Abstract
An epizootic of hemorrhagic disease associated with Epizootic hemorrhagic disease virus serotype 2 (EHDV-2) infections in yaks from 5 herds occurred in Colorado between August 21 and October 3, 2012. Affected yaks presented with fever, lethargy, anorexia, dyspnea, and swollen conjunctivae. Ulcerated dental pads, mucoid sanguineous nasal discharge, petechial hemorrhages in multiple organs, pulmonary edema, and serosanguinous fluid in the thorax, abdomen, and pericardial sac were observed at necropsy. Blood and tissue samples from 8 yaks with similar clinical signs and necropsy findings were positive for EHDV-2 by reverse transcription polymerase chain reaction and 5 yaks were seropositive for EHDV. Tests for malignant catarrhal fever (Ovine herpesvirus 2), Bovine viral diarrhea virus, Bovine herpesvirus 1, Foot-and-mouth disease virus, and Vesicular stomatitis virus were negative. The findings indicate that yaks are susceptible to infection with EHDV-2 and exhibit the clinical signs, and gross and histologic lesions of hemorrhagic disease observed in other ruminant species.
Epizootic hemorrhagic disease (EHD) occurs in several native, free-ranging ruminant species in North America due to infection with Epizootic hemorrhagic disease virus (EHDV) and Bluetongue virus (BTV). 6 Cases of EHD occur on the Front Range of Colorado during the late summer and fall when climatic conditions favor the arthropod vector, Culicoides sonorensis. In addition, BTV and EHDV antibodies are detected in serum samples from domestic animals such as cattle, sheep, goats, and alpacas every year (Colorado State University Veterinary Diagnostic Laboratory [CSU-VDL], unpublished data, Fort Collins, CO) indicating that these viruses are enzootic in Colorado. Yaks (Bos grunniens) are a relatively new addition to livestock in Colorado. Hemorrhagic disease has previously been described in a 3-year-old yak from a zoo in 1970 4 ; however, the report was published at a time when the specific viral agent could not be definitively identified and the vector was unknown. The current report describes clinical disease supported by molecular virological findings, and gross and histologic lesions associated with EHDV serotype 2 (EHDV-2) infection of yaks in the United States.
On August 21, 2012, a 4-year-old yak cow (Bos grunniens) with a 2-day history of anorexia and nasal discharge was examined. On August 23, the cow and her 2-month-old calf had fever (40°C and 40.4°C, respectively), swollen conjunctivae, and increased respiratory rate. In addition, the calf had a markedly reddened nasal epithelium. The 2 affected yaks were from a group that included a bull and 2 additional cows with calves; none of the other yaks were clinically ill. The cow and calf had been to a county fair on August 1 where they were stabled next to a petting zoo with sheep. The yaks had not been vaccinated with bovine vaccines. On August 24, the owner reported that the cow had sores on her dental pad and had difficulty eating. The calf was depressed and also had sores on its dental pad as well as red, blotchy patches on its nose. Whole blood (ethylenediamine tetra-acetic acid) from the cow was submitted for Ovine herpesvirus 2 (OHV-2) polymerase chain reaction (PCR) 7 and EHDV 13 and BTV reverse transcription (RT)-PCR 14 ; the EHDV RT-PCR test was positive, and the results of the BTV RT-PCR and OHV-2 PCR tests were negative. Antibodies to Bovine herpesvirus 1 (BHV-1; commonly known as Infectious bovine rhinotracheitis virus), Bovine viral diarrhea virus 1 and 2 (BVDV-1 and -2), Bovine respiratory syncytial virus, and Bovine parainfluenza virus 3 were not detected by serum neutralization assay.
The yak calf died on August 29 and was submitted for necropsy and ancillary tests. On gross examination, foam exuded from the nares, the conjunctivae were edematous, and there were ulcerated areas on the dental pad and under the tongue. Over 90% of the lung was firm and edematous; the cranial ventral aspect was the most consolidated. Multiple petechiae were noted on the serosal surface of the rumen (Fig. 1A), epicardial surface of the heart (Fig. 1B), at the apices of the papillary muscles, and on the pulmonary artery. The liver was distinctly yellow with rounded edges. Fresh samples of lung and spleen were positive for EHDV RNA sequences and negative for BTV by RT-PCR. On September 4, 14 days after first examination, the yak cow, mother of the calf described earlier, was found in lateral recumbency, and was euthanized. At necropsy, a mucoid, sanguineous discharge exuded from both nares, and ulcers were noted on the dental pad (Fig. 1C). The lungs were diffusely edematous with foam and fluid in the airways (Fig. 1D). Several liters of fluid were found in the abdomen and thoracic cavity; approximately 200 ml of serosanguinous fluid was found in the pericardial sac. Multiple petechiae were found on the serosal surfaces of viscera, epicardial, and endocardial surfaces and extended into the myocardium. The contents of the gastrointestinal tract were dark and appeared to contain blood; however, evidence of mucosal ulceration was not found. The spleen was negative for EHDV, BTV, and BVDV by RT-PCR and negative for OHV-2 by PCR.
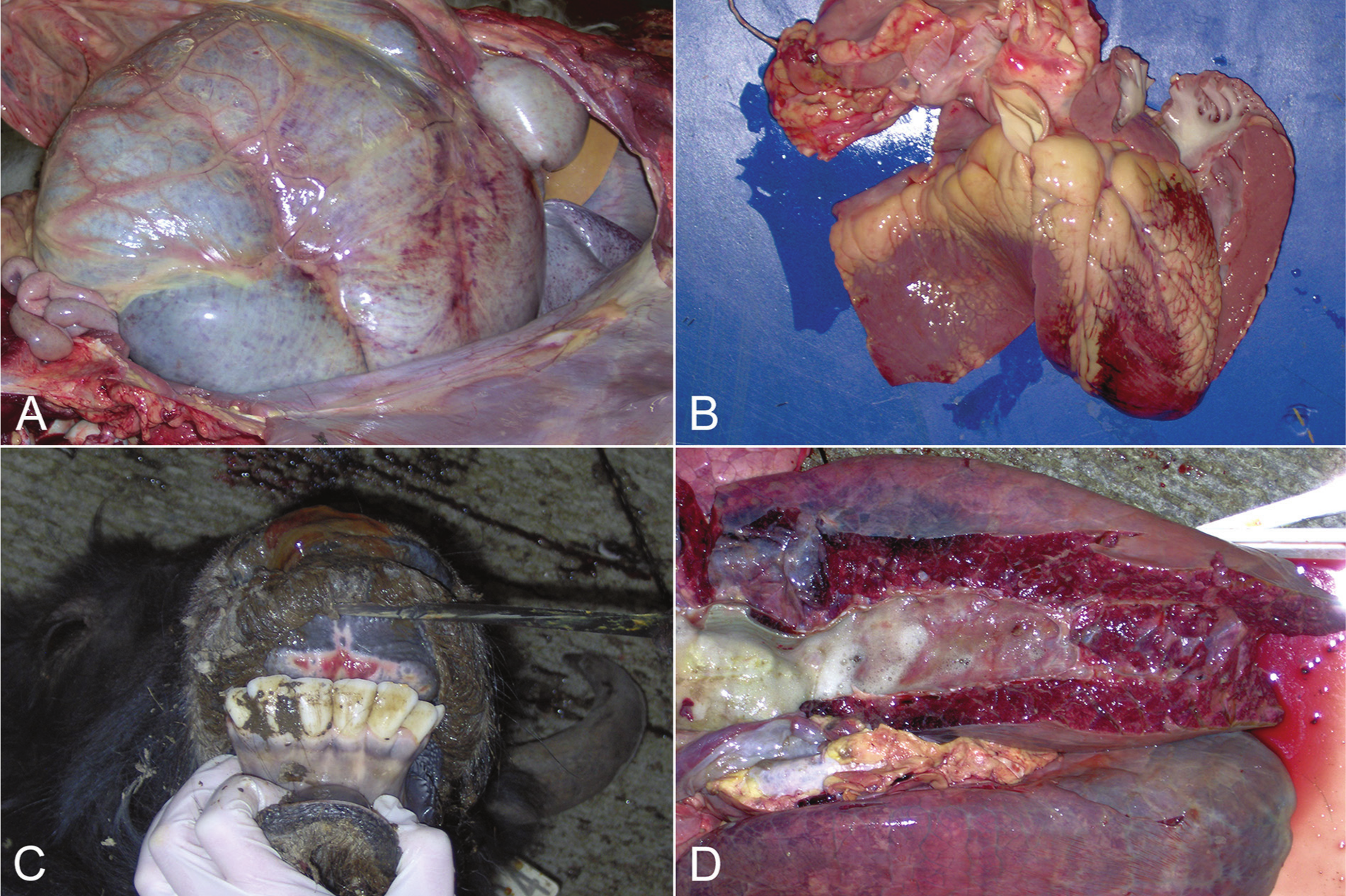
Yak (Bos grunniens).
On histologic examination, both animals had marked pulmonary edema characterized by flooding of alveolar spaces and expansion of the intralobular septa by eosinophilic, proteinaceous fluid and a variable number of foamy macrophages and neutrophils. There were also regions of acute hemorrhage into both alveoli and intralobular septae. The calf had evidence of acute aspiration pneumonia consistent with the grossly observed change in the cranial ventral portion of the lung. Areas of hemorrhage in the heart were acute, and blood vessels near the affected regions had reactive endothelial cells and perivascular edema that occasionally dissected between cardiac myocytes. Oral ulcers were more chronic in the cow, with fibrin on the surface, mixed inflammation, and reactive fibrosis within the ulcer bed. Small foci of hemorrhage were present in the mucosa, submucosa, and occasionally adjacent to blood vessels in the muscularis of the intestinal tract of the calf. Prominent bile stasis was observed in the liver of the calf. The spleen of the cow had moderate lymphoid hyperplasia with sinus histiocytosis and erythrophagocytosis.
Between August 21 and October 3, 2012, blood, serum, or tissue samples were submitted from 9 other yaks from 4 additional Colorado yak herds with similarly affected animals. One or more blood or fresh tissue samples submitted from yaks from each of the 4 herds were EHDV positive by RT-PCR. Serum samples from 1 affected animal from each of these 4 herds were strongly seropositive to EHDV antigen a and weakly seropositive to BTV by agar gel immunodiffusion. b Two of the 4 seropositive yaks had blood or tissues concurrently positive for EHDV by RT PCR, and 2 seropositive yaks were negative for viral RNA. Individual animals from these 4 herds were tested and found to be negative for OHV-2 DNA, Bacillus anthracis DNA, BVDV antigen, BHV-1 antigen, and Rabies virus antigen in brain. Two yaks from 1 herd were negative for Foot-and-mouth disease virus by RT-PCR performed at the National Veterinary Services Laboratories. To date, attempts to isolate EHDV or other viruses from blood and tissue samples have been unsuccessful.
Total RNA was extracted from spleen, lung, or the buffy coat from whole blood using a commercial kit c and following the manufacturer’s instructions. The EHDV and BTV multiplex flank RT-PCR was performed using 5 μl of RNA and a commercial kit. d The flank reaction was set up in a final volume of 25 μl using 5 μl of the RNA, 6.68 μl H2O, 0.6 μM each of EHDF1(5’-AAGTTCTTCGTCGACTGCCATC-3’), EHDR1(5’-GGTTGCACCATCTGGCCATAAT-3’), BTVF1 (5’-AGTTCTCTAGTTGGCAACCACC-3’), BTVR1 (5’-TTCACGGCTTCATCCATCATCC-3’) primers1, 1× kit buffer, d 1× Q solution, d 0.4 μM each deoxyribonucleotide triphosphate (dNTP), 1 μl of enzyme mix, and 4.8 μ of RNA inhibitor. e Cycling conditions are as follows: reverse transcription (50°C, 30 min, then 95°C, 15 min), followed by 35 cycles (95°C, 30 sec; 55°C, 45 sec; 72°C, 45 sec), and a final extension (72°C, 5 min). Each 50-μl nest reaction consists of 2 μl of the flank reaction added to a mixture of 34.3 μl of H2O, 0.6 μM of EHDF2 (5’-AGCATTATCACCACAGTGG ACG-3’), EHDR2 (5’-GCCAATCTATATGCCGCATGTG-3’), BTVF2 (5’-CACCACAATGGACTTGCAGTCA-3’), and BTVR2 (5’-GACATCCTCCTCGTATGGTTGA-3’) primers1, and 0.2 μM each dNTP, 1× PCR buffer, 1 mM of MgCl2, 2.5 units Taq, f and 0.1% Triton-X100. g Cycling conditions are as follows: initial melt (95°C, 5 min), followed by 35 cycles (95°C, 30 sec; 55°C, 45 sec; 72°C, 45 sec), and a final extension (72°C, 5 min).
All EHDV RT-PCR–positive samples were further genotyped using EHDV-2–specific primers, and all yielded EHDV-2 amplicons. 1 The EHDV-2 PCR was performed exactly as the preceding flank and nested reactions were, with the following exceptions: flank primers2 consisted of 0.6 μM of EHDV-2-VP2-49 (5’-CGCATAATAATGAAGAAGAG-3’), EHDV-2-VP2-633 (5’-AGCGTTTTGTCACATCCTG-3’), and Nest2 primers consisted of 0.6 μM of EHDV-2-VP2-170i (5’-GCTGAGAACGATAGGAGACG-3’) and EHDV-2-VP2-436i (CTAATCCAACTCGCCGC), with 50°C annealing temperature in both flank and nest cycles.
Clinical signs commonly described for EHDV-positive cases in yaks include fever, depression, inappetence, conjunctival swelling, ocular and nasal discharge, dyspnea suggestive of respiratory infection, ulcerative gingivitis of the dental pad with mild bleeding from the mouth, and varying degrees of lameness. 5 Similar clinical signs and lesions have been described in EHDV-2, 3 EHDV-6, 11 and EHDV-7 16 infections in cattle. A severe drop in milk production has been reported, 16 and abortions or the birth of deformed calves may result. 10 A similar critical gestation time frame for fetal losses due to EHDV has not been established in yaks. The list of differential diagnoses for EHD cases with hemorrhagic enteritis includes coccidiosis, Clostridium spp., Salmonella spp., or hemorrhagic bowel syndrome. For the systemic hemorrhagic lesions, BTV, Foot-and-mouth disease virus, vesicular stomatitis, malignant catarrhal fever (OHV-2), BVDV, and BHV-1 should be considered.
Several reasons may account for the apparent epizootic of hemorrhagic disease in 5 yak herds in Colorado during the late summer and fall of 2012. One explanation would be the emergence of a new serotype or strain of EHDV into a susceptible population of yaks. Exotic orbiviruses have been detected in wild species in the United States (www.usaha .org/Portals/6/Reports/2011/report-btbr-2011.pdf), and a recombinant virus, EHDV-6, appears have spread westward since 2006. 1 However, the EHDV in the yak cases presented herein has been characterized as EHDV-2, a genotype that has been isolated from domestic and wild species in the western United States for many years. 12 Identification of EHDV-2 does not exclude the possibility of infection with a more virulent strain of the same genotype, such as Ibaraki virus. 10 To determine the genetic relatedness between the 2012 yak EHDV-2 and other isolates will require viral RNA sequence comparisons.
A second explanation for the EHD cases may be found in meteorological conditions for the year 2012, which was the warmest on record in Colorado and other states, and the driest since 2002 (http://www.ncdc.noaa.gov/temp-and-precip/ranks.php). A notable number of EHD cases were observed in white-tailed deer (Odocoileus virginianus) and other wild ruminants in multiple states in 2012 (http://www.nwhc.usgs.gov/publications/wildlife_health_bulletins/WHB_2012-05_Hemorrhagic.pdf). The relationship between temperature and the life cycle of Culicoides spp. vector is well established; higher temperatures result in a decreased extrinsic incubation period, increased number of eggs laid per female,8,15 and increased amounts of BTV antigen per female, 9 culminating in an increased transmission potential for the virus. Not surprisingly, there is a direct relationship between temperature and risk of infection. 2
The possibility that EHD may have occurred in yaks previously, but was not recognized, is suggested by a single necropsy report from the records of CSU-VDL. On November 6, 2007, a 7-month-old male yak was submitted for necropsy. The yak was examined by a veterinarian approximately 1 month before death for oral lesions and, after testing, was found to be negative for vesicular stomatitis. The owner noted lethargy and inappetence prior to death. At necropsy, multiple ulcers were noted on the lips, nose, and soft palate; pulmonary congestion and edema, splenic lymphoid depletion, and petechial hemorrhages on the surface of the heart and within the endocardium were observed. Virus isolation, as well as BTV and EHDV RT-PCR, performed on pooled tissues were negative, possibly reflecting the time between the onset of clinical signs to necropsy. Gross and histologic lesions similar to the cases presented in the current report were recorded, with the exception of a chronic active hepatitis.
The negative EHDV RT-PCR result on splenic tissue from the yak cow, obtained 14 days after first examination and 11 days after the EHDV RT-PCR–positive blood sample was obtained, may be the result of clearance of the virus by the immune response. Cattle have a prolonged viremic stage of BTV infection in the absence of clinical signs; however, the period of viremia in yaks is not known. Experimental infection studies are needed to determine the pathogenicity of EHDV-2 for this species as well as to define the period of viremia, timing of the immune response, and onset of disease. In conclusion, yaks are susceptible to infection with EHDV-2 and exhibit the clinical signs and gross and histologic lesions of hemorrhagic disease observed in other ruminant species.
Footnotes
Acknowledgements
The authors thank Drs. Keith Roehr and Greg Farrand for furnishing clinical observations and samples from suspect cases, and Drs. Alana Garner and Paula Schaffer for their assistance with the necropsies. The authors thank Dr. William C. Wilson for helpful discussion and for advice on virus isolation and RNA extraction methods.
a.
Catalog no. E-01, Veterinary Diagnostic Technology Inc., Denver, CO.
b.
Catalog no. B-01, Veterinary Diagnostic Technology Inc., Denver, CO.
c.
RNeasy Mini Kit, Qiagen Inc., Valencia, CA.
d.
OneStep RT-PCR Kit, Qiagen Inc., Valencia, CA.
e.
RNasin, Invitrogen Corp., Carlsbad, CA.
f.
Biolase Taq Kit, Bioline USA Inc., Taunton, MA.
g.
Sigma-Aldrich, St. Louis, MO.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
