Abstract
A 15-d-old female crossbreed calf was referred because of paraplegia since birth. Clinical examination revealed a skin defect covered by hair on the dorsal midline in the thoracic area of the spine. Thoracolumbar spinal cord neuroanatomic localization was determined based on neurologic examination. Computed tomography of the thoracolumbar spine revealed incomplete fusion of the vertebral arches from T6 to T10 and duplication of the vertebral arch of T7. At the level of T6-T7, duplication of the spinal cord with 2 segments completely separated by a septum of hyperattenuating, probably cartilaginous, tissue was noted. Histologically, the spinal segments had different degrees of duplication. Three central canals were detected in one region. Genetic investigation for the presence of methylenetetrahydrofolate reductase (MTHFR) polymorphism, which has been investigated in both human and veterinary medicine as a possible cause of neural tube defects and abortion, was carried out and was negative in both the calf and her dam.
A 15-d-old female crossbreed calf was referred to the Mobile Clinic Service, Veterinary Teaching Hospital (VTH), Department of Veterinary Science of Turin (Italy) because of inability to stand on the pelvic limbs since birth. Because of podalic presentation, she had been delivered by Cesarean section performed without complications by the referring veterinarian. The dam had given birth to other normal calves previously; pedigree information about the sire was unavailable.
Clinical examination, performed in the field, revealed a skin defect covered by hair on the dorsal midline in the thoracic area of the spine. After clipping and cleaning of the area, the lesion was seen to be ~10 cm long surrounded by severely inflamed, hyperemic, and edematous tissues. Physical examination revealed other malformations including mild lateral mandibular deviation and mild arthrogryposis affecting both thoracic limbs. Neurologic examination performed by a board-certified neurologist (A D’Angelo) revealed a moderately obtunded mental status. The calf was able to stand on thoracic limbs if supported, but the pelvic limbs were plegic. Postural reactions were normal on both thoracic limbs. Cranial nerve examination was normal, except for a bilaterally absent menace response presumably because of the animal’s young age. Patellar, cranial tibial, flexor withdrawal, and perineal reflexes were all normal in both pelvic limbs. Carpal extensor and withdrawal reflexes in both thoracic limbs were difficult to evaluate because of tendon contracture, but were considered to be normal. No pain on palpation of the paraspinal muscles was noted. Thoracolumbar spinal cord neuroanatomic localization was diagnosed based on the neurologic examination; further possible secondary intracranial involvement was suspected on the basis of mental status. Complete blood count (CBC) showed mild neutrophilia (7.64 × 109/L, reference interval [RI]: 1.1–3.6 × 109/L) probably as a result of inflammation at the site of the defect. The biochemistry profile was otherwise unremarkable.
Computed tomography (CT; Highspeed Fx/i CT, GE Healthcare, Little Chalfont, UK) revealed incomplete fusion of the vertebral arches from T6 to T10, associated with duplication of the vertebral arch of T7. The spinous processes appeared completely merged at the level of T11. The vertebral canal was elliptical at the level of T7, where the transverse diameter was increased from 2.2 cm to 2.57 cm and the spinal cord duplicated, with 2 segments completely separated by a septum of hyperattenuating (+111 HU), probably cartilaginous, tissue (Fig. 1). Iodinated nonionic contrast medium (Iomeron 400, Bracco, Milan, Italy) was administered by lumbosacral puncture. The post-contrast phase showed homogeneous distribution of the contrast medium up to T6. From this point to 3.2 cm cranially, the contrast medium surrounded the 2 spinal cord segments, enhancing 2 different subarachnoid spaces, and flowed dorsally from the right spinal cord segment to the skin, creating the appearance of a dermoid sinus associated with a meningocele (Fig. 2).
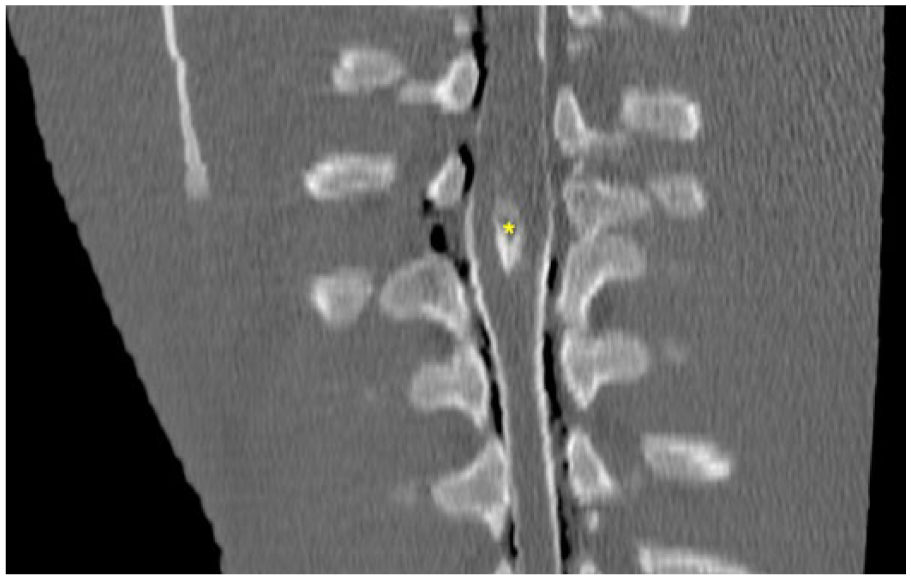
Myelo-CT dorsal reconstruction of the thoracic spine of a calf with a neural tube defect. Asterisk (*) indicates the fibrocartilaginous septum dividing the spinal cord into 2 hemicords.
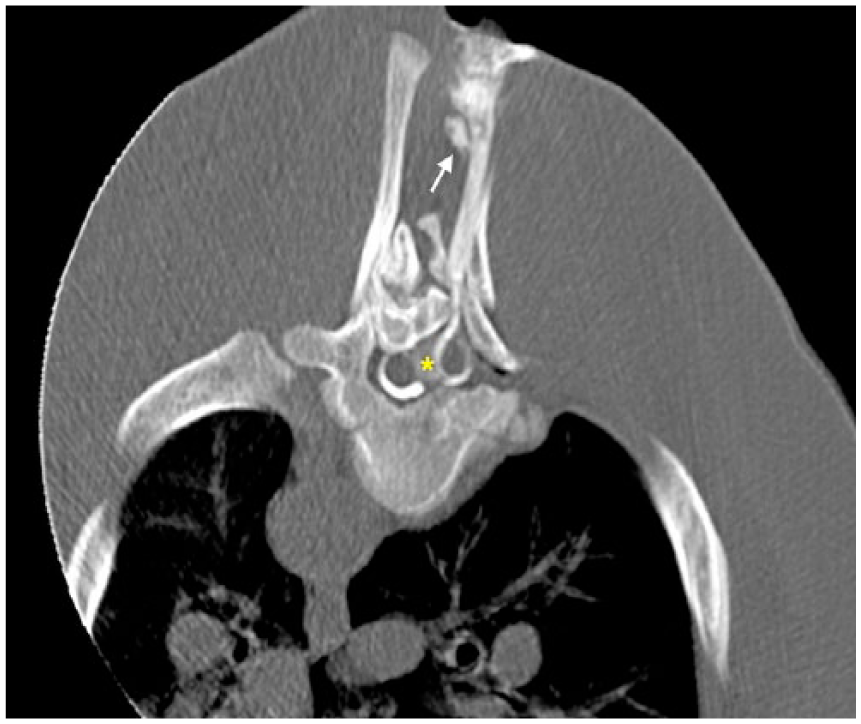
Transverse myelo-CT at the level of T6-T7 of a calf with a neural tube defect. Asterisk (*) indicates the fibrocartilaginous septum dividing the spinal cord into 2 segments. Hemicords are surrounded by contrast medium. Arrow indicates the outflow of the contrast medium towards the skin.
A cerebrospinal fluid (CSF) sample was collected by lumbosacral puncture just before iodinated nonionic contrast medium administration. The sample was slightly yellow but clear at gross evaluation. CSF analysis revealed increased total microprotein concentration (5.38 g/L, RI: <0.4 g/L), increased total nucleated cell count (0.11 × 109/L, RI: 0–0.01 × 109 /L), and marked blood contamination with a total erythrocyte count of 0.5 × 109 cells/L (RI: 0/L). Differential leukocyte count revealed mixed mononuclear pleocytosis with numerous activated vacuolated macrophages (61%) and occasional leukophagocytosis, activated lymphoid cells (30%), and neutrophils (9%).
The calf was euthanized given the poor prognosis, and postmortem examination was carried out. Upon dissection of the skin, the cutaneous lesion led to an oval opening into the vertebral canal at the level of T6-T7. After removing the spinal cord, a thick fibro-cartilaginous septum was noted to arise ventrally from the dura mater at level T6-T7, penetrate into the center of the spinal cord, and split the cord into 2 hemicords. At gross examination, the spinal cord was progressively enlarged at the level of T6, with complete duplication of the cord surrounding a central defect at the level of T7 and thickening of the meninges closely adherent to the bone (Fig. 3). Brain and spinal cord were fixed in 10% neutral-buffered formalin, processed routinely, and stained with hematoxylin and eosin for histology. The spinal cord was transversally sectioned at several levels to inspect it for macroscopic and microscopic lesions. The spinal segments showed different degrees of duplication. Partial fusion of 2 spinal cords with central compression and deformation was noted in the rostral section (at the level of T6) where 2 normally organized hemicords, each with a central canal, were detected (Fig. 4, section a). Spinal cord duplication was more evident caudally, with 2 smaller and regularly organized hemicords (Fig. 5, section b). Moving caudally, histologic duplication of the spinal cord was complete where it was separated into 2 well-organized but atrophic sections (Fig. 6, section c). Severe disorganization of the neuroparenchyma was noted at the level of T7-T8 on transverse section after resolution of the split into 2 segments. Here, partial tripartition of the spinal cord was suspected given the presence of 3 central canals, 2 completely, and 1 irregularly but not completely, surrounded by ependymal cells. Disseminated nonsuppurative perivascular cuffs with foci of neovascularization and gliosis were detected in the neuroparenchyma (Fig. 7, section d). This segment of the spinal cord had severe necrosuppurative meningitis, possibly related to bacterial infection because of the direct connection between the central canal and the environment. Multifocal meningeal fibrosis was also observed, particularly in the section caudal to the area of cord duplication. The inflamed soft tissue surrounding both hemicords represented a portion of the fibrocartilaginous septum observed on the CT images and arising from the ventral dura mater. Moderate, diffuse chronic meningitis associated with nonsuppurative perivascular cuffs was detected in the white and gray matter of the cerebellum and brainstem, particularly in the submeningeal and subventricular areas. No lesions were detected in the other parts of the central nervous system.
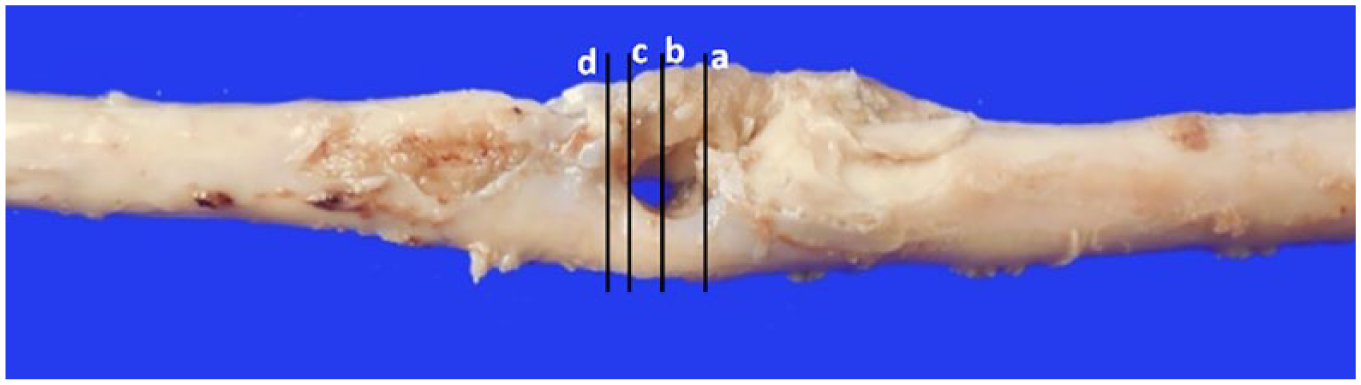
Dorsal view of the enlarged spinal cord with progressive duplication in 2 segments at the level of T7 of a calf with a neural tube defect. The central “hole” corresponds to the midline fibrocartilaginous spur observed in CT images. a, b, c, d = levels of inclusion corresponding to the histologic sections of Figs. 4–7; level “a” is most cranial.
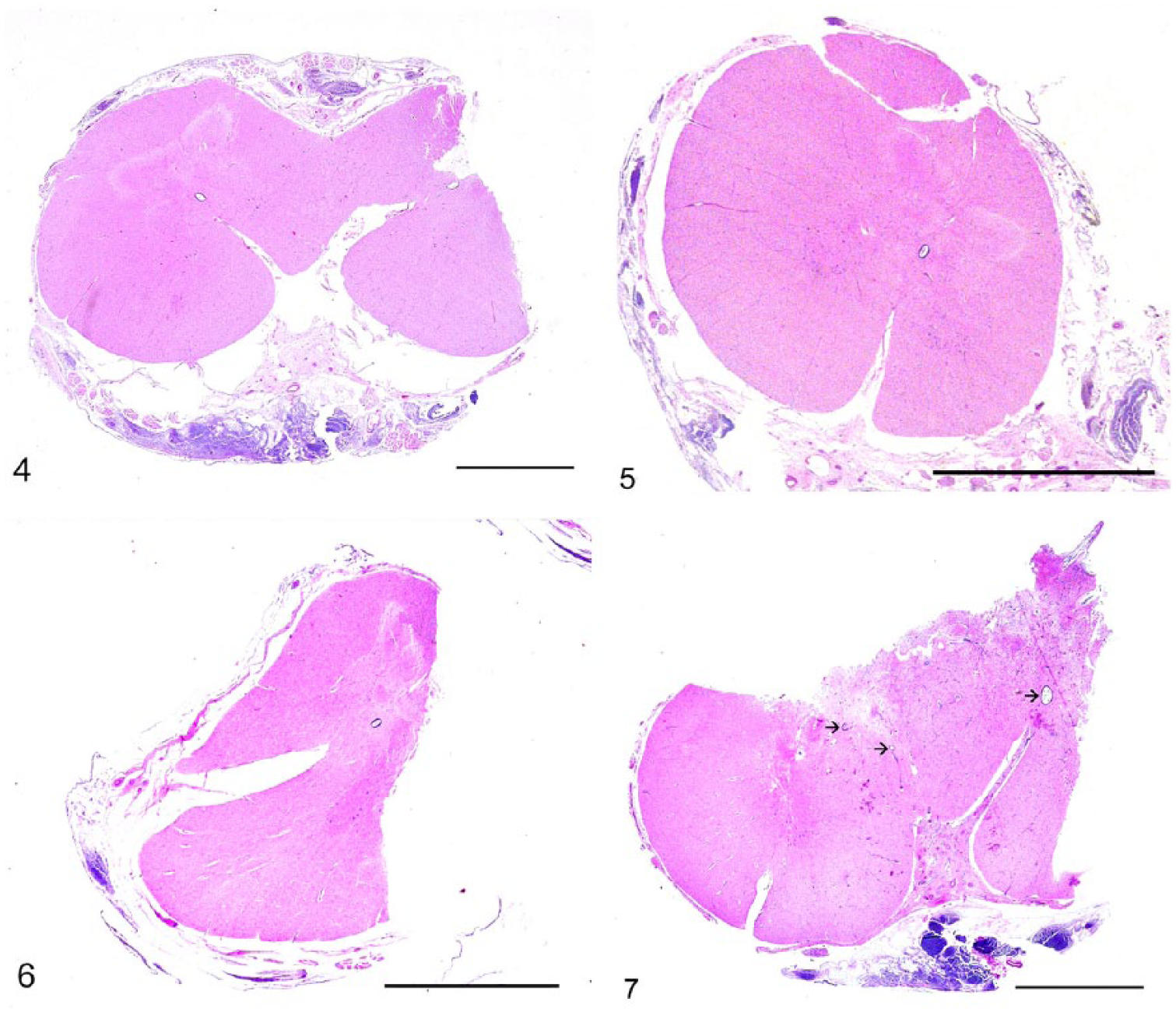
Histologic sections of the spinal cord of a calf with a neural tube defect. H&E. Bars = 5 mm.
A CSF sample was submitted for the detection of Toxoplasma gondii [by means of nested real-time PCR (rtPCR)] and of Neospora caninum (by means of simplex rtPCR). A spleen sample of the calf taken during autopsy was submitted for the detection of Schmallenberg virus and bovine viral diarrhea virus by reverse-transcription rtPCR. These investigations were carried out at the Istituto Zooprofilattico Sperimentale del Piemonte e della Valle d’Aosta; results were negative.
In order to exclude, as reported in human medicine,1,4,10 correlation of the malformations with deficiency of methylenetetrahydrofolate reductase (MTHFR), and subsequently of folate, further genetic investigation was carried out based on a previous study. 12 A muscle sample of the calf was taken during autopsy and stored at −80°C; a muscle sample of the dam was also collected at slaughter. Genomic DNA was obtained from muscle (NucleoSpin tissue kit, Macherey-Nagel, Düren, Germany) according to the manufacturer’s protocol. DNA purity was evaluated by absorbance readings on an ultraviolet spectrophotometer (NanoDrop 2000, Thermo Fisher Scientific, Waltham, MA). The primer set reported previously was used to amplify a fragment of MTHFR exon 4. 12 The primers for exon 7, designed on the bovine genomic sequence AC_000173.1, were TGGAGGCCATTGTCTGGAGTAT (forward) and CGAGAGGTAGTGGGCAAAGA (reverse). Real-time PCR reactions were performed in 25-μL volumes consisting of 0.03 U/μL of HotStarTaq DNA polymerase (Qiagen, Hilden, Germany), 0.2 mM each of deoxyribonucleotide triphosphate, 0.5 μM of each primer, and 50–100 ng of DNA template. The rtPCR profile consisted of an initial activation step at 95°C for 15 min, followed by 35 denaturation cycles at 95°C for 60 s, annealing at 54°C for 60 s, and extension at 72°C for 60 s. A final extension step of 72°C for 7 min was added to all reactions. Amplifications were carried out (GeneAmp PCR system 2720 thermal cycler, Thermo Fisher Scientific, Life Technologies Italia, Monza, Italy). Amplicons were resolved on 2.0% agarose gel. Amplified fragments were cycle sequenced (ABI Prism 310 genetic analyzer, ABI Prism BigDye terminator v.1.1, Thermo Fisher Scientific) by the dideoxy chain termination method with fluorescent dye terminators. Sequencing on both strands was performed using the PCR primer. The resulting sequences were compared and aligned with the human messenger RNA sequence U09806.2 using the NCBI BLASTn suite-2 sequences software (https://blast.ncbi.nml.nih.gov/Blast.cgi); 198-bp and 205-bp fragments were generated from exons 4 and 7 of MTHFR, respectively. The amplicon sequences were compared and aligned with corresponding human sequences to identify the position corresponding to C677T and A1298C (Figs. 8, 9). Both calf and dam were homozygous for the 2 single nucleotide polymorphisms (SNPs) showing the genotypes CC and AA for the normal alleles. Our results for exon 4 are in agreement with previous investigations. 12 Regarding the mutation in exon 7, a C/T polymorphism has been reported in Holstein cows in China 12 in position 1484 (NM_001011685.1), corresponding to position 1308 in the human sequence, whereas no polymorphism was found at nucleotide 1474 (NM_001011685.1), corresponding to 1298 in the human sequence.

Fragment of MTHFR exon 4 aligned with the human reference messenger RNA sequence. The boxes correspond to the mutation site.
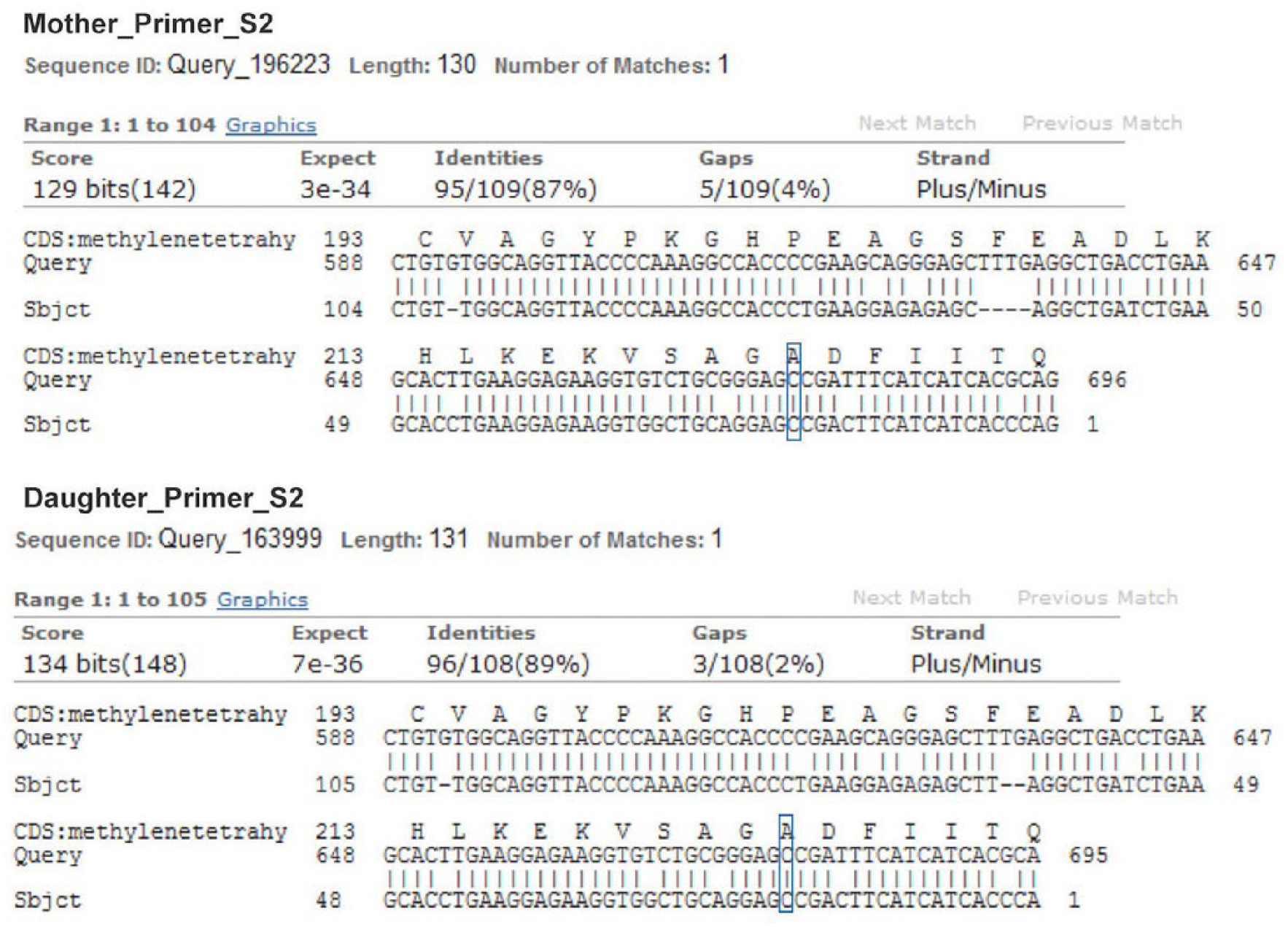
Fragment of MTHFR exon 7 aligned with the human reference messenger RNA sequence. The boxes correspond to the mutation site.
In our calf, based on the results of CT investigation in association with autopsy, a condition of spina bifida and meningocele was associated with various degrees of duplication of the spinal cord referred to as diastematomyelia and diplomyelia. In veterinary medicine, duplication of the spinal cord is defined as diastematomyelia (from the Greek diastema = cleft) when the duplication is complete; the 2 histologically well-organized spinal cords are usually separated by a bony partition and contained in their own meningeal sheaths. 13 When the 2 spinal cords are merged and covered by the same meninges, and histologic disorganization of the white and gray matter is present, this condition is referred to as diplomyelia (diplouz = double). 17 Tripartition of the spinal cord, also known as trifid cord, was noted at the level of T7-T8 in our calf. To our knowledge, this condition has only been histologically reported once, in 2010. 18 Very few cases have been described in human medicine. 7
Spinal cord malformations are an uncommon finding in large animals.3,13 Usually occurring in the thoracolumbar segments, such malformations are often associated with vertebral column malformations.2,3 In fact, a close correlation exists between the embryologic development of these 2 structures: the mesenchyme that gives rise to the axial skeleton is derived from the sclerotomal portion of the somites. These bilateral segmental structures originate from the paraxial mesoderm and are located next to the neural tube and notochord. They begin to develop during the third week of gestation. Over the following 2 wk, differentiation of these somites is influenced by the adjacent structures: the notochord and the neural tube stimulate the secretion of epimorphin, which induces sclerotome cells to move close to the notochord and the neural tube and promote differentiation into vertebral cartilage and bone. 6
In human medicine, there is lack of consensus on the classification and terminology used to describe these malformations. Diplomyelia was classically defined as spinal cord duplication, whereas diastematomyelia referred to spinal cord splitting. 15 Replacement of these terms has been proposed with the general term “split cord malformations,” which are further classified as type I and II based on easily identifiable imaging hallmarks.8,9 Type I is characterized by the presence of a rigid bony or cartilaginous septum that gives rise to 2 dural tubes containing completely separated hemicords, whereas type II lacks a rigid septum (sometimes only fibrous or fibrovascular) and the 2 spinal cords are contained within a single dural tube. 8 On the basis of the human classification, our case could be defined as split cord malformation type I.
The embryogenesis of spinal cord tripartition is not well understood. The presence of more than one accessory neuroenteric canal is thought to be involved. Moreover, it is not known whether predisposing factors for the development of diplomyelia exist. Several have been associated with the onset of spina bifida and other neural tube defects in human medicine. 4 Various environmental and genetic factors have been studied, including geography, maternal age, maternal diet, maternal diabetes and obesity, and exposure to antiepileptic drugs. The most significant finding to date has been the protective effect of folic acid consumption during pregnancy, with 60–70% reduction in the incidence of neural tube defects. 1
An essential nutrient for mammalian cell growth, folic acid is involved in the conversion of homocysteine to methionine and in the synthesis of purine and pyrimidine, all essential components for fetal development. Folate deficiency leads to elevated homocysteine levels in the blood, which is thought to be one of the causes of delayed neural tube closure. The conversion of homocysteine to methionine requires the reduction of 5,10-methylenetetrahydrofolate to 5-methylenetetrahydrofolate, which constitutes the primary form of serum folate. This reaction is promoted by the 5,10-methylenetetrahydrofolate reductase enzyme.1–3 Genetic studies have identified 2 alleles and various mutations of the MTHFR enzyme that can cause a deficiency of this enzyme. 16
It has been proposed that MTHFR polymorphism exists in Holstein cattle, and that one of these genotypes is associated with a higher risk of abortion and higher homocysteine plasma concentration during pregnancy. 12 Further studies are needed to better understand the role of this mutation and resulting hyperhomocysteinemia in the development of neural tube defects. Results of genetic investigation for the presence of MTHFR polymorphism in both the calf and the dam in our case were negative.
To our knowledge, there are few reports of split cord malformations in veterinary medicine. The animal in our report was female, as were those described in 1983 (Vitellozzi G, et al. Un caso di diplomielia in un vitello [A case of diplomyelia in a calf]. Atti SISVET, October 5–8, 1983; Abano Terme, Italy) and by other authors.5,14,18 Large case series and retrospective studies on this type of malformation in human medicine have highlighted its higher prevalence among females, with a female-to-male ratio of 1.6:1 to 3:1.9,11
The veterinary literature provides no guidelines for an appropriate CT imaging technique to diagnose diplomyelia. As reported in 2010, ultrasound examination of a 40-d-old crossbreed female calf confirmed diplomyelia in the lumbosacral region of the spinal cord. 14 The authors remarked that, because the vertebral spinous processes in human neonates are not yet ossified, ultrasound evaluation of the spinal cord without acoustic shadowing can be performed. However, the only acoustic window in calves is at the lumbosacral junction, which allows for evaluation of just 1 cm of the spinal cord. 14 In the case of split cord malformation reported in 2010, diplomyelia was diagnosed by magnetic resonance imaging (MRI). 18 Compared to MRI, CT takes considerably less time for image acquisition, with a shorter time required for general anesthesia and better evaluation of bony structures. CT images allowed the diagnosis of spina bifida, meningocele, diplomyelia, and diastematomyelia (or split cord malformation type I according to the human terminology), confirmed on postmortem examination, in our case.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
