Abstract
Bovine tuberculosis (TB) is endemic in Kuwait; cattle identified as TB-positive using the caudal fold test (CFT) are culled. We used a Bayesian approach to estimate the sensitivity (Se) and specificity (Sp) of the IFNγ assay and ELISA, which are not routinely used in Kuwait in CFT-negative dairy cattle. Blood samples from CFT-negative cattle (
Between May 2014, and November 2016, blood samples (10 mL) from 38 dairy farms were collected into heparinized tubes immediately before cattle were tested by CFT. Samples were collected and transported to the laboratory to avoid the extremely hot temperatures of Kuwait. Immediately upon arrival at the laboratory, within 4–6 h after collection, blood samples were divided into 3 aliquots that were stimulated using bovine and avian purified protein derivative (PPD) and phosphate-buffered saline (PBS), and incubated overnight at 37ºC in a humidified atmosphere as described elsewhere.
18
Samples were then centrifuged, and plasma was recovered and stored until the CFT-negative status of the animals was demonstrated, which was the case for 384 animals. Subsequently the IFNγ assay (Prionics, Schlieren, Switzerland) and ELISA (IDEXX Laboratories, Westbrook, ME) were performed according to manufacturers’ instructions. Although this was a convenience sample, and the number of samples that would be CFT-negative was unknown at the time of collection, the number of CFT-negative cattle (
A Bayesian approach was used to estimate the Se and Sp of the ELISA and the IFNγ test.
2
,
3
,
8
,
14
Bayesian approaches have been used previously to estimate the accuracy of TB tests in endemic settings
1
given the lack of a reliable gold standard test for the disease. Model parameters (including test Se and Sp and disease prevalence [
Prior distributions and source of data used to estimate the accuracy of 2 bovine tuberculosis tests in caudal fold test–negative cattle of Kuwait using a Bayesian latent class model and in the absence of a gold standard. Uncertainty was measured as the 95% confidence level that the parameter was higher (>) or lower (<) than a certain value.
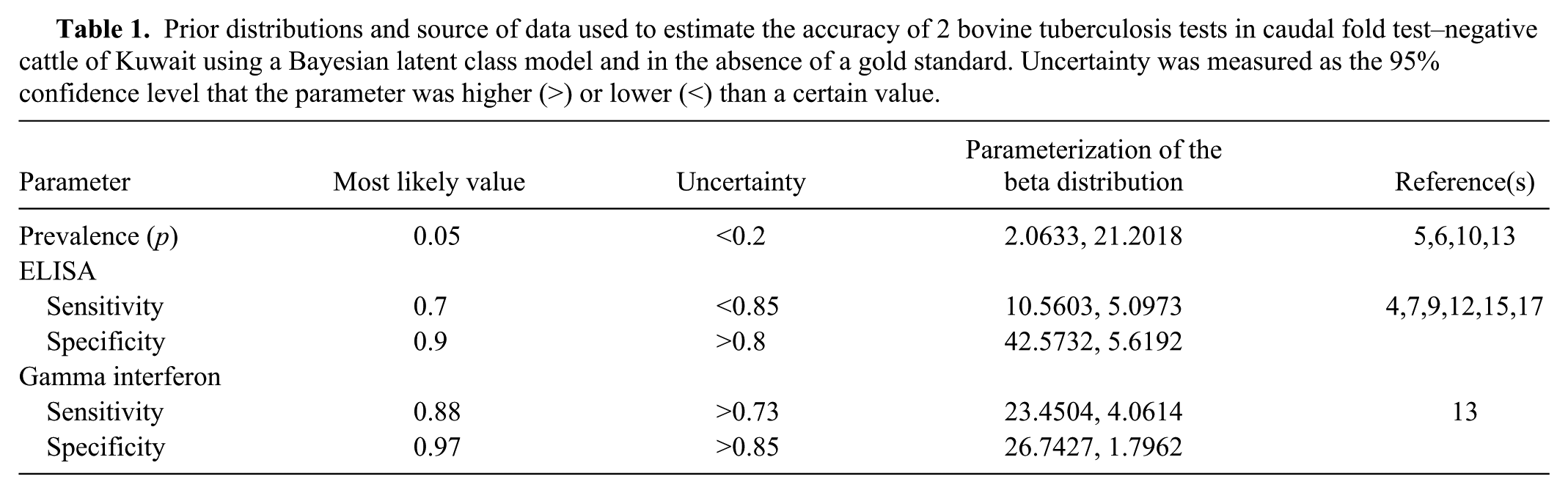
Because Kuwait is a small country, no TB testing occurs prior to animal movement, and most farms are small (<5 cattle per farm), a single population was assumed. Results from the IFNγ assay and the ELISA may be independent because they are based on the detection of cellular and humoral immune responses, respectively. However, IFNγ and ELISA results may still be correlated and, thus, we preferred to explicitly assess the correlation between test results. 2 , 3 Robustness of the model to the definition of the prior distributions was evaluated by alternatively assuming uniform (0, 1) prior distributions for the parameter values. All calculations were performed using WinBUGS software, 8 and results were computed for 200,000 iterations of the model after the initial 500 iterations were discarded. The autocorrelation and trace plots of the simulations were visually assessed, and the Gelman–Rubin statistic was computed to evaluate model convergence.
There were 55 (14.3%) IFNγ assay–positive, 40 (10.4%) ELISA-positive, and 5 (1.3%) IFNγ-and-ELISA–positive samples among CFT-negative dairy cattle of Kuwait. The remaining 284 (74.0%) samples were negative to both tests. The estimated (posterior) Se and Sp of the IFNγ assay were 85.0% (95% CI: 67.6–95.3%) and 90.4% (86.7–95.3%), respectively. Results are similar to those reported elsewhere, with Se and Sp of the IFNγ assay estimated to be 73.0–100% and 85.0–99.6%, respectively. 6 , 13 Slightly lower values were estimated for the Se (61.1%; 95% CI: 33.1–84.6%) and Sp (85.4%; 81.7–88.8%) of the ELISA, compared to the IFNγ assay, in agreement with a number of studies that reported a substantial variation in ELISA results. 4 , 7 , 9 , 12 , 13 , 15 , 17 Disease prevalence (95% CI) in CFT-negative cattle was estimated as 2.6% (0.5–9.5%). Different PPDs were used for the IFNγ assay (Tuberculin, Prionics, Lelystad, The Netherlands) and the CFT (Bovituber, Synbiotics, Lyon, France), which may explain, at least in part, the differences in the test results. Furthermore, extreme weather conditions of Kuwait challenged the requirement of keeping samples at room temperature. Samples were refrigerated to resemble conditions observed in countries where the IFNγ assay was standardized; however, it is unclear how temperature and storage conditions may have affected our results. As expected, the correlation between the ELISA and IFNγ assay was nil, with median values approximating 0 for both infected and uninfected animals. Such findings may be related to detection of infection status at different stages, 11 leading to an opportunity for in-parallel use and interpretation of those tests, particularly the IFNγ assay, to increase TB diagnostic sensitivity in Kuwait. Results were sensitive to the prior parameterization of the model, and thus, validity of the model is conditional to the values assumed for the prior distributions, which is an assumption of Bayesian analysis. In alignment with World Organization for Animal Health recommendations, 19 combined and systematic application of CFT and IFNγ may improve bovine TB control in Kuwait.
Footnotes
Acknowledgements
The support provided by the management and other departments of the Kuwait Institute for Scientific Research is gratefully acknowledged. We thank the Public Authority for Agriculture Affairs and Fish Resources (PAAFR) and the Kuwait Union of Dairy Producers for technical cooperation provided during the study, and Dr. Y Al-Azemi for technical assistance. We are grateful to OMICS RU project SRUL02/13 in the RCF Research Core Facility at Kuwait University for the use of unit facilities.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Research was funded by the Kuwait Foundation for the Advancement of Sciences (KFAS).
