, pancreatic exocrine insufficiency, and gallstones. Gastric NE carcinomas in humans are known to behave particularly aggressively, have an extremely poor prognosis, and rarely produce somatostatin. 26
Although well described in other species, the primary neuroendocrine carcinoma of the stomach described in the current report represents one of the first recorded in a bearded dragon with multiple hepatic metastases. Based on this single case, the tumors can be particularly aggressive and have a poor prognosis. Hence, it is particularly important for clinical veterinarians to be aware of their existence, especially in bearded dragons presenting for nonspecific lethargy, anorexia, and weight loss, or gastrointestinal disturbances, and any collections of symptoms that mimic somatostatinoma in humans.
Acknowledgements. The authors thank Ms. Anik Va-sington for graphic assistance, Ms. Misty Bailey for editorial support, and Dr. Matti Kiupel for immunohisto-chemistry support.
Footnotes
a.
Reptisun, Zoo Med, San Luis Obispo, CA.
b.
Neodesha Plastics Inc., Neodesha, KS.
c.
Baytril, Bayer, Shawnee Mission, KS.
d.
Sporanox, Janssen, Titusville, NJ.
e.
Dako, Carpinteria, CA.
f.
Model S3400, Dako, Carpinteria, CA.
g.
MIG-N3, BioGenex, San Ramon, CA.
h.
Cell Marque, Rocklin, CA.
i.
Envision+ System HRP Anti-mouse, Dako, Carpinteria, CA.
j.
EMbed-812, Electron Microscopy Services, Hatfield, PA.
k.
Reichert OMU3, Reichert Microscope Services, Depew, NY.
l.
Hitachi H800, Hitachi High-Technologies Europe GmbH, Krefeld, Germany.
