Abstract
An 8-y-old lactating Holstein cow was presented to the herd veterinarian with a history of acute ataxia, and because of poor prognosis, the animal was euthanized and submitted for postmortem examination. A lumbar vertebra was incompletely fractured and had an adjacent subdural hematoma. The pancreas had a hard texture with exaggerated nodular appearance, and on cut surface, the pancreatic ducts appeared fibrotic and contained scores of round-to-faceted pancreatoliths of 0.5–2.0 mm diameter. These calculi extended from the major ducts to the pancreatic duct, sphincter, and into the duodenal lumen. Microscopically, the pancreas had severe acinar atrophy and dissecting glandular fibrosis. The major pancreatic ducts were ulcerated with fibrosis and inflammation. In humans, chronic pancreatitis has been linked to osteoporosis and increased risk of bone fractures. Manual evaluation of ribs at the time of autopsy did not reveal abnormalities in bone pliability or fragility in our cow.
An 8-y-old lactating Holstein cow with no previous indication of ill health was presented to the herd veterinarian because of ataxia, originating from what the owner presumed was an acute traumatic injury. On clinical examination, the cow had an exaggerated arched back and was reluctant to move but was ambulatory. She exhibited an exaggerated posture with hindlimbs slightly extending behind her and almost rigid; there was minimal flexion of the stifle and hock bilaterally. Injury was suspected because there was some degree of heat and swelling over the lumbar vertebrae. The prognosis given for the return to full function was poor, and the owner elected euthanasia and autopsy.
On postmortem examination, the cow was in good body condition with no obvious external abnormalities other than some lumbar swelling. Noteworthy gross anatomic lesions were limited to the pancreas and spinal column. The pancreas had a hard texture with exaggerated nodular appearance, and on cut surface, major pancreatic ducts appeared fibrotic and filled with a thick paste composed of myriads of calculi (Fig. 1). The concretions were homogeneously white with a rock-like texture and varied from rounded-to-faceted, 0.5–2.0 mm diameter. The calculi extended down the pancreatic duct and sphincter, and into the duodenal lumen (Fig. 1). The longissimus dorsi and ventral psoas muscles were hemorrhagic and edematous. Further dissection of the spine revealed an incomplete fracture of vertebra L4 involving the right transverse process and part of the vertebral body. A parasagittal section of the lumbar spine exposed a localized (5 × 1 cm) subdural hematoma adjacent to the fracture site. The skeleton was examined carefully, but there was no further gross evidence of fractures or bone calluses. Tissues were fixed in formalin and processed for histopathology.
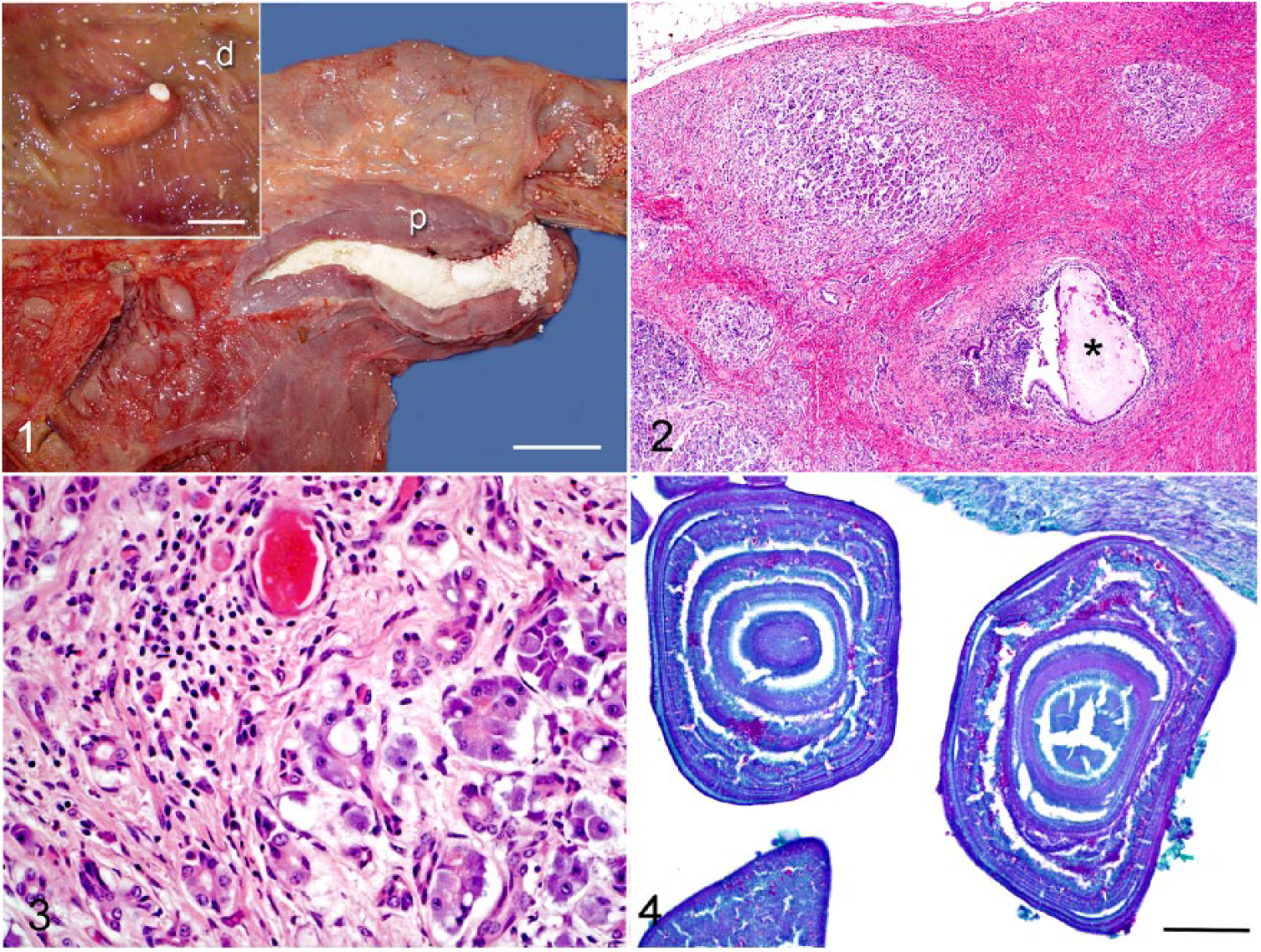
Pancreatolithiasis in an 8-y-old Holstein cow.
Microscopic examination of demineralized pancreas revealed moderate-to-severe atrophy and dissecting fibrosis of pancreatic acini (Figs. 2, 3). Pancreatic ducts were distended, and their walls were thickened by fibrosis and moderate infiltration of eosinophils, lymphocytes, and plasma cells. The ductal epithelium was mostly ulcerated but showed occasional foci of epithelial hyperplasia. Neutrophils, eosinophils, and lymphocytes also infiltrated the underlying lamina propria, particularly around the ulcerated epithelium. The ductal lumens contained many acellular laminated bodies composed of amorphous amphophilic material (Fig. 4). The pancreatoliths were non-birefringent, and negative by von Kossa and periodic acid–Schiff (PAS) staining, except for pinpoint material between the laminated plates that stained positively with PAS (Fig. 4).
Pancreatic calculi have been recognized in human and veterinary medicine for many years.1,12 The clinical condition associated with pancreatic calculi is known as “pancreolithiasis” or “pancreatolithiasis,” and the stones are referred to as pancreatoliths (Greek lithos = stone).19,20 Although rare in veterinary medicine, pancreatolithiasis is documented in cattle and cats,18–20 and has been reproduced experimentally in dogs. 16 According to one report, 19 the first account of bovine pancreatolithiasis dates back to 1846.
Pancreatolithiasis in our cow was regarded as an incidental finding because clinical signs leading to euthanasia were purely neurologic, and had resulted from a lumbar fracture. Bovine pancreatolithiasis is asymptomatic, 8 and generally an incidental finding during autopsy or detected by meat inspectors at the abattoir.1,11 Studies of adult cattle revealed that the frequency of pancreatolithiasis at the abattoir was 2.6% in Rumania and 0.82% in Denmark,1,19 and the weight of individual pancreatic calculi was 0.2–24 g. 1 The incidence of pancreatolithiasis in North America is unknown, and this condition is likely overlooked during postmortem examination unless the gross pancreatic lesions are sufficiently severe to prompt close inspection of the gland by the pathologist, as was the case for our cow. The mineral composition of the calculi was not determined in our cow, but mineral analyses of human, bovine, and feline pancreatoliths showed that the calculi were mainly calcium carbonate and calcium phosphate.1,19,20 The calculi in the cow in our study were negative by von Kossa stain, but the pancreatic tissue had been demineralized for histologic examination. As reported previously for cattle and humans, pancreatic calculi were associated with chronic ductal inflammation and chronic fibrosing pancreatitis.1,2 Bovine pancreatic calculi were initially regarded as the cause of pancreatic ductal inflammation rather than the result of inflammation, as is now widely accepted.11,19
The pathogenesis of pancreatolithiasis is incompletely understood, but for many years it has been considered a sequel to chronic pancreatitis in which intraductal exudate and locally secreted proteins form a plug and act as nidi for concentric stone formation.10,15 Some studies estimate that as many as 50% of human patients with chronic pancreatitis develop pancreatoliths. 17 Pancreatitis, the prelude of pancreatolithiasis, starts with the stagnation of pancreatic secretions known as pancreastasis, which results in abnormal discharge of pancreatic enzymes through the basolateral membrane, rather than through normal apical exocytosis. 2 These events trigger a surge of reactive oxygen species, and make pancreatic acinar cells a perpetual source of chemokines and cytokines. 3 The anomalous basolateral release of pancreatic enzymes and release of chemokines and cytokines causes vicious cycles of local inflammation and unsuccessful repair leading eventually to pancreatic atrophy, fibrosis, and intraductal mineralization. 2 Chronic pancreatitis often progresses to endocrine dysfunction and diabetes mellitus in humans, but these sequelae have not been investigated in cattle. 17
A protein known as lithostathine, or pancreatic stone protein (PSP) or regenerating protein (Reg), is synthesized by pancreatic acinar cells, and secreted into pancreatic ducts where it mixes with other pancreatic secretions.4,17 Lithostathine is the main protein present in pancreatoliths. 4 Lithostathine is a non-enzymatic protein found in the pancreatic juice of humans and animals,4,5 and for many years this protein was believed to inhibit calcium carbonate precipitation, thus preventing formation of pancreatic calculi. 13 However, subsequent investigations have put into question the real role of lithostathine in preventing formation of calculi. 5 Lithostathine has been identified in the pancreatic secretions of rodents, dogs, pigs, and cattle, but its beneficial effect in preventing lithogenesis remains unexplored. 4 Information regarding the pathogenesis of bovine pancreatolithiasis is sparse, but it most likely follows the triad of pancreastasis–pancreatitis–pancreatolithiasis as reported in human medicine.2,15 Pancreatoliths and chronic pancreatitis always occur together in cattle.11,19
In humans, chronic pancreatitis is a well-recognized risk factor for osteopenia and osteoporosis, which may result in bone fragility and susceptibility to pathologic fractures.6,14 According to one study, one-third of human patients with chronic pancreatitis develop osteoporosis. 6 The mechanism by which chronic pancreatitis predisposes the skeleton to develop osteoporosis is still debatable, but some researchers suggest nutritional malabsorption as a result of abnormal enzymatic digestion. 6 Epidemiologic studies ruled out malabsorptive vitamin D deficiency in patients with pancreatitis and osteoporosis, but suggested a possible relationship between pancreatitis and systemic inflammation with abnormal bone remodeling.7,9
Another Holstein cow with ataxia caused by a lumbar fracture that also had pancreatitis and pancreolithiasis was reported in Alberta, Canada in 1994. 8 This 1994 report along with our findings could suggest that, as demonstrated in humans, a connection exists between chronic pancreatitis–pancreatolithiasis with bone fractures in cattle. Subjective evaluation by manually bending the ribs at the time of autopsy did not reveal abnormalities in bone pliability or fragility. However, definitive diagnosis of osteoporosis would have required a bone density scan or stereologic analysis of bone trabeculae, which were not performed in our cow.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
