Abstract
Chronic dermatomycosis was identified in 3 central bearded dragons (Pogona vitticeps), held as companion animals by the same owner. Clinical signs of dermatomycosis included subcutaneous masses as well as crusty, erosive, and ulcerative skin lesions. The facial region was affected in 2 of the 3 cases. Masses were surgically excised, and histology confirmed necrotizing and granulomatous inflammatory processes associated with fungal hyphae. Two of the bearded dragons were euthanized because of their deteriorating condition. In both cases, postmortem histology confirmed systemic fungal infections despite treatment of 1 animal with itraconazole. In the third bearded dragon, therapy with voriconazole at 10 mg/kg was initially effective, but mycotic lesions reappeared 15 months later. Nannizziopsis chlamydospora was identified by PCR and subsequent DNA sequencing in 2 of these cases.
Dermatomycosis associated with Nannizziopsis sp. is documented in lizards, terrestrial and aquatic snakes, and crocodiles. 6 Hyphomycete fungi of the family Nannizziopsiaceae isolated from bearded dragons include N. chlamydospora, N. draconii, N. barbata, N. guarroi, and N. vriesii.1–7,9 Clinical signs include swellings, subcutaneous masses, and skin lesions.1–5,9 Antifungal treatments were not always successful in resolving the skin lesions, or skin lesions recrudesce, resulting in the animals being euthanized and, at postmortem, systemic fungal infections were occasionally identified.2,4,5 Three central bearded dragons (syn. inland bearded dragon; Pogona vitticeps) from the same owner were privately held as companion animals. Two bearded dragons were male and 1 was female and all were 6 years of age. They were habituated in 2 terrariums (2.2 m2 and 1.2 m2, photoperiod 10–12 h, ultraviolet source, temperature 23–40°C, cage substrate sand), fed insects twice a week, vegetables and fruit daily, and supplemented with a calcium and vitamin powder once a week. Males were housed separately, and alternately paired with the female bearded dragon. The 3 lizards were presented to the clinic because of chronic skin problems. One male bearded dragon (case 1) had crusty skin lesions on the lower mandible and a subcutaneous mass near the left shoulder; the second male bearded dragon (case 2) also had crusty, erosive, and ulcerative skin lesions on the lower mandible; and the female bearded dragon (case 3) had a subcutaneous mass near the left shoulder (Fig. 1). A routine fecal examination revealed Isospora amphiboluri and flagellates, resulting in the treatment of all 3 bearded dragons with toltrazuril (10 mg/kg orally) a and metronidazole (40 mg/kg orally) b twice within a 2-week interval.

Female bearded dragon (Pogona vitticeps; case 3) with a mass near the left shoulder. Image courtesy P. Scholze.
The subcutaneous masses were surgically excised in cases 1 and 3, and histological examination of the masses revealed necrotizing and granulomatous dermatitis and panniculitis associated with fungal hyphae. Case 1 was treated with itraconazole (5 mg/kg, orally every 48 h) c for 4 months and topical iodine solution, d and skin lesions improved; however, 6 months later, the skin lesions recurred. The condition of cases 1 and 2 deteriorated, and they were euthanized.
Postmortem examination revealed granulomatous masses in liver, heart, and lungs in both cases. Case 1 additionally had granulomatous masses with intralesional fungal hyphae in kidneys, tongue, and intestine, as well as in the celomic serosa near the surgical scar at the left shoulder. Large numbers of fungal hyphae could also be seen inside blood vessels of numerous organs. Case 2 additionally had multifocal chronic deep granulomatous dermatitis with necrosis and ulceration and intralesional fungal hyphae (Fig. 2). Bacterial and fungal culture of a skin lesion from case 2 yielded moderate numbers of Enterococcus sp. and low numbers of Nannizziopsis sp. In case 3, therapy with voriconazole (10 mg/kg, orally every 24 h) e was administered. Serum biochemistry performed on a blood sample taken from the tail vein showed no significant deviations from reference intervals. To evaluate whether the antimycotic treatment could be stopped, a skin swab was taken on day 32 of voriconazole treatment. Mycologic examination revealed low numbers of Nannizziopsis sp. Later, a skin swab was negative, so voriconazole treatment was stopped on day 99. One year later, the lizard was free from new skin lesions, and the result of fungal culture of a skin swab was negative. However, 3 months later, the bearded dragon was presented again with a small mass near the operation site on her left shoulder. The mass was removed again, and isolates were consistent with Nannizziopsis sp. The voriconazole treatment was started again and is ongoing at the time of manuscript writing.
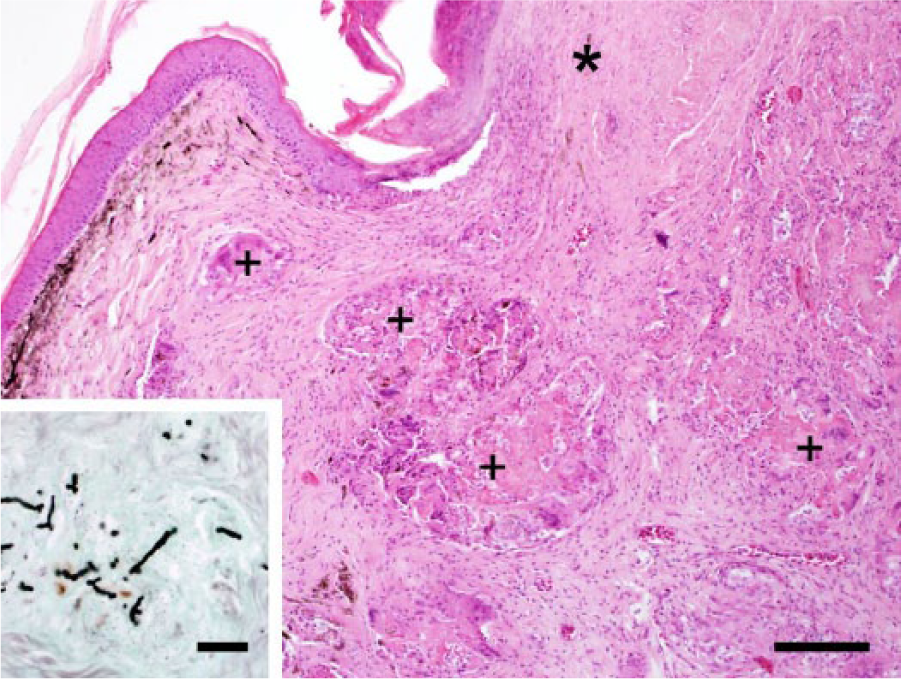
Male bearded dragon (Pogona vitticeps; case 2). Chronic fungal dermatitis with granulomas (+) surrounded by dermal fibrosis, and focal necrosis of epidermis and dermis (*). Hematoxylin and eosin. Bar = 150 μm. Inset: dermal granuloma with stained septate and branching fungal hyphae. Grocott methenamine silver. Bar = 40 μm.
The isolation of bacteria and fungi from cases 2 and 3 was performed as described previously. 5 After cultivation at 28°C for 14 days on Sabouraud dextrose agar with gentamicin and chloramphenicol, f the isolates were regrown on the same medium under the same conditions. Finally, DNA extraction and PCR amplification of the first and second ribosomal internal transcribed spacer regions (ITS1, ITS2), 5.8S ribosomal (r)RNA gene, D1 and D2 domains of the 28S rDNA (D1-D2), and a fragment of actin were amplified as described before.5,7 BLASTn with default settings (http://www.ncbi.nlm.nih.gov/BLAST) was used to compare obtained sequences to nonredundant DNA database sequences. The sequences were aligned with the most similar sequences of established species deposited in GenBank in order to define the levels of relatedness. 8 The same software was used for phylogenetic analyses. Maximum-likelihood tree based on Tamura 3-parameter–corrected nucleotide distances obtained from the combined DNA sequence data from 3 loci (D1-D2, actin and ITS) with 1,000 bootstraps was used to reconstruct phylogenetic trees. Comparison with corresponding actin and ITS sequences showed that both isolate sequences shared high similarity scores (>99%) with N. chlamydospora strains UTHSC 04-2056 and UTHSC 06-1419 (HF547879, HF547881, HF547871, NR_111522). Comparison with corresponding 28S rRNA sequences revealed highest DNA sequence similarities with N. vriesii strain CBS 407.71 (KC989736) and ATCC 22444 (AY176715), but none of the 28S rRNA sequences belonging to distinct Nannizziopsis species was deposited in GenBank. Nevertheless, analysis of actin and ITS1-5.8S-ITS2 sequences of both fungal isolates unequivocally demonstrated its affiliation with N. chlamydospora strains. The partial nucleotide sequences of actin (KR912175, KR912176), ITS1-5.8S-ITS2 (KR349443, KR349444), and D1-D2 (KR063659, KR063660) were deposited in GenBank (Fig. 3).
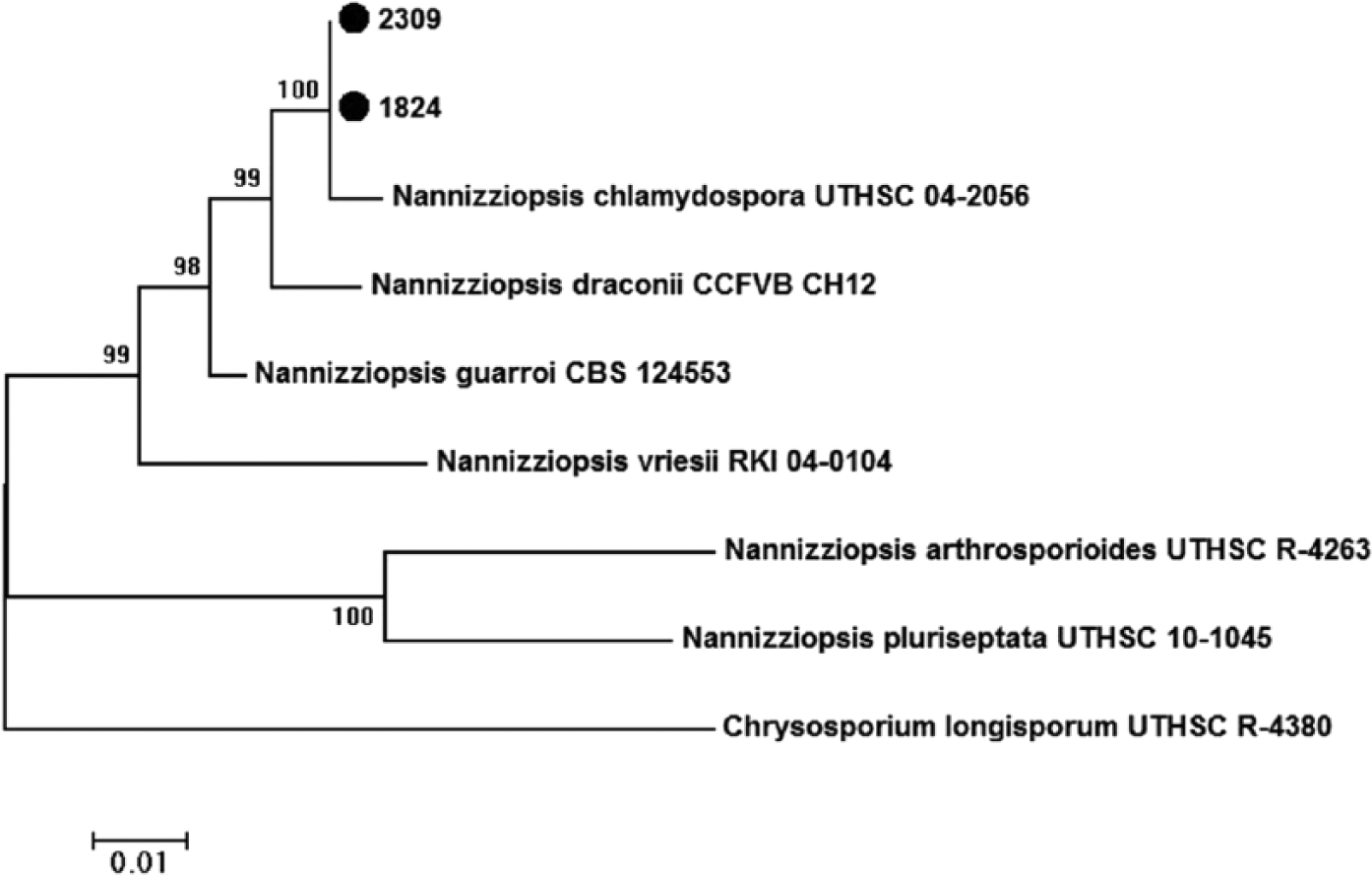
Maximum-likelihood tree based on Tamura 3-parameter corrected nucleotide distances obtained from the combined DNA sequence data from 3 loci (D1-D2, actin, and ITS). Bootstrap values (expressed as percentages of 100 replications) >75% are shown at branching points. The sequence generated during this study was labeled (●).
Dermatomycosis in bearded dragons caused by members of the family Nannizziopsiaceae is reported in Europe, the United States, and Australia.1–5,9 Captive-born bearded dragons are affected as well as wild-caught animals. 4 Clinical signs include swellings and discolored, crusty, ulcerative to necrotic skin lesions of the facial region, body, limbs, or tail.1–5,9 Clinical signs of dermatomycosis in the present 3 cases were similar to skin lesions caused by other Nannizziopsis species,1–5,9 and the site of infection also included the facial region in 2 cases. Identification of N. chlamydospora by PCR and subsequent DNA sequencing in 2 cases strongly suggests that this fungal species was the causative agent of the dermatomycosis. The source of infection in these cases remains unknown, but it is very likely that environmental management played a role in the development of the infections and potentially in the relapse, because the owner had not changed the cage substrate on a regular basis. However, the relapses could also be explained by treatment failure. Despite antifungal therapy with itraconazole or voriconazole, relapses were observed after 6 and 15 months, respectively. Because of the chronic nature of this type of dermatomycosis, regular clinical examination by the owners for early detection of skin lesions or swellings is recommended. Animals with suspicious skin lesions should quickly be isolated from the group and veterinary medical attention sought to confirm the diagnosis in order to maximize treatment success.
Footnotes
Acknowledgements
We thank P. Scholze for kindly providing Figure 1 included in this article.
Authors’ note
S Schmidt-Ukaj and I Loncaric contributed equally to this work.
Authors’ contributions
S Schmidt-Ukaj and I Loncaric drafted the manuscript and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All authors critically revised the manuscript and gave final approval. S Schmidt-Ukaj contributed to conception and design of the study and contributed to acquisition, analysis, and interpretation of data. I Loncaric contributed to conception and design of the study and contributed to analysis and interpretation of data. J Spergser contributed to analysis of data. B Richter contributed to conception and design of the study and contributed to acquisition and analysis of data. M Hochleithner contributed to acquisition, analysis, and interpretation of data.
a.
Baycox (25 mg/mL solution), Bayer Austria, Vienna, Austria.
b.
Anaerobex film-coated tablets (500 mg), G.L. Pharma GmbH, Lannach, Austria.
c.
Sporanox capsules (100 mg), Janssen-Cilag Pharma GmbH, Vienna, Austria.
d.
Betaisodona solution standardized, Mundipharma, Vienna, Austria.
e.
V-fend film-coated tablets (200 mg), Pfizer Ltd., Kent, England.
f.
Sabouraud dextrose agar with chloramphenicol and gentamicin, BBL, Heidelberg, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
