Abstract
Introduction
Aortic aneurysm (AA) is a focal dilation of the aorta characterized by progressive loss of elastin, death of smooth muscle cells (SMCs), loss of endothelial cell (EC) lining, and persistent inflammatory response driven by innate immune cells such as monocytes and macrophages. 1 Increased circulating blood monocytes, known as monocytosis, is associated with AA. Subsequently, monocyte infiltration and differentiation to macrophages is a major contributor to macrophage accumulation in AAs.2–5 Macrophages can induce cytotoxicity and oxidative stress in SMCs and ECs, promoting the de-differentiation and proliferation of SMCs as well as disrupting the endothelial barrier.6–9 Pentagalloyl glucose (PGG) is a polyphenol compound that has been shown to stabilize and promote vascular elastic fiber formation. 10 When PGG was delivered with albumin nanoparticles conjugated to degraded elastin-targeting antibodies (PGG-ELN-NPs), it reversed AA growth in elastase, calcium chloride (CaCl2), or angiotensin II (AngII) mediated AA rodent models associated with decreased number of macrophages.11–15 While its interactions with elastin and other extracellular matrix components have been more thoroughly studied, 16 the cellular mechanisms of aneurysm resolution by PGG are not well understood. In aneurysmal tissues, PGG-ELN-NP treatment reduced the expression of CD80, a M1 macrophage inflammatory phenotype marker, suggesting PGG modulates either macrophage infiltration or polarization status.11,12,14 Treatment of ECs with similar compounds such as tannic acid and ethyl gallate decreases monocyte and leukocyte adhesion by reducing expression of adhesion molecules like vascular cell adhesion molecule 1 (VCAM-1), intercellular adhesion molecule 1 (ICAM-1), and E-selectin.17,18 Thus, PGG may directly reduce monocyte adhesion contributing to AA resolution; however, this effect has not been explored.
PGG-ELN-NP treatment was shown to reduce tumor necrosis factor-alpha (TNF-α) expression as well as increase the expression of interleukin-10 (IL-10).11–15 IL-10 and TNF-α can be highly expressed by macrophages.19,20 IL-10 is reduced in mouse and human AAs while TNF-α and TNF-α converting enzyme (TACE), which converts membrane-bound TNF-α to soluble TNF-α, is overexpressed. 21 Consequently, the expression of TNF-α is localized to areas of macrophage infiltration in human AAs. 22 NF-κB is considered a crucial transcription factor in inflammatory macrophages as it is a master regulator of associated genes, including TNF-α. 23 The NF-κB subunit p65 and its inhibitor IκBα are respectively up- and down-regulated in abdominal AA patients. 24 Given this, we sought to determine if PGG can suppress activated p65 NF-κB in THP-1-derived macrophages, increasing the ratio of IL-10/TNF-α and CD206/CD80 expression thus reducing subsequent macrophage-induced vascular cell pathology.
Interestingly, we have also observed that PGG-ELN-NP mediated AA reversal drastically reduces systemic and local levels of TGF-β. While reduction of TGF-β was associated with disease resolution, inhibiting TGF-β directly in AngII-infused ApoE −/− mice or elastase-induced AA causes mortality due to aortic rupture.11,25,26 In the hereditary condition Marfan Syndrome, patients spontaneously develop AAs and have increased TGF-β signaling. Ablation of myeloid cell TGF-β type II receptor (Tgfbr2) in the Fbn1C1039G/+ mouse model of Marfan Syndrome attenuated AA formation and macrophage infiltration. 27 Conversely, SMC-specific Tgfbr2 deletion in ApoE −/− mice on a high-fat diet caused de-differentiation of contractile SMCs, which exacerbated vascular pathogenesis; however, this same deletion was protective against elastase-induced abdominal AA.28,29 Further supporting the role of TGF-β in pathological SMC-macrophage crosstalk, atherosclerotic macrophages exhibit a distinct phenotype that can be replicated in vitro by maturing rat bone marrow cells in the presence of macrophage-colony stimulating factor and SMC-conditioned medium or recombinant TGF-β1. This distinct phenotype is suppressed by a TGF-β antibody, TGF-β receptor inhibitor, or conditioned media from TGF-β–depleted SMCs, suggesting SMC-derived TGF-β is critical to macrophage-induced vascular pathogenesis and remodeling. 30
Furthermore, elastic fiber degradation in AA causes cleavage of microfibril-bound latent TGF-β, which can subsequently be activated by matrix metalloproteases (MMPs) expressed by inflammatory macrophages such as MMP-2 and 9.31–33 Previous studies examining the influence of PGG treatment on TGF-β expression were restricted to whole serum or tissue analysis, limiting our understanding of the effects of PGG and the major biological sources as well as functions of this cytokine. Thus, this study aims to assess further the cellular mechanisms underlying the anti-inflammatory and vaso-protective properties of PGG. We sought to determine if the aneurysm-reversing effect of PGG may be partially due to the influence of PGG on monocyte adhesion, the inflammatory polarization and mitochondrial oxidation status of macrophages, prevention of macrophage induced human vascular cell oxidative stress and cytotoxicity, as well as cellular and extracellular matrix specific modulation of TGF-β release.
Materials and methods
Cell culture
Primary human aortic endothelial cells (HAoECs) and human aortic smooth muscle cells (HAoSMCs) were purchased from PromoCell and used between passages four and six for all experiments. Monocultures of HAoECs and HAoSMCs were maintained in 75 cm2 tissue culture-treated vented flasks in a 37°C and 5% CO2 environment in Endothelial Cell Medium and Smooth Muscle Cell Medium II respectively (Promocell). THP-1 monocytes, as well as macrophages, were maintained in RPMI media (Thermo Fisher Scientific) supplemented with 10% fetal calf serum (Thermo Fisher Scientific),
Ex vivo aorta culture
To perform an ex vivo study, whole-length infrarenal aortas until iliac bifurcation were extracted from male CD® Sprague-Dawley rats (n = 4; weight ∼700g) euthanized by saline perfusion under isoflurane anesthesia into ice cold sterile Moscona’s buffer solution (8g NaCl, 1.7g glucose, 1g NaHCO3, 0.2g KCl, 0.005g NaH2PO4 in 1 L DI water; pH adjusted to 7.2). The animal study followed Clemson University IACUC guidelines (protocol# AUP2022-0423) for humane animal treatment and complied with relevant legislation. Aortas were washed thrice with Dulbocco’s Phosphate Buffered Saline (DPBS) containing 1% penicillin/streptomycin. Upon cleaning, whole-length aortas were cut into 3-4 mm pieces of aortic rings and placed in ultra-low adherent 12-well plates for culture.
After overnight equilibration in Dulbocco’s Modified Eagles Medium (DMEM – Sigma Aldrich) supplemented with 10% fetal calf serum (Thermo Fisher Scientific),
Monocyte adhesion assay
HAoECs and HAoVSMCs were cultured with 0.1% DMSO (Vehicle Control) or 10 µg/mL PGG for 72 h in 6-well plates. THP-1 Monocytes were added to existing media of HAoECs and HAoVSMCs at 100k cells per well and incubated at 37°C, 5% CO2 for 30 min. Unbound cells were removed by washing 3x with warm PBS. Analysis was conducted on triplicates with N = 3 biological replicates, five representative fields of view were captured at 10x magnification, and images were analyzed using ImageJ.
Macrophage differentiation from THP-1 monocytes and conditioning of medium
1 × 106 THP-1 Monocytes were plated in 6-well plates and stimulated with 10 ng/mL phorbol 12- myristate 13-acetate (PMA, Sigma Aldrich) for 48 h. Differentiated Macrophages were washed with warm PBS and rested for 48 h in a fresh medium. The medium was then replaced with fresh medium containing 0.1% DMSO (vehicle control) or 10 ug/mL PGG, and cells were stimulated for 72 h.
Analysis of cellular and mitochondrial ROS
CellROX or MitoSOX (Invitrogen) were added to the fresh culture medium and incubated following the manufacturer’s instructions. Five representative fields of view were captured at 10x magnification, and images were analyzed using ImageJ.
Estimation of mitochondrial membrane polarization
The number of cells with high mitochondrial activity was estimated in THP-1 monocyte-derived macrophages following stimulation with 0.1% DMSO (vehicle control) or 10 µg/mL PGG for 72 h by staining cells for 30 min with 20 nM tetramethylrhodamine (TMRM). Cells were subsequently analyzed by fluorescence microscopy. Analysis was conducted in triplicates on N = 3 biological replicates, five representative fields of view were captured at 200x total magnification and images were analyzed using ImageJ.
Viability and proliferation of human vascular cells exposed to macrophage-conditioned medium
The previously collected macrophage-conditioned medium was used to replace the medium of confluent human aortic smooth muscle cells cultured in separate 6-well plates – the HAoVSMCs were exposed to conditioned medium for another 72 h then stained for Live/Dead cells. Analysis was conducted in triplicates on N = 3 biological replicates; five representative fields of view were captured at 2x magnification, and images were analyzed using ImageJ.
Determination of IL-10, TNF-α, and TGF-β expression
Supernatants were centrifuged at 2000g for 10 min at 4°C to remove cellular debris, and cytokine concentrations in supernatants were quantified using TGF-β1, TNF-α, and IL-10 ELISA kits (Invitrogen) according to manufacturer’s protocol.
Immunofluorescence
For immunofluorescence of the cultured differentiated THP-1 derived macrophages, following 3-days stimulation with vehicle or PGG, cells were washed with PBS, fixed with ice-cold methanol for 10 min, and incubated for 20 min with Background Buster (Innovex) to block non-specific antibody bindings. Cells were subsequently incubated overnight with CD208, CD80, phospho-p65 NF-κB (1:300 dilutions) primary antibodies. The next day, cells were washed in DPBS and incubated with appropriate fluorophore-conjugated secondary antibodies (1:100 dilutions) at room temperature for an hour. Following washing with DPBS and DAPI nuclear staining, the cells were imaged using a Keyence BZ-810 fluorescent microscope.
ImageJ analysis
All images were processed and analyzed with ImageJ 1.45 s (National Institutes of Health, USA), where fluorescence intensities and particle counts were quantified. 8-bit red-green–blue fluorescent images were converted into binary images, and a threshold was set for color saturation of fluorescence for stained cells or fluorescent beads. Integrated density data or particle counts representing the number of stained cells, were obtained. Average values of the integrated density data obtained from three independent experiments were statistically analyzed.
Statistical analysis
Data is shown as mean ± standard error of the mean (SEM). Unpaired students’ t-tests or repeated measures, such as one-way ANOVA with Tukey’s post-hoc tests, were performed to determine significance between groups. Results were considered statistically significant if p values were <.05.
Results
Pentagalloyl glucose decreases the adhesion of THP-1 monocytes to HAoECs, HAoSMCs, and elastase-damaged rat aortas
Pre-treatment of human aortic primary cell monolayers with PGG drastically reduced the adherence of THP-1 monocytes to both HAoECs, HAoSMCs (Figure 1), suggesting PGG modulates cell-macrophage interactions. HAoEC & HAoSMC monolayers after 72 h of treatment with Vehicle or PGG and incubation with THP-1 monocytes captured with phase contrast imaging at 10x total magnification. ns: p > .05, ***: p < .001 (N = 3).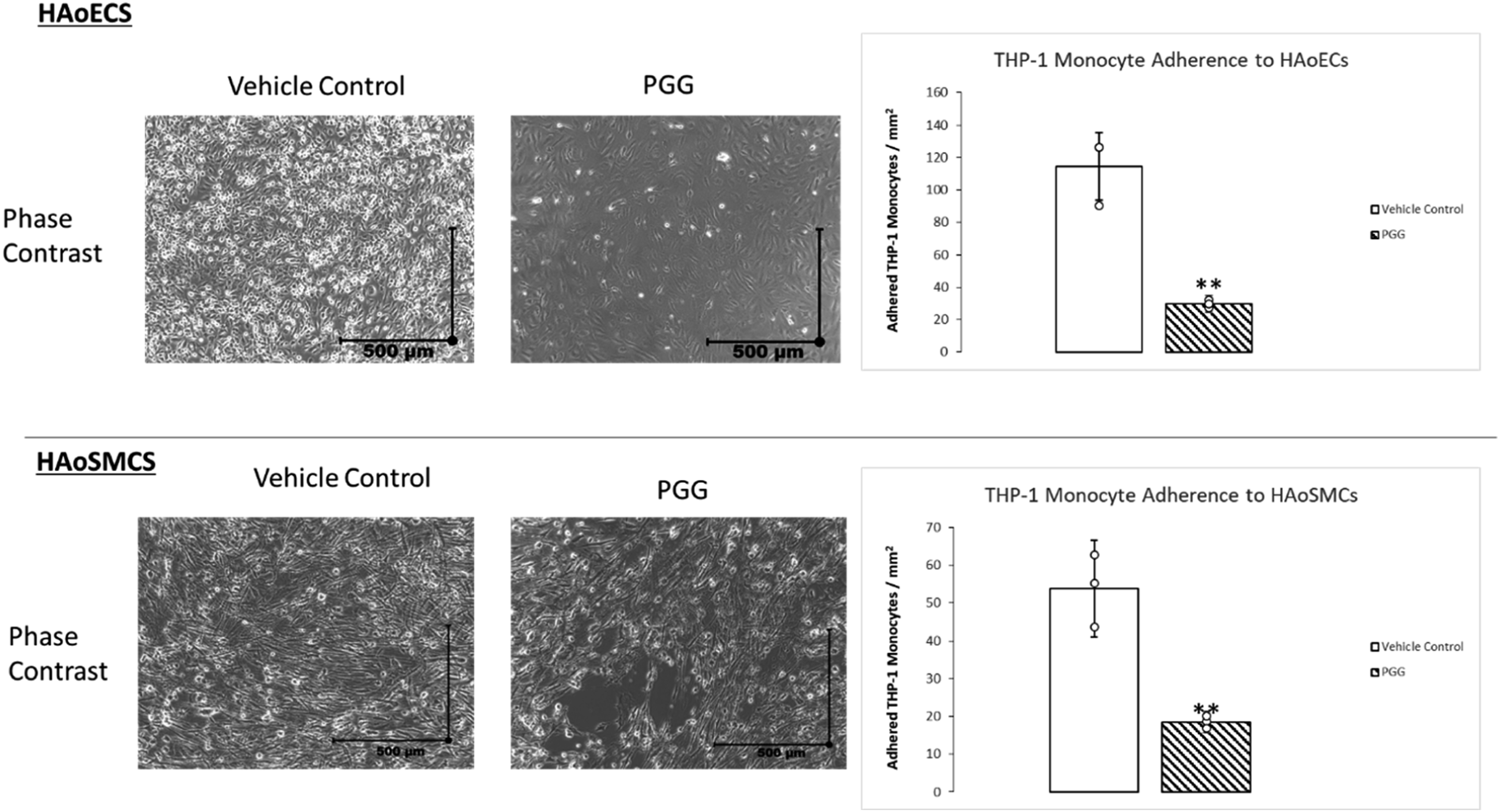
Pentagalloyl glucose induces characteristics of anti-inflammatory macrophage polarization and suppresses expression of phosphorylated NF-κB p65 subunit
To determine if PGG affects anti-inflammatory macrophage polarization, macrophages were examined after 72 h of stimulation with a vehicle or 10 µg/mL PGG. As shown in (Figure 2), the expression of CD80 was low on THP-1-derived macrophages, and the addition of PGG drastically increased the expression of CD206 and the proportion of CD206-expressing cells. Results of macrophage inflammatory markers following stimulation with 0.1% DMSO (vehicle control) or 10 µg/mL PGG for 72 h. (A) CD206, CD80, and DAPI staining of THP-1 monocyte derived macrophages (N = 4). (B) Levels of IL-10 and TNF-α in THP-1 derived macrophage conditioned medium (N = 12). (C) Phosphorylated p65 NF-κB staining of THP-1 monocyte-derived macrophages (N = 3). *: p < .05, **: p < .01, ***: p < .001.
Pentagalloyl glucose increases mitochondrial ROS and polarization of macrophages
To determine the antioxidant properties of PGG in macrophages derived from THP-1 monocytes CellROX, MitoSOX, and TMRM staining was performed. Stimulation of macrophages post PMA differentiation macrophages with PGG for 3 days differentially affected cellular and mitochondrial ROS, suppressing the former and increasing the latter associated with an increase in mitochondrial polarization (Figure 3). Previous studies have shown that PMA stimulation does not affect total ROS production in THP-1 derived macrophages.
34
CellROX, MitoSOX, and TMRM staining of THP-1 monocyte-derived macrophages following differentiation and 72-h stimulation with 0.1% DMSO (vehicle control) or 10 μg/mL PGG (N = 3). ns: p > .05, **: p < .01, ***: p < .001.
Pentagalloyl glucose reduces macrophage conditioned medium induced ROS in HAoECs and HAoSMCs
Given that increased oxidative stress is observed in many vascular diseases, we sought to determine if macrophages increased this effect and if this could be suppressed by PGG.
35
Exposure to a macrophage medium broadly induces cellular and mitochondrial ROS production in HAoECs and HAoSMCs, which is blunted by the addition of PGG. While a reductive trend was observed, PGG did not significantly reduce mitochondrial ROS in HAoECs (Figure 4). HAoEC (A) and HAoSMC (B) monolayers stained with CellROX and MitoSOX after treatment with macrophage-conditioned medium for 72 h captured with Cy5 filter at 50x total magnification. Exposure to a macrophage-conditioned medium drastically increased HAoSMC cellular and mitochondrial ROS while PGG significantly repressed these effects ns: p > .05, ***: p < .001 (N = 3).
Pentagalloyl glucose mitigates macrophage-conditioned medium-induced cytotoxicity and differentially affects the proliferation of HAoECs and HAoSMCs
PGG addition suppressed cytotoxicity in HAoECs and HAoSMCs; however, proliferation was suppressed in HAoSMCs while proliferation was increased in HAoECs, as seen in Figure 5. HAoEC (A) and HAoSMC (B) monolayers were stained for Live/Dead after treatment with a macrophage-conditioned medium for 72 h and captured with Cy5 and GFP filters at 10x total magnification. Exposure to a macrophage-conditioned medium drastically increased HAoSMC proliferation and cytotoxicity, while PGG significantly repressed these effects (N = 3). Ns: p > .05, **: p < .01, ***: p < .001.
Pentagalloyl glucose reduces macrophage-conditioned medium and elastase-induced TGF-β release
TGF-β secretion in the medium before and after conditioned medium stimulation of HAoECs (N = 9) or HAoSMCs (N = 5 or 6) was evaluated by ELISA (Figure 6(A)). No significant difference was observed between untreated and media control cells. TGF-β was not detected in PMA-induced macrophages or THP-1 monocytes conditioned medium alone (Data not shown). Similar effects were seen in aortic organ cultures. Elastase treatment increased the expression of TGF-β, and PGG treatment of the aorta significantly reduced it (Figure 6(B)). (A) Levels of TGF-β in macrophage/HaoEC and HAoSMC conditioned medium analyzed via ELISA. Exposure to a macrophage-conditioned medium drastically increases the expression of TGF-β from HAoECs and HAoSMCs, which is suppressed by the addition of PGG. (N = 6). (B) Levels of TGF-β in ex vivo aorta conditioned medium analyzed via ELISA. Exposure to elastase drastically increases the expression of TGF-β, which is suppressed by the addition of PGG (N = 4). *: p < .05, **: p < .01, ***: p < .001.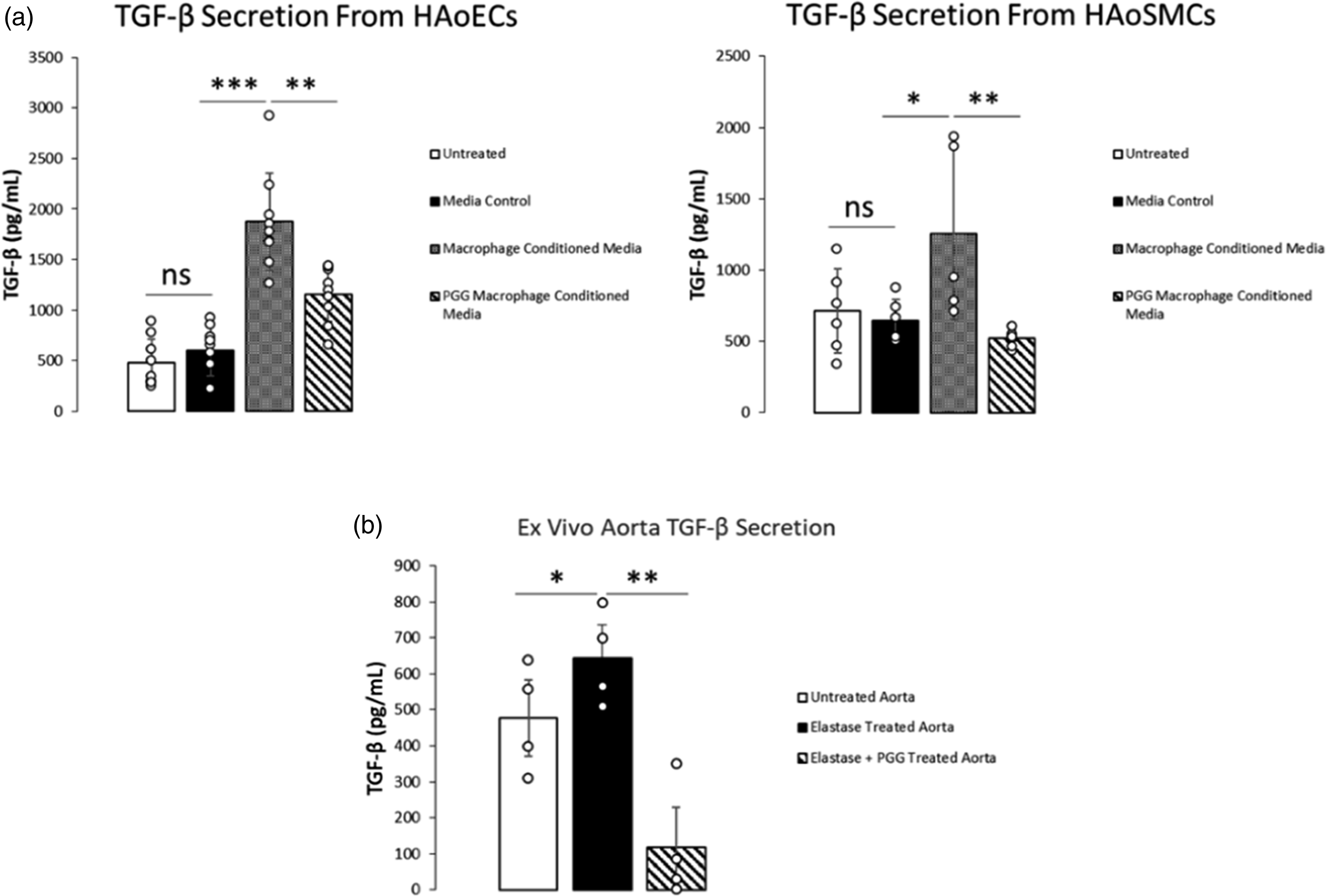
Discussion
In this study, we sought to determine underlying cellular mechanisms behind the effect of PGG on macrophage-induced inflammation observed in vascular diseases such as AA. PGG pre-treatment of HAoECs and HAoSMCs significantly prevented THP-1 monocyte adhesion (Figure 1). The effect of PGG to reduce monocyte adhesion is likely a key factor in its vaso-protective capabilities, reducing the number of infiltrating monocytes and their subsequent differentiation to macrophages.
PGG also drastically increased the expression of CD206 (mannose receptor), a classical surface marker of anti-inflammatory polarization, in THP-1-derived macrophages (Figure 2(A)). Subsequently, we observed an increase in IL-10 secretion as well as a reduction of TNF and phosphorylated NF-κB p65 subunit expression PGG treated macrophages (Figure 2(B) and (C)). Given their well-known roles in inflammation associated with AA, the reduction of TNF and increase in IL-10 secretion from macrophages by PGG stimulation may contribute to the resolution of abdominal AA in vivo. Single nucleotide polymorphisms of TNF-α (−308 G/A, rs1800629) are associated with increased plasma levels of TNF-α as well as increased risk of abdominal AA incidence among heterozygous carriers.36,37 Overexpression of IL-10 or suppression of TACE/TNF-α signaling by genetic knockdown or pharmacological antagonism can inhibit AA growth, elastin degradation, and macrophage infiltration in mice.38–41 There is substantial regulatory feedback between these elements which are implicated in macrophage biology and AA pathogenesis. In elastase-perfused rabbits, IV administration of recombinant IL-10 resulted in reduced aneurysm growth associated with the reduction of NF-κB and the presence of arginase-1 positive macrophages, another marker of anti-inflammatory polarization. 42 Subsequent knockdown of p50 or p65 NF-κB subunits using siRNA-loaded NPs mitigated aneurysm growth and mortality due to aortic rupture in elastase/anti-TGF beta mouse models associated with reduction of infiltrating macrophages as well as TNF-α. 26 IL-10 overexpression increased the number of mannose receptor (CD-206) positive macrophages, reducing TNF-α expression in aneurysmal tissues. 40 Direct intraperitoneal or intravenous administration of CD-206 expressing macrophages to AngII-infused or CaCl2 injured models of AA in mice prevented the further growth of abdominal AAs and was associated with increased expression of IL-10 in aneurysmal tissues.43,44 Thus, PGG-mediated macrophage alteration likely participates in AA resolution.
It was observed that PGG treatment reduced macrophage cellular ROS but increased mitochondrial ROS and mitochondrial membrane potential (Figure 3), characteristic of M2 anti-inflammatory type polarization. Oxidative stress contributes to AA pathogenesis; however, specific manipulation of cellular and mitochondrial reactive oxygen species (ROS) may influence the polarization of anti-inflammatory macrophages. It has been shown that PGG and other polyphenols possess broad ROS scavenging capabilities; however, this effect cannot account for the observed anti-inflammatory effects in macrophages. It was shown previously that broad scavenging of ROS in bone marrow-derived macrophages by N-acetyl-
Exposure to a macrophage-conditioned medium drastically increased cytotoxicity and cellular ROS in HAoSMC, while the addition of PGG repressed these effects. While there was a reduction trend, mtROS was not reduced significantly (Figures 4(B) and 5(B)). In HAoECs, similar effects were observed for ROS generation and cytotoxicity induced by macrophage-conditioned medium (Figures 4(A) and 5(A)). Exposure to a macrophage-conditioned medium drastically increases the expression of TGF-β from HAoECs and HAoSMCs, which is suppressed by the addition of PGG (Figure 6(A)). This confirms previous research from our lab showing a reduction of TGF-β expression in PGG-ELN-NP treated aneurysmal tissues and demonstrates that both HAoECs and HAoSMCs contribute to dysregulated TGF-β signaling induced by macrophages. Similarly, elastic fiber degradation by elastase induces an increase in TGF-β release, which is suppressed by the addition of PGG post-digestion (Figure 6(B)). In this study, it seems the primary source of TGF-β is due to cellular secretion, as PGG treatment of elastase digested aortas decreased TGF-β concentration in conditioned medium below that of undamaged aortas (Figure 6(B)).
Our results show that TGF-β dysregulation and macrophage inflammation are likely intrinsically linked processes with cells such as ECs and SMCs producing TGF-β in response to inflammatory stimuli, contributing to feedback accentuating macrophage-based inflammation. TGF-β expression and inflammatory feedback between ECs, SMCs, and macrophages is likely a critical factor in the progression or resolution of vascular diseases such as AA. In the context of the results of this study, PGG may prevent monocyte adhesion in vivo by regulating cell surface adhesion molecules and suppressing TGF-β secretion and elastin peptide fragment release (due to its binding to elastic fiber). Given the effects of PGG on THP-1-derived macrophages and subsequent conditioned medium stimulation of human vascular cells, and the reversal of AA from previous mouse studies strongly suggest PGG promotes an anti-inflammatory macrophage phenotype. The effect of PGG on macrophage polarization in vivo may be due to the reduction of TGF-β secretion from ECs and SMCs or can occur via an NFkB-dependent mechanism, as we observed in this study.
Footnotes
Acknowledgment
We greatly acknowledge support from SCBioCRAFT center at Clemson University.
Author contributions
G.H. conceived and performed all experiments in the manuscript and prepared the first draft of the manuscript. F.Z., S.A., and H.Z. helped with experimental methods, and NRV planned and oversaw the research and finalized the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health grants to NRV (R01HL133662, P30GM131959) and Hunter Endowment at Clemson University.
Ethical statement
Data availability statement
All data generated or analyzed during this study are included in this published article. The details will be made available by the corresponding author when requested.
