Abstract
Tunga penetrans is the smallest biting flea known. In cattle, infestation by T. penetrans (tungiasis) typically affects the skin of the distal legs, udder, prepuce, and perianal area. A detailed clinical and pathologic description of bovine tungiasis, together with electron microscopy and molecular diagnostics to establish the identity of the parasite are described. Ninety percent of the cows and heifers and 80% of the bulls in a herd in northwest Argentina had proliferative and ulcerative skin lesions affecting the coronary band, interdigital space, heels, and rudimentary toes of the fore and/or rear limbs, teats, and/or prepuce. These proliferative lesions had multiple large cavities filled with hemorrhagic fluid, necrotic debris, and Tunga spp. parasites. Histologically, the skin showed diffuse papillary epithelial hyperplasia with severe orthokeratotic hyperkeratosis, and it was multifocally ulcerated and inflamed. Multifocally, sections of arthropod parasites were observed embedded in the epidermis and dermis with the posterior end toward the surface. Images of wet mounts and scanning electron microscopy of the parasite showed morphologic characteristics compatible with Tunga spp. Polymerase chain reaction followed by sequencing of the cytochrome c oxidase subunit II and the internal transcribed spacer region indicated 99% homology to published T. penetrans sequences. Tungiasis should be considered as a differential diagnosis for proliferative lesions in skin of cattle.
Keywords
Proliferative lesions of the skin and adnexa in cattle can be associated with various etiologies including bacterial, viral, fungal, and parasitic agents. 4 Insects, such as Tunga penetrans, have also been described as causing proliferative dermatitis in cattle, although information about the pathology of the lesions produced by this parasite in cattle is very limited.2,9,12
The genus Tunga includes 13 species of sand fleas, of which T. penetrans is the most frequently reported, parasitizing several wild and domestic mammalian species including human beings, cattle, dogs, cats, pigs, goats, sheep, monkeys, wild rodents, coatis, and armadillos.6,13 The disease produced by the female sand flea of the genus Tunga is known as tungiasis. T. penetrans (also known as sand flea, nigua, pique, chigger, or bug of the foot) is the smallest known biting flea (approximately 1 mm diameter). 3 The gravid female penetrates the skin of the host and once in the skin undergoes significant hypertrophy reaching a size of up to 1 cm. 5 It breathes, defecates, expels the eggs, and stays in contact with the air via its abdominal cone, leaving an opening of approximately 250–500-µm diameter in the skin, which can be a port of entry for microorganisms. 5 No drug treatment is known to be effective to eliminate burrowed sand fleas, although insect repellents seem to help in preventing infestation. 3 The treatment of both human and animal tungiasis consists mainly of surgical extraction of the fleas, followed by cleansing and topical antibiotics to prevent secondary infections. Although topical ivermectin, metrifonate, and thiabendazole have been reported to be effective in killing the fleas and facilitate their manual removal, these treatments do not remove the flea from the skin and do not provide relief for the painful Tunga spp. lesions.1,7,8 Preventive fumigation of problem fields and pens with pyrethroid substances at the beginning of the dry season seems to help in preventing infestations (authors’ unpublished observation).
In human beings, tungiasis by T. penetrans is endemic in several areas of South and Central America, and Africa. 14 Isolated cases of this disease have also been reported in India and Italy.6,14 Originally T. penetrans existed only in the Americas from where it propagated to other parts of the world. 3 Both in human beings and animals, tungiasis is mainly associated with sandy soils and dry weather, but it has also occasionally been seen in rainforests. 3
Bovine tungiasis has been described in several South American countries with most reports originating from Brazil.10,12 The disease in cattle can be produced by 3 species of Tunga (i.e., Tunga penetrans, Tunga trimamillata, and Tunga hexalobulata),2,10 although T. penetrans is by far the most frequently reported species of Tunga associated with bovine tungiasis.
Tungiasis by T. penetrans in cattle usually affects the skin of the distal feet, udder, prepuce, and perianal area. Prevalences between 2.5% and 68% have been described in bovine herds.2,12 Lesions in bovine tungiasis are painful, pruritic, and often secondarily contaminated with opportunistic bacteria. 12 Treatment is rarely applied to cattle and typically the outcome of infection is culling due to severe lameness, secondary infections, and the drop in production associated with these lesions.
Previous reports of bovine tungiasis have described only the epidemiological and clinical characteristics of the disease; while the identification of Tunga spp. has been traditionally limited to morphological features alone.2,12 The clinical and pathological changes produced by different species of Tunga spp. are very similar to each other, and determination of the species present cannot be achieved by these features alone. The current study presents a detailed pathologic description of bovine tungiasis together with the use of electron microscopic findings and molecular techniques that confirmed the identity of the parasite.
In 2007, the owner of a breeding herd of Brangus cross cattle (n = 1,430, including cows, heifers, steers, bulls, and calves) in the province of Salta, northwest Argentina, reported a high incidence of digital lesions, mainly in the adult cattle. The farm was visited by one of the authors (R Marin), and all the cows and heifers (n = 520) and bulls (n = 30) were examined clinically with special emphasis on feet, udders, and in the case of the bulls, prepuce. Acute clinical signs consisted of intense pruritus, pain, licking, and swelling of affected areas (mostly feet and teats), and obstruction of the teat canal. The latter led to severe reduction in milk production and mastitis, which in turn was responsible for loss of condition and death of a large number of calves. Animals that had been affected for several days presented hoof deformities, anorexia with severe loss of condition, lameness, and marked reduction of reproductive indexes. The bulls showed marked loss of libido.
An adult cow with feet and teat lesions similar in nature and severity to those seen in most animals of the herd was euthanized, and a full necropsy, including thorough gross examination of the skin, with particular emphasis on limbs and udder, was performed. Samples from leg and teat lesions were collected in 10% buffered formalin (pH 7) and processed routinely for the production of 4-µm thick hematoxylin and eosin–stained sections. Selected sections of skin were also stained with Steiner, Gram, and an indirect immunoperoxidase technique for Treponema spp. using a commercial kit a according to previous descriptions. 11 For the latter, a rabbit polyclonal anti–Treponema spp. antibody b was used as a primary antibody. Skin from the interdigital space of a cow with papillomatous digital dermatitis and from the interdigital space of a normal cow, were used as positive and negative controls, respectively.
Samples from the legs and teat lesions from the euthanized cow were aseptically collected, inoculated onto 5% blood agar plates, and incubated aerobically or anaerobically for 48 hr. Colonies were examined on Gram-stained smears and finally identified by conventional biochemical tests. Direct smears of fine-needle aspirates from the leg lesions were also examined by dark field microscopy.
Fleas from the environment and from the skin of several affected animals were collected, fixed initially in 70% ethanol, and postfixed in Karnovsky fixative in 0.1 M sodium phosphate buffer (Sorenson), then washed using 0.1 M sodium phosphate. Dehydration was accomplished in increasing concentrations of ethanol through 100% ethanol and critical point dried. c The fleas were mounted on aluminum stubs and sputter coated with gold using a commercial coater. d The samples were viewed on a scanning electron microscope (SEM), e and digital images were obtained.
DNA from 3 fleas was extracted using a commercial blood and tissue kit f according to the manufacturer’s instructions. Two different sets of primers specific for T. penetrans were designed, one to detect cytochrome c oxidase subunit II (COII; accession no. DQ 844706.1) and another to detect 5.8S ribosomal RNA gene internal transcribed spacer (ITS; accession no. DQ844725.1). The following COII primers with expected amplicon size of 468 bp were used: forward 5′-AATTTACTCACCGAATATTAATAGAAAGTCAA-3′, and reverse 5′-CTATGATTTGCTCCACAGATTTCTG-3′. The following ITS primers with expected amplicon size of 396 bp were used: forward 5′-CTAATTGCGCGTCAACAT GTG-3′ and reverse 5′-AAGCGTGGAGGTTTC GAGTTC- 3′. The PCR amplification was carried out using commercial kit g consisting of 5 μl of 10× Advantage 2 buffer, g 1 μl of 10 mM deoxyribonucleotide triphosphate mix, 1 μl of each primer, 20 μl of extracted DNA, 21 μl of diethylpyrocarbonate-treated water, and 1 μl of 50× Advantage 2 polymerase mix g for a total reaction volume of 50 μl. The following cycling conditions were used: 94°C for 1 min, 25 cycles of 94°C for 15 sec, 55°C for 15 sec, and 70°C for 45 sec, following with a final elongation of 70°C for 5 min and a 4°C forever hold step. The PCR products were run on 1.8% agarose gel with addition of EZ VISION dye, h and the expected sizes of bands were noted. Each band was cut out, purified i according to manufacturer’s instructions, and submitted to the sequencing facility of the University of California–Davis.
Ninety percent of the cows and heifers (468/520) and 80% of the bulls (24/30) had proliferative skin lesions affecting 1 or more of the following locations: fetlock area, rudimentary toes, coronary band, interdigital space and heels of the fore and/or rear limbs, teats, and/or prepuce. Proliferative lesions in most animals were ulcerated and bleeding.
In the euthanized adult cow, 4 legs and 4 teats were affected. Proliferative and hemorrhagic lesions of varying sizes (3 cm × 2 cm × 2 cm to 15 cm × 10 cm × 6 cm) were present on all 4 feet distal to the fetlock joint, including the coronary band and interdigital space (Fig. 1), and the teats (Fig. 2); there was also abnormal hoof growth (Fig. 3). Cut section of the leg and teat lesions revealed multiple large cavities that contained hemorrhagic fluid, necrotic debris and Tunga spp. parasites. Subcutaneous edema extended distally from the carpi and tarsi to the coronary band. Lesions in the teats were similar but less severe. No other significant gross abnormalities were observed in the rest of the carcass.
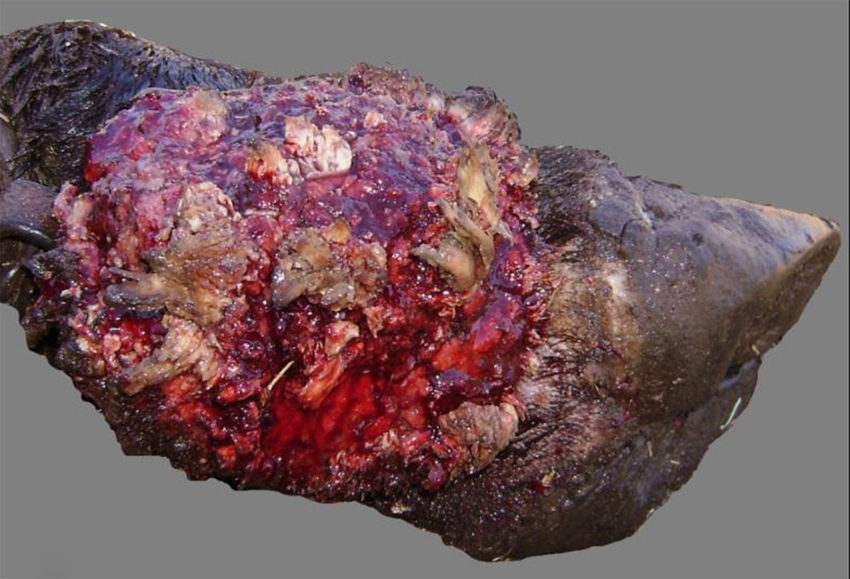
Cow. Distal limb with proliferative and ulcerative dermatitis and coronitis due to Tunga penetrans.
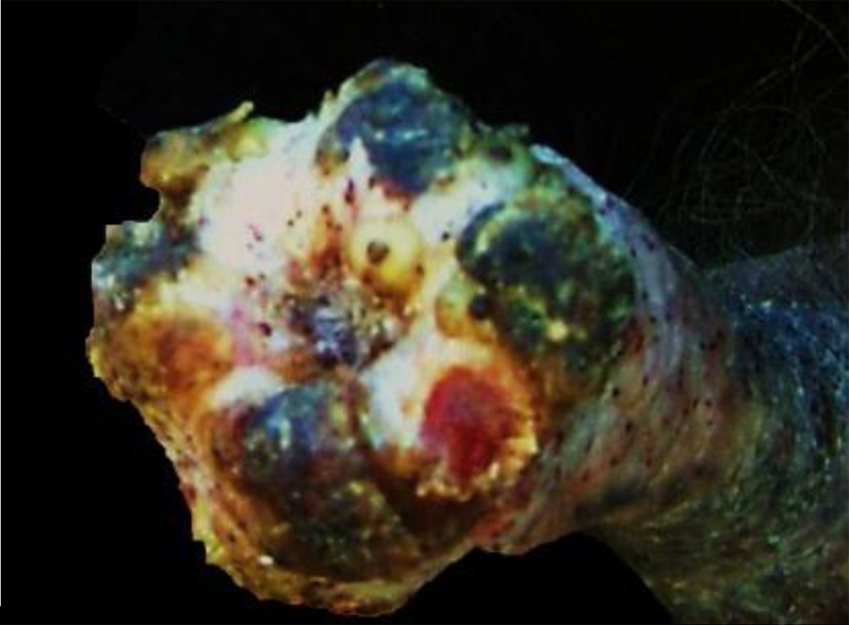
Cow. Udder with proliferative and ulcerative dermatitis due to Tunga penetrans.
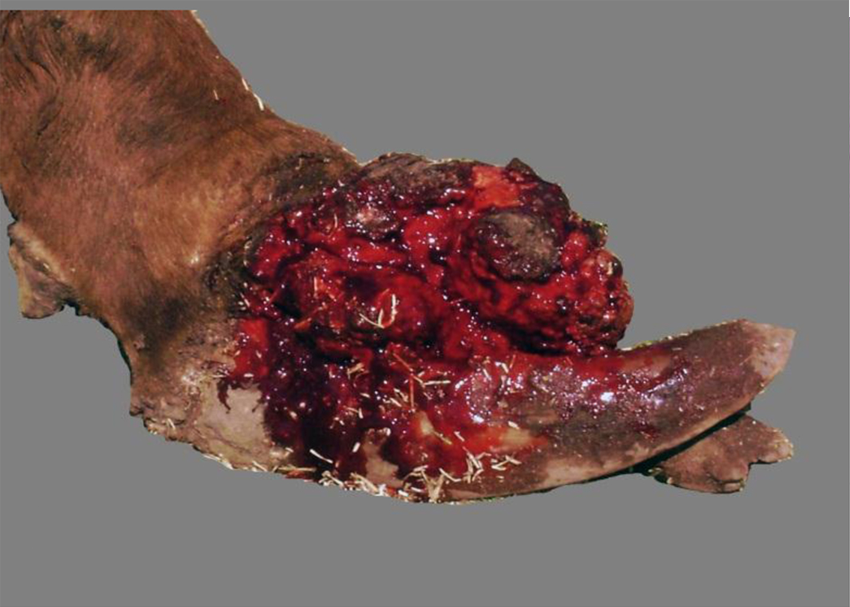
Cow. Distal limb with proliferative and ulcerative dermatitis, and coronitis due to Tunga penetrans. Notice deformed hooves.
Histologic findings were similar in leg and teat lesions. The epidermis showed diffuse papillary hyperplasia with severe orthokeratotic hyperkeratosis (Figs. 4, 5). Multifocally, there was deep chronic ulceration covered by large crusts composed of extravasated red blood cells, lymphocytes, plasma cells, macrophages, neutrophils, eosinophils, fibrin, cell debris, and bacterial rods and cocci. The dermis showed diffuse infiltration by lymphocytes, plasma cells, macrophages, eosinophils, and neutrophils (which was most severe surrounding parasites), fibroplasia with proliferating blood vessels oriented perpendicular to the surface (granulation tissue), and pockets of necrosis and neutrophilic infiltration, which included both viable and dead neutrophils. Hematoxylin and eosin– and Steiner-stained sections of skin showed superficial invasion of mixed bacteria, which multifocally invaded the superficial dermis. This bacterial population was composed by a mixture of Gram-positive and -negative organisms. Immunohistochemical staining for Treponema spp. was negative in all sections examined.
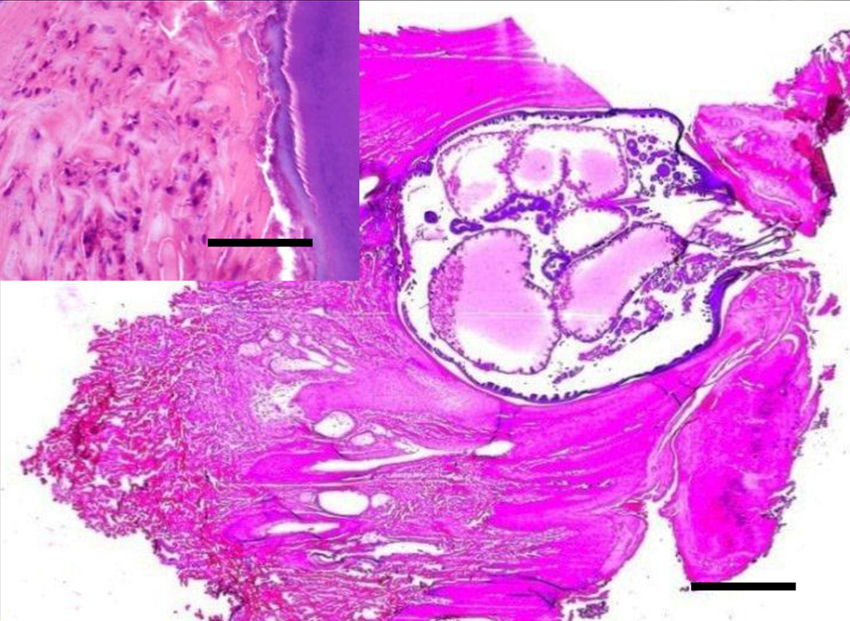
Cow. Subgross section of skin with prominent orthokeratotic hyperkeratosis. Observe an intradermal and epidermal section of an arthropod compatible with Tunga penetrans, with posterior end toward the surface. The parasite shows a thick cuticle, prominent hypodermic cells lining the body cavity, distended digestive tract sections, and posterior end with chitin. Hematoxylin and eosin (HE). Bar = 100 μm. Inset: higher magnification of the external parasite wall (right) and dermal host tissue (left) showing neutrophilic and eosinophilic infiltration. HE. Bar = 20 μm.
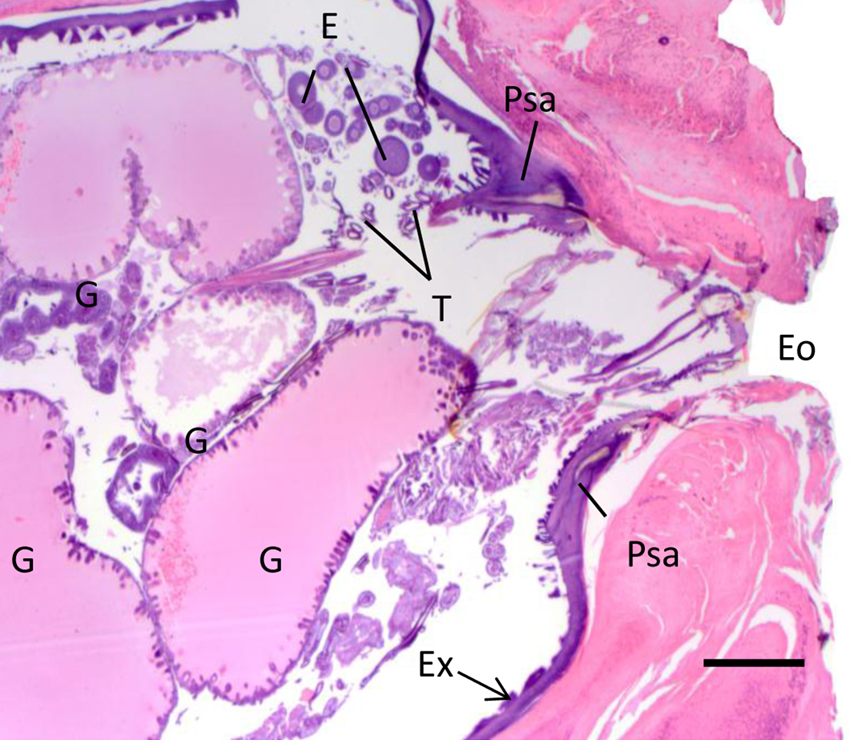
Cow. Higher magnification of the posterior end of Tunga penetrans in the skin showed in Figure 3. The image shows the thick basophilic cuticle of the exoskeleton (Ex), distended intestinal loops (G), trachea (T), developing eggs (E), pale-staining areas (Psa), and external orifice (Eo). Hematoxylin and eosin. Bar = 40 μm.
Multifocally, sections of arthropod parasites were embedded in the epidermis and dermis with the posterior end oriented toward the surface (Figs. 4, 5). The parasites were approximately 2 mm × 3 mm in length, with a thick body wall lined by a prominent hypodermal layer and cuticularized structures on the external surface. The head presented a well-developed lacinia and epipharynx. Within the body cavity there were myriad, approximately 100 µm diameter eggs at various stages of development. The eggs were round and solid or targetoid (Fig. 5). Long eosinophilic muscle fibers with central elongated nuclei, and tubular transverse sections of intestine lined by prominent epithelium, trachea, and pale-staining areas were also seen scattered within the parasite body wall (Fig. 5).
Bacterial cultures of leg and teat skin samples produced almost pure growth of Pseudomonas spp. Spirochetes were not seen on direct smears of fine-needle aspirates from the leg lesions examined by dark field microscopy.
Light microscopy of wet mounts (Figs. 6, 7) and SEM images (Figs. 8, 9) of male and non-gravid female parasites showed a relatively flattened head. The genal groove, characteristic of T. penetrans, was visible on wet mounts and SEM of both sexes (Figs. 6–9), as was the absence of setae on the medial border of the tibia (Fig. 10). Sexual dimorphism was characteristic of the genus (i.e., the male possessing a prominent copulatory apparatus and the female having a distinct genital depression).

Light micrograph of a non-gravid female Tunga penetrans in left lateral recumbency. T: tibia of left hind leg; GD: genital depression. Wet mount. Bar = 200 μm.

Light micrograph of an adult male Tunga penetrans in left lateral recumbency. T: tibia of left hind leg; CA: copulatory apparatus. Wet mount. Bar = 200 μm.

Nongravid female Tunga penetrans in left lateral recumbency. GD: genital depression; A: antenna; E: eye; GG: genal groove; LP: labial palps. Scanning electron microscopy. Bar = 200 μm.
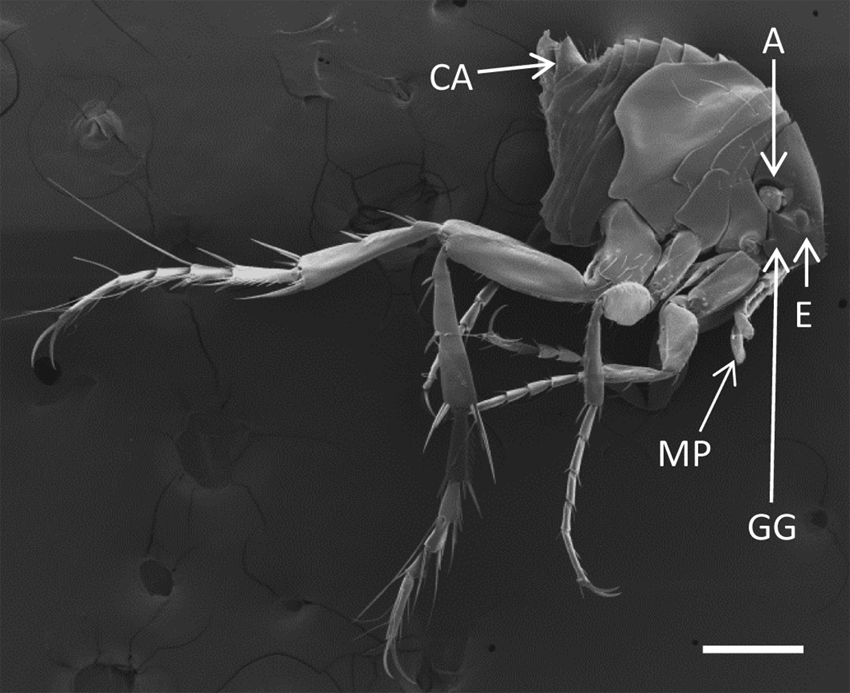
Male Tunga penetrans in left lateral recumbency. CA: copulatory apparatus; A: antenna; E: eye; GG: genal groove; MP: maxillary palps. Bar = 200 μm.
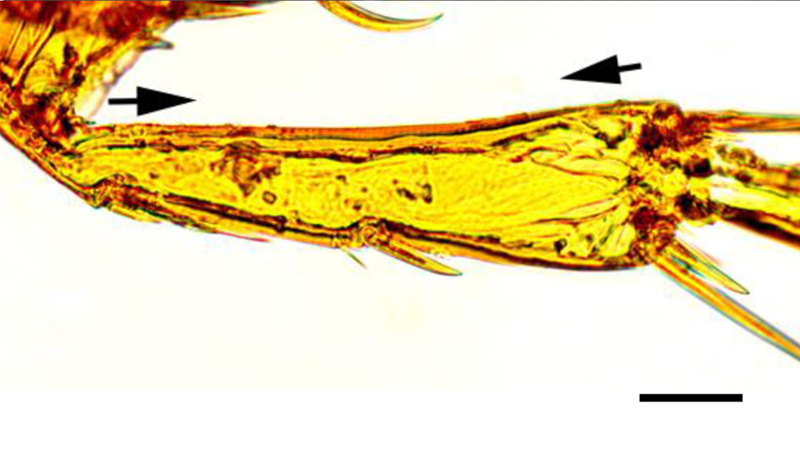
Light micrograph of the tibia of a female Tunga penetrans. The region between the arrows shows the lack of setae that distinguishes T. penetrans from Tunga trimamillata. Wet mount. Bar = 40 μm.
Sequencing results for COII indicated 99% homology to published T. penetrans sequences. There was a 3-bp mismatch. The sequence for the ITS region of T. penetrans also indicated 99% homology to the known published sequences of this parasite with a 5-bp mismatch.
A diagnosis of tungiasis was made based on the clinical, gross, and histologic findings. Light microscopic and SEM examination of the parasites allowed identification of the parasite as T. penetrans. Pale-staining areas were seen within the parasite wall. Similar pale-staining areas have been previously described in the inner and outer portions of the sand flea’s posterior region of the exoskeleton. 14 These pale areas seem to be unique for T. penetrans, and they are considered to be useful identifying features. Further confirmation of the species as T. penetrans was provided by sequencing of the COII and ITS, both of which showed 99% homology to the published sequences of this parasite. Even though both fragments had a few base pair mismatches to the currently published sequence data, the primers were designed on areas that would amplify regions conserved to T. penetrans species, and sequenced fragments significantly differed from T. trimamillata, which allowed ruling out the latter. Other causes of proliferative dermatitis were ruled out based on clinical, gross, and microscopic examination, as well as immunohistochemical staining for Treponema spp., and bacterial cultures.
Although 13 species of Tunga have been described, only 3 of them (i.e., T. penetrans, T. trimamillata, and T. hexalobulata) have been described in cattle, with the first one being most frequently seen.2,10 Differentiation of the 3 species of Tunga that affect cattle based on morphology of the parasite alone is difficult and frequently inaccurate. The use of molecular methods in the current study proved to be a quick and efficacious method to identify Tunga at a species level. Identifying fleas to the species level may prove useful to select appropriate control and treatment measures if differences in sensitivity and pathogenesis are found between flea species in the future.
Between 80% and 90% of the animals in the study farm had proliferative lesions consistent with tungiasis. Similar prevalences were observed in some neighboring farms (authors’ unpublished observations). To the best of the authors’ knowledge, this high prevalence has not been previously reported in cattle or other animal species. Tunga penetrans thrives in semitropical or tropical climates with sandy soils, as was present in the study farm. However, a high prevalence of tungiasis was not always seen in neighboring farms with similar conditions. The reason for the high prevalence of disease seen in the current study is unknown; further epidemiological studies are necessary to clarify this issue.
Tungiasis should be considered as a differential diagnosis for proliferative lesions in skin and adnexa of cattle. Molecular methods are very useful for rapid identification of the parasite at the species level. Fleas and/or full thickness skin samples are required for diagnosis.
Footnotes
Acknowledgements
All SEM work was completed in the Electron Microscopy Laboratory, Department of Medical Pathology and Laboratory Medicine, School of Medicine, University of California at Davis. The authors thank Ms. Patricia Kysar for her technical assistance and the field staff of San Jose de Pocoy, Salta, Argentina, for their help in several ways.
a.
ABC Vectastain kit, Vector Laboratories Inc., Burlingame, CA.
b.
Rabbit polyclonal anti-treponema antibody, CAHFS-Davis, CA.
c.
Critical point dryer: Tousimis 931.GL Autosamdri, Tousimis Research Corp., Rockville, MD.
d.
Pelco auto sputter coater SC-7, Ted Pella Inc., Redding, CA.
e.
Philips XL30 TMP, FEI Co., Hillsboro, OR.
f.
DNeasy blood and tissue kit, Qiagen NV, Venlo, The Netherlands.
g.
Advantage 2 polymerase kit (catalog no. 639206), Clontech Laboratories Inc., Palo Alto, CA.
h.
EZ Vision dye, Amresco Inc., Solon, OH.
i.
QIAquick gel extraction kit, Qiagen NV, Venlo, The Netherlands.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
