Abstract
Isolates of Gram-negative anaerobic bacteria from reptiles have only occasionally been identified to the genus and species level in the veterinary medical literature. In particular, reports identifying Porphyromonas spp. from infections in reptiles are scarce. The present report describes unique Porphyromonas isolates obtained from necrosuppurative infections in central bearded dragons (Pogona vitticeps). The isolates grew in the presence of oxygen, were strongly hemolytic, and did not produce detectable black, iron porphyrin pigment. Biochemical identification kit numeric biocodes gave high but unreliable probabilities (>99.9%) for identification as Porphyromonas gingivalis. Partial 16S ribosomal RNA gene sequences of the isolates were identical to each other and shared 91% identity with those of Porphyromonas gulae. The isolates may represent a new reptile-associated Porphyromonas species.
Anaerobic Gram-negative bacteria are common among the resident flora of animals, and some are frequently associated with polymicrobial, necrosuppurative diseases. Taxonomy within this group of bacteria has evolved rapidly, but many species remain uncharacterized. The genus Porphyromonas was proposed 13 to accommodate 3 species of black colony-producing, anaerobic, Gram-negative bacteria that were formerly classified within the genus Bacteroides. There are currently 16 validly named Porphyromonas species. Porphyromonas gingivalis is a key pathogen in human periodontal disease, 4 whereas dog and cat gingival isolates, once identified as animal biotypes of P. gingivalis, were characterized as the new species Porphyromonas gulae, 5 which is also a recognized pathogen in canine periodontitis. A bacterin, consisting of Porphyromonas denticanis, P. gulae, and Porphyromonas salivosa (current name, Porphyromonas macacae), is now available as an aid in preventing and reducing the severity of this disease in dogs. 12 Recent phylogenetic analysis, 11 based on 16S ribosomal (r)RNA gene sequences, found that P. gingivalis–like isolates from Australian marsupials are grouped into 2 clades. One clade included the type strain of P. gulae as well as isolates from other nonhuman mammals. The second clade consisted of isolates that only originated from marsupials and was suggested 11 to represent a putative new marsupial Porphyromonas species. The current report describes the isolation and preliminary identification of a reptile-associated Porphyromonas variant that may represent a new species and is perhaps one of the first reports describing Porphyromonas-associated clinical infections in reptiles.
Patient 1 was a privately owned 8-year-old female central bearded dragon (Pogona vitticeps) that presented with a 4 × 2–cm left facial abscess, just ventral to the left mandible and including part of the “beard” (Fig. 1). There was no known history of trauma. After anesthesia with isoflurane, a followed by analgesia with butorphanol b (1.5 mg/kg administered once intramuscularly), and an aseptic surgical preparation of the skin, the abscess was lanced, debrided, and left open to heal by second intention. A portion of the interior lining of the abscess wall was sampled immediately after the abscess was lanced and was placed in a commercial transport medium c and submitted for aerobic and anaerobic bacterial cultures. After debridement, the wound was flushed repeatedly with a solution of povidone–iodine diluted 1:200 in sterile saline, and a silver sulfadiazine cream d was applied daily for approximately 3 weeks. There was complete recovery from the facial abscess, but 6 months later, the patient had a 4-day history of anorexia and a 1-day history of dehydration and depression. At presentation, there were palpable, firm, coelomic masses, with no associated vascular supply on ultrasonographic evaluation. Despite treatment with fluids and ceftiofur e (15 mg/kg subcutaneously [SQ]), the lizard died the next day. The pathology report indicated that the lizard had a fibrinous yolk coelomitis with intralesional filamentous bacteria. A culture was not performed.

Central bearded dragon (Pogona vitticeps). Patient 1 at time of surgical preparation to lance a 4 × 2–cm left facial abscess and collect a sample of the inner lining of the abscess for culture.
Patient 2 was an adult, female central bearded dragon of undetermined age that was obtained for research but that had not yet been used in any research. For the 6 months prior to its death, the lizard maintained a good appetite, was in fair body condition, maintained a body weight between 340 and 362 g, and was otherwise healthy. Approximately 1 week prior to death, the lizard exhibited weakness, anorexia, and weight loss (300 g). It had been treated with fenbendazole f and sulfadimethoxine g 2 months prior to death for a mild case of coccidiosis and pinworms in the colony. In the pathology report, the diagnosis was moderate, chronic multifocal granulomatous pneumonia with intralesional bacteria and eosinophilic intracytoplasmic inclusion bodies in many epithelial cells. The gallbladder contained many small choleliths ranging in size from 2 to 5 mm. Fresh tissue was not saved for virus isolation. A swab of the fluid found within a large bulla in the distal right lung was submitted for bacterial culture.
Patient 3 was an approximately 8-year-old, male central bearded dragon that was obtained for research and that had only been used once for phlebotomy purposes. After 6 months, it presented with a 15 × 5–cm area of thickened, darkened, and dry skin that occupied most of the dorsum. When the affected skin was lifted, pus and necrotic tissue were observed underneath. A swab containing exudates collected from the subcutaneous tissues beneath the skin scab was subjected to bacterial and fungal culture. After 7 weeks of daily wound cleaning with chlorhexidine scrub, h wound flushing with saline, topical application of amphotericin-B i (0.5 ml of 100 mg/ml) and then silver sulfadiazine, d and administration of injectable antibiotics (ceftiofur, e 15 mg/kg SQ every 5 days) and oral itraconazole j (5 mg/kg every 5 days), the lizard's skin wound had healed well. Two weeks after discontinuing medication, it was suddenly found upside down in the water dish, dead. At necropsy, there was a hepatic abscess, a sample of which was submitted for bacterial and fungal cultures.
Using standard methods, routine diagnostic cultures were performed by the University of Tennessee, College of Veterinary Medicine Clinical Bacteriology and Mycology Laboratory (Knoxville, Tennessee). Cultures from each patient yielded relatively high levels of polymicrobial bacterial growth, including high levels (>1,000 colonies) of an unidentified Gram-negative anaerobic rod. Other accompanying organisms were as follows: from patient 1, Pseudomonas aeruginosa (>100 colonies) and Morganella morganii (>100 colonies); from patient 2, Pseudomonas aeruginosa (>1,000 colonies); and from the initial culture of patient 3, P. aeruginosa (>1,000 colonies), M. morganii (>1,000 colonies), Enterococcus (>100 colonies), alpha hemolytic Streptococcus sp. (>100 colonies), a small aerobic Gram-positive rod (which was not further identified; >75 colonies), and a hyaline, Chrysosporium-like mold (few colonies).
The unidentified organism was first recognized in cultures from patients 1 and 2 as very small colonies surrounded by a large zone of complete (“beta”) hemolysis after 24–48-hr incubation in 7% CO2 at 35°C on Columbia agar c and colistin–nalidixic acid agar c plates, each of which contained 5% sterile sheep blood. k The initial isolate from patient 3 was observed among other organisms growing on phenylethyl alcohol agar c supplemented with 5% sterile defibrinated sheep blood k and incubated in an atmosphere containing 5% hydrogen, 10% CO2, and 85% nitrogen at 37°C. In cultures from patients 1 and 2, primary growth was also subsequently observed on Centers for Disease Control and Prevention (CDC) anaerobic blood agar a plates incubated in the anaerobic atmosphere described above. The isolates appeared microscopically as small, approximately 0.5–0.8-μm × 0.8–3-μm, Gram-negative rods and coccobacilli. The phenotypic appearance and biochemical reactions of the isolates from each patient were identical. The isolates grew in aerobic, elevated-CO2 and anaerobic atmospheres, with the best growth occurring in the latter atmosphere. Isolated colonies on CDC anaerobic blood agar (for 7 days) were convex, slightly glistening, light gray, and 1–2 mm in diameter. The isolates were catalase-positive and strongly hemolytic on sheep blood agar plates (Fig. 2). Black- or brown-pigmented colonies were not observed on any of the blood-containing media, including anaerobic laked Brucella blood agar containing 5% laked sheep blood l incubated for 14 days (Fig. 3). The isolates did not agglutinate sheep red blood cells in a microplate assay with a hemagglutinating strain of Bordetella bronchiseptica as a positive control. 2 The isolates produced a weak acidic reaction throughout triple sugar iron agar slants incubated aerobically at 37°C and were urease negative and spot indole positive. Susceptibilities to special potency antibiotic disks were determined on CDC anaerobic blood agar plates incubated anaerobically for 3 days. No visible zones of growth inhibition were observed surrounding colistin (10 μg), vancomycin (5 μg), kanamycin (1,000 μg), or nalidixic-acid (30 μg) disks. c

Centers for Disease Control and Prevention anaerobic blood agar plate showing isolated hemolytic colony of the variant Porphyromonas sp. after 7 days of anaerobic incubation at 37°C.
Reactions of each isolate in a commercial biochemical test kit m designed for anaerobic bacteria gave the microcode (034747) that identified the isolates as P. gingivalis with 99.9% probability. However, the biofrequencies of the microcode were considered very rare, rendering the microcode questionable and the probabilities unreliable. Re-isolation and repeat testing were recommended in each case. Positive test results included ortho-nitrophenyl-beta-D-galactosidase, alpha D-glucosidase, n-acetyl-beta-D-glucosidase, alkaline phosphatase, leucyl-glycine arylamidase, glycine arylamidase, arginine arylamidase, serine arylamidase, pyrrolidonyl arylamidase, and indole production. Negative test results were observed for urease, beta-D-disaccharidase, alpha-L-arabinosidase, beta-D-glucosidase, alpha-D-galactosidase, alpha-L-fucosidase, proline arylamidase, and phenylalanine arylamidase. Three of these test reactions (ortho-nitrophenyl-beta-D-galactosidase, usually negative [98%]; alpha-D-glucosidase, usually negative [100%]; and glycine arylamidase, usually negative [81%] for P. gingivalis) were repeatedly nonsupportive of the identification of the isolate as P. gingivalis. However, a reaction in 1 of 3 such tests (ortho-nitrophenyl-beta-D-galactosidase, reported positive) was consistent with that reported for the type strain of P. gulae. The commercial identification system did not contain reactions for P. gulae in its database.

Anaerobic Brucella blood agar (LBA) after 3 days of anaerobic incubation at 37°C. Beginning at upper left and proceeding counterclockwise, quadrants contain the following: variant Porphyromonas isolates from patients 1–3, respectively, and an arbitrarily selected clinical isolate showing the black, iron protoporphyrin pigment that is typically associated with many species of Porphyromonas.
Species identification was further pursued with each isolate by partial 16S rRNA gene DNA sequencing. Template DNA was extracted from individual colonies by physical disruption with glass beads n and subjected to polymerase chain reaction (PCR) with universal eubacterial primers designed to amplify a portion of the 16S rRNA gene. 14 The primers, Broad 1 (GCGGATCCTGCAGAGTTTGATCCTGGCTCAG) and Broad 2 (GGCTCGACCGGGTTACCTTGTTACGACTT), correspond to bases 8–27 and 1492–1510 of the Escherichia coli small ribosomal subunit gene. Polymerase chain reactions were performed with a commercial Taq polymerase premix o containing reaction buffer and nucleotides. Thermocycler conditions included 1 cycle of denaturation and enzyme activation at 95°C for 1.5 min, 30 cycles of annealing at 50°C for 30 sec, extension at 72°C for 2.5 min, and denaturation for 1 min at 94°C. This was followed by a final cycle of annealing at 50°C for 2 min, extension at 72°C for 5 min, and holding at 4°C. The PCR amplification products were subjected to electrophoresis in 1% agarose gel containing 1 μg/ml of ethidium bromide and were visualized by ultraviolet transillumination. The PCR products were of expected size, and single-strand DNA was removed by exonuclease treatment p before submission to the University of Tennessee Molecular Biology Resource Facility (Knoxville, Tennessee) for bidirectional sequencing with the PCR primers. Nucleotide sequence data were analyzed using a commercial software program. q Alignment of the sequences obtained from each isolate showed that they shared 100% identity. The 16S rRNA gene sequences of the isolates were compared to sequences in the GenBank database using the BLASTn algorithm (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi). 1 The best match to a recognized species, with 90% nucleotide similarity over 1428 nt, was to P. gulae (4 strains, including type species).
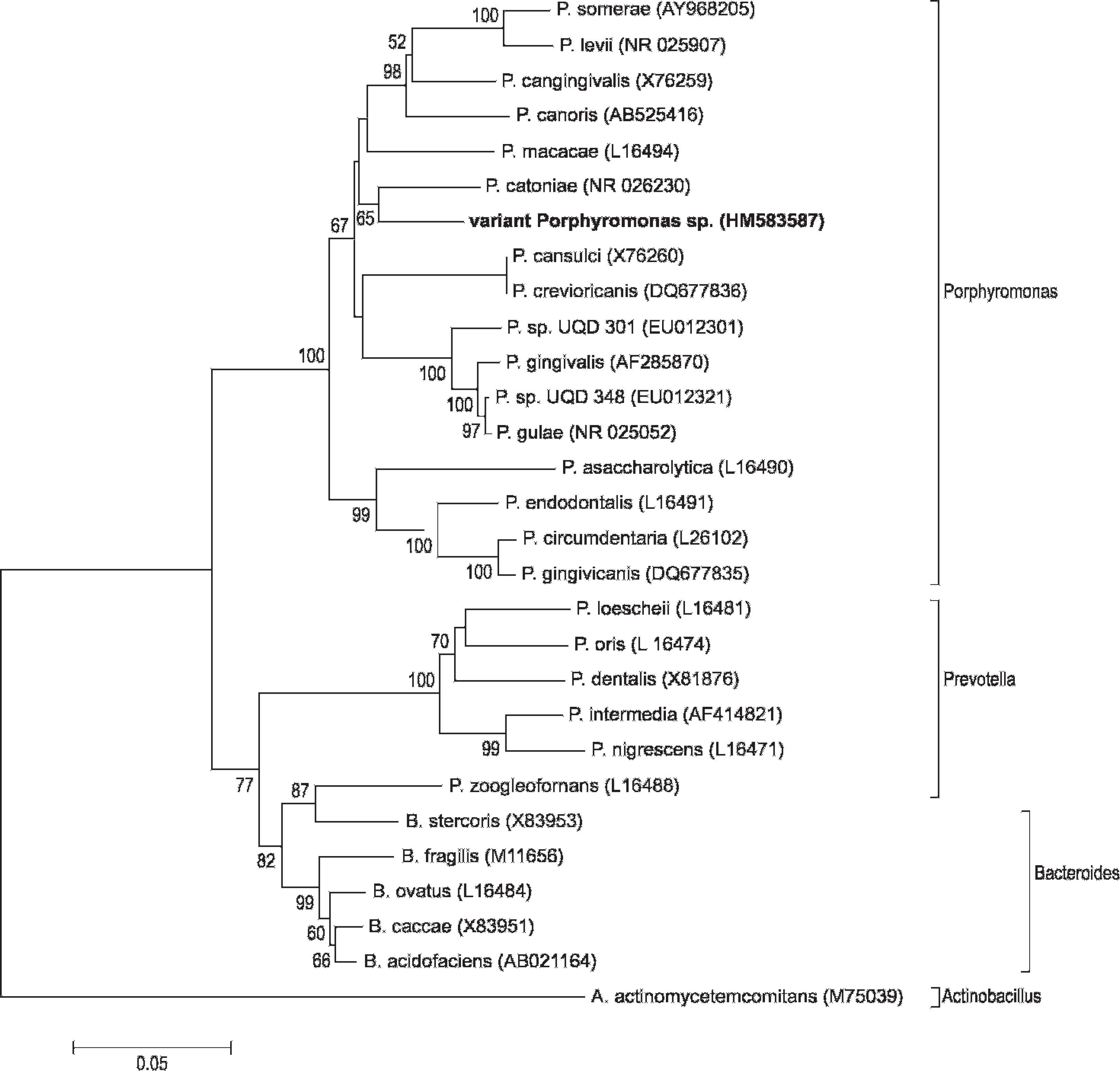
16S ribosomal RNA gene sequence–based phylogenetic tree of central bearded dragon variant Porphyromonas sp. and related GenBank entries from the family Bacteroidaceae. Sequence accession numbers are given in parentheses. The tree was generated by the neighbor-joining method, and the bootstrap values, shown at the branches, were calculated from 1,000 replicates. Evolutionary analyses were conducted in Molecular Evolutionary Genetics Analysis (MEGA) software version 5.0. r
The P. gulae type species was described as obligately anaerobic and hemagglutinating (but with no mention of having hemolytic activity); it was also described as producing brown to black pigment on aging of the cultures. 5 The isolates described in the present study were initially recognized as a result of their strong hemolytic activity on blood agar incubated in an aerobic atmosphere with elevated CO2. Hemolytic activity is a property of many P. gingivalis strains and has been observed in some Porphyromonas catoniae strains. 3,19 Evidence of oxygen metabolism and growth in aerobic conditions has also recently been observed in P. gingivalis. 9 Naturally occurring, nonpigmented strains of Porphyromonas have been reported in several species 17,18 and are consistently found in the species P. catoniae. 19 The present isolates did not have detectable hemagglutinating activity; however, in P. gingivalis, hemagglutination, hemolysis, and pigment formation are properties associated with an integrated iron capture system mediated by multiple domains within surface arginine and lysine cysteine proteases known as gingipains. 8,10
Isolates in the current study from central bearded dragons were most closely related to members of the genus Porphyromonas but differed significantly from existing species. The bearded dragon isolates had 91% 16S rRNA gene similarity with P. gulae in an initial BLAST search and were, thus, more divergent than a recent collection of marsupial Porphyromonas strains that were considered a potential new species, with 94.9% 16S rRNA gene similarity to P. gulae. 11 A phylogenetic tree generated from 16S rRNA gene sequences (1409 nt) from the bearded dragon isolates and related Bacteroidaceae indicated a possible closer relationship to P. catoniae than to P. gulae (Fig. 4). Direct alignment of 16S rRNA sequences from the bearded dragon isolates with those of type strains of P. catoniae and P. gulae indicated an equivalent nucleotide similarity (90%) to both species. Sequence from a representative isolate (10–1288) has been submitted to the GenBank database and assigned the accession number HM583587.
Many Porphyromonas spp. are animal parasites; thus, phylogenetic diversity seen within species of this genus may parallel the phylogenetic diversity of their animal hosts. Descriptions of characterized Porphyromonas isolates from nonmammalian sources are lacking in the veterinary medical literature. 15 The extent of their distribution in healthy bearded dragons or other reptiles is unknown. The sources of infection presented in the current report were not known but may have been secondary to underlying factors such as trauma, aspiration, and chronic fungal infection in patients 1–3, respectively. Periodontal disease was not noted in the patients and was not evident in remaining bearded dragons in the affected research colony. Gingival swabs collected from patient 3 prior to its death and from 7 other apparently healthy bearded dragons yielded 2 isolates (including one from patient 3) that were subsequently confirmed to be the same Porphyromonas variants. An identical organism was also recovered from the hepatic abscess observed at necropsy in patient 3. To date, multiple isolates from 6 bearded dragons (5 of which have died) have been obtained by the authors of the present study. Although isolates of the Porphyromonas species identified in the current study were associated with infections in bearded dragons, the study did not establish if these isolates were the primary causative agents of the infections reported. In fact, isolation of the same variant of Porphyromonas species from healthy bearded dragons indicates that these are probably residents of normal microflora. Whether this species is capable of establishing primary necrosuppurative infections in bearded dragons has yet to be determined. Employing subcutaneous chamber and oral challenges, several animal models have been used to compare virulence characteristics among other Porphyromonas species. 6 Additional phenotypic and genotypic characterization is required to determine if the bearded dragon isolates represent a different species.
Resistance to colistin, vancomycin, kanamycin, and nalidixic acid, as observed with isolates in the present study, is common among the Bacteroides fragilis group of anaerobic Gram-negative bacteria. 7 Porphyromonas spp. are unique among Gram-negative bacteria in that they are customarily susceptible to special potency vancomycin disks, and this trait is useful for their preliminary identification. 16 However, a naturally occurring, nonpigmented variant of Porphyromonas endodontalis was 10 times more resistant to vancomycin than were typical strains of this species, while having equivalent susceptibilities to 21 other antimicrobials. 17
The present report indicates that the unique Porphyromonas sp. may be a significant, albeit opportunistic, bacterial pathogen in necrosuppurative diseases of central bearded dragons. Veterinary microbiologists should be alerted to the possible identification of similar hemolytic, aerobically growing, nonpigmented Porphyromonas sp. in clinical samples from reptiles and should report their occurrence to allow further analyses of host range and pathogenicity.
Acknowledgements. The authors thank Brian Johnson, Rupal Brahmbhatt, Randall Kania, and Anik Vasington for technical assistance.
Footnotes
a.
IsoFlo®, Abbott Laboratories, Abbott Park, IL.
b.
Torgugesic® (10 mg/ml), Fort Dodge Animal Health, Fort Dodge, IA.
c.
BBL™, BD Diagnostic Systems, Sparks, MD.
d.
Silver sulfadiazine 1% cream, PAR Pharmaceutical Companies Inc., Woodcliff Lake, NJ.
e.
Exceed®, ceftiofur crystalline-free acid (200 mg/ml), Pfizer Inc., New York, NY.
f.
Panacur®, suspension 10%, Intervet Inc., Millsboro, DE.
g.
Sulfadimethoxine, oral suspension 5%, Pfizer Inc., New York, NY.
h.
ChlorHex-Q Scrub®, chlorhexidine scrub, Vedco Inc., St. Joseph, MO.
i.
Novaplus®, amphoterocin B, 50-mg bottle diluted with 10 ml saline (5 mg/ml), X-Gen Pharmaceuticals Inc., Big Flats, NY.
j.
Itraconazole (10 mg/ml suspension), Centocor Ortho Biotech Inc., Horsham, PA.
k.
Hemostat Laboratories, Dixon, CA.
l.
Anaerobic laked Brucella blood agar (LBA; catalog no. 145), Anaerobe Systems, Morgan Hill, CA.
m.
RapID ANA II System, ERIC version 1.0.771, Remel Inc., Lenexa, KS.
n.
BioSpec Products Inc., Bartlesville, OK.
o.
Premix Taq™, Takara Bio Inc., Otsu, Shiga, Japan.
p.
ExoSAP-IT, USB Corp., Cleveland, OH.
q.
Lasergene® v7.2, DNASTAR Inc., Madison, WI.
