Abstract
A 19.5-y-old, male domestic yak (Bos grunniens) with a history of sudden unexpected death was submitted for autopsy. The yak had hemoabdomen, and a large blood clot was attached to the liver and forestomachs. The hepatic artery had a saccular aneurysm with a 1-cm tear. The arterial wall at the site of the rupture was thin, and the luminal surface was roughened with yellow streaks. The arterial wall adjacent to the rupture was thickened, white, firm, and less elastic than normal arterial walls. Cause of death was concluded to be acute exsanguination. Similar cases with sudden death have been reported in domestic cattle and humans. No risk factors, such as nutritional deficiencies, genetic predisposition, or blunt trauma, were identified in this case, and there was no gross or histologic evidence of generalized vascular disease.
Aneurysms of abdominal arteries, including the aorta, result from a number of underlying causes leading to progressive loss of structural integrity, hemodynamic changes, and possible rupture.3,11 Abdominal aortic aneurysms in humans that rupture have a mortality rate of up to 90%. 10 The low survival rate is a result of the difficulty in detecting aneurysms prior to rupture, 10 and the rapid nature by which exsanguination and shock occur upon rupture. 18 Practitioners on the human and animal side of medicine strive to further understand the causes of aneurysms. We document herein a novel case of hepatic artery aneurysm in a domestic yak (Bos grunniens).
A 19.5-y-old, 503 kg, castrated male domestic yak was presented to the Veterinary Diagnostic Laboratory (VDL) at Michigan State University (East Lansing, MI) for postmortem examination following a sudden and unexpected death. The animal had no recent health concerns. Annual wellness examination consisting of a complete blood count and biochemical analysis, performed 6 mo earlier, was within normal limits.
At autopsy, the yak was in good body condition with adequate hydration and abdominal and subcutaneous fat stores. The mucous membranes and sclera were pale, but otherwise the external appearance of the animal was unremarkable. The abdomen contained a large amount of clotted and unclotted blood, with the largest clots localized at the caudodorsal and caudoventral hepatic surfaces and right lateral serosal surfaces of the forestomachs. The hepatic artery was markedly dilated adjacent to the branching of the celiac artery into the hepatic and gastroduodenal branches, with a substantial amount of clotted blood in the surrounding area (Fig. 1). The vessel wall in this area was focally thin with aneurysmal dilation. The hepatic artery had ruptured at the site of the aneurysm and the tear, ~1 cm in length, had roughened, dark-red edges (Fig 2). The luminal surface of the artery was focally roughened with yellow streaks at the site of the rupture. The arterial wall adjacent to the rupture was thickened, and the affected wall was white, firm, and less elastic compared to grossly normal arterial wall proximal and distal to the thickening and aneurysm (Fig. 2). Grossly, all other major arteries and veins of the abdomen and thorax were unremarkable, including but not limited to the vena cava, aorta, pulmonary vessels, carotid arteries, and the celiac artery and its branches distant to the aneurysm. The cause of death was determined to be acute exsanguination via rupture of a hepatic artery aneurysm. To better understand the changes, and possibly identify an etiology, sections of the described vessels were taken for histologic examination; liver samples were submitted for mineral analysis, including copper.
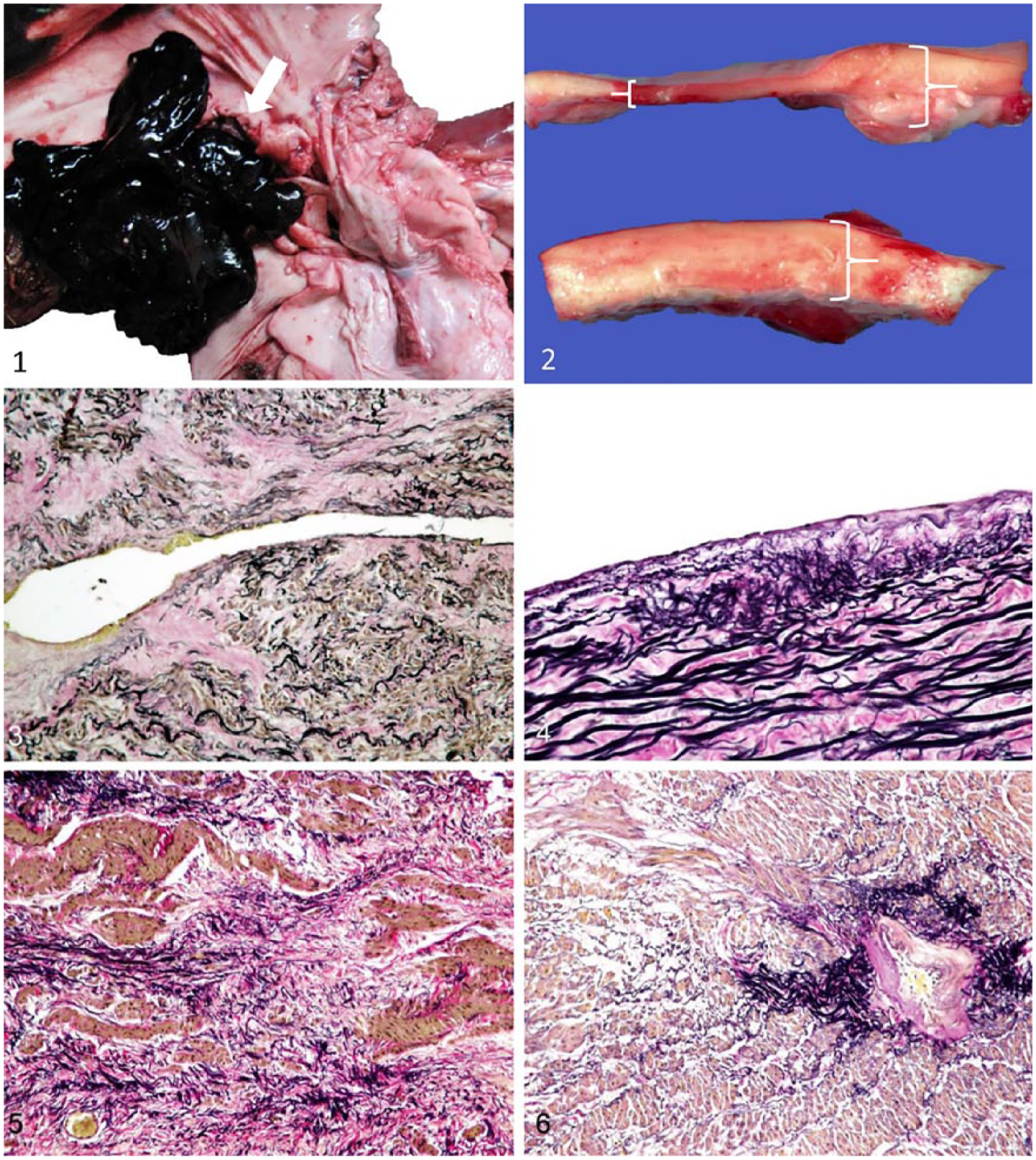
The intima of the affected artery is roughened (arrow), and a large blood clot is attached to the tear in the aneurysm.
On microscopic examination, samples taken from the rupture site were very thin; areas adjacent to the thinning and rupture were twice the thickness of comparable vessels. Because an age-matched control animal was not available, sections of celiac artery away from the lesion were used as the primary comparator. Histologically, other abdominal arteries, and the pulmonary and carotid arteries, were unremarkable. At the margins of the rupture site, the tunica intima was fragmented or completely absent. Superficial portions of the tunica media were also absent (Fig. 3). The internal elastic lamina was often lost or disrupted, and there were focal areas of small elastic fiber fragmentation and disarray (Fig. 4). Within the tunica media, elastic fibers were not parallel; there were focal areas of smooth muscle hyperplasia alternating with areas of degeneration and fibrosis, and focally extensive mineralization (Fig. 5). The adventitial vasa vasora had marked intimal thickening with smooth muscle hyperplasia and luminal restriction, and were commonly surrounded by dense aggregates of elastic fibers (Fig. 6). A few areas had subendothelial accumulation of small numbers of inflammatory cells. The vessel wall proximal and distal to the aneurysm was thickened by smooth muscle hyperplasia. There was focal extensive mesothelial papillary proliferation on the serosal surface of affected vessels. The histologic nature of these lesions is consistent with a degenerative pathogenesis, and absence of significant inflammation and necrosis made immune-mediated or infectious disease unlikely.
The liver copper concentration was 232 µg/g, which was within the reference interval (50–300 µg/g) for domestic bovine species. Reference values for hepatic mineral concentrations were not available for domestic yak.
Most sudden deaths in cattle are the result of respiratory disease, acute metabolic disturbances, or bloat. 7 However, 4% of sudden deaths in feedlot cattle result from cardiovascular disease, primarily pulmonary artery aneurysm. 14 This most often affects a single animal in a group. Acute rupture of the aorta and other blood vessels can result from multiple causes, but is often the result of a previously weakened structure. In veterinary medicine, causes of rupture of major vessels and exsanguination include copper deficiency in pigs, 19 a genetic predisposition in Friesian horses 15 or cattle with bovine Marfan syndrome, 16 suspected collagen disorders and degenerative mitral valve disease in several small-breed dogs, 17 as well as blunt trauma.
Exsanguination as a result of abdominal artery aneurysm rupture has been recognized previously in Holstein dairy cattle. 12 Such cases identified both an acute form, characterized by fibrin and hemorrhage at the rupture site, and a chronic form, characterized by proliferation of the intima of the vessel wall and rupture of the internal elastic lamina at the site of rupture. 12 In bovine Marfan syndrome, vascular elastin is abnormal because of decreased fibrillin production, leading to an increased risk of rupture. 16 The pathogenesis appears to be an initial and persistent weakening within the vessel wall, followed by attempts at repair with smooth muscle hypertrophy, development of granulation tissue, and intimal thickening before eventual rupture. 16 We ruled out bovine Marfan syndrome lesions in our case based on the absence of other clinically described skeletal and ocular abnormalities. 9
In pigs and turkeys, copper deficiency results in defects in lysyl oxidase, leading to decreased and abnormal layering of elastin, which may contribute to increased propensity for aortic aneurysm and rupture.8,13 Trace mineral deficiency in captive wild bovid species continues to be a concern when separating the animals from their natural environment and diet. 5 Concentrations of copper in forage can vary with the season, resulting in fluctuations in the diet. 2 Copper deficiency can also be induced artificially by elevated levels of sulfur or molybdenum in the diet. 21 Yaks are considered to be vulnerable to copper deficiency because of a lower capacity to absorb dietary copper compared to other bovine species. 20 However, the yak in our case was determined to have normal liver copper values at the time of death compared to normal domestic bovine reference values. We cannot comment on possible historical fluctuations given lack of serial copper testing.
In humans, abdominal aortic aneurysm affects 5–9% of the population > 65-y-old, 1 and rupture in these patients often results in sudden death.1,4 Rupture of an aneurysm most commonly results from trauma or cardiac stress, often induced by high-intensity exercise. Aneurysms can be caused by arteriosclerosis, weakened vascular walls, or decreased blood flow rate. 6 Cigarette smoking, atherosclerosis, arterial hypertension, and dyslipidemia are considered to be risk factors for developing abdominal aortic aneurysm. 4 Other factors thought to play a role include proteolytic enzymes such as elastases, collagenases, and matrix metalloproteinases; oxidative stressors such as nitric oxide; and inflammation. 4 Additional proposed mechanisms for aneurysms of the abdominal arteries with subsequent rupture include deposition of antigen–antibody complexes resulting in fragmentation and breakdown of elastin. 22 Primary defects in collagen, as is seen with Ehlers–Danlos syndrome in humans, may cause weakening of the vessel wall with secondary fracturing of elastin. 12
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
