Abstract
The gamma interferon (IFN-γ) test has been used for many years as an ancillary test in the detection of bovine tuberculosis. We investigated the effect of skin testing and the length of time between blood collection and processing on the performance of the IFN-γ test. A series of blood samples were taken from groups of experimentally infected cattle (
The gamma interferon (IFN-γ) test used in the detection of tuberculosis in cattle was developed in the early 1990s. 15 Subsequently, there has been widespread use of this test as an ancillary procedure to intradermal skin testing. New Zealand was the first country to use the IFN-γ test as a routine procedure in a bovine tuberculosis control program. 8 Validation studies were carried out in New Zealand to determine how best this test could be adapted to meet the local conditions, including the transport of samples to a central laboratory and the use of the caudal fold skin test. 12 The major modification to the test was to process blood samples up to 30 h postcollection, enabling the use of overnight courier services for the forwarding of samples to a central laboratory. In response to a concern that prior skin testing could affect the responses to the IFN-γ test, routine testing was restricted to 8 d following caudal fold skin testing. This condition was based on limited studies 11 that found inhibition of responses immediately following skin testing. Further studies have cast doubt on the effects of skin testing on IFN-γ responses. 14 A confounding factor was whether cattle were tested with the comparative cervical skin test, which used bovine and avian purified protein derivatives (PPDs), or the caudal fold test, which used only bovine PPD. In a 2010 review, 14 a need was identified for further studies investigating the effect of skin testing on IFN-γ testing. We therefore investigated factors that could be modified to make the test more practicable, including prior skin testing and delays in the processing of blood samples. Currently, delays in overnight courier deliveries result in some samples arriving at the laboratory >30 h postcollection, which requires animals to be rebled. These effects were examined using both experimentally and naturally infected cattle.
Twenty-four 15-mo-old, Friesian-cross, female cattle were obtained from a herd that was accredited free of tuberculosis and came from areas in New Zealand where both farmed animals and wildlife were free of bovine tuberculosis. The cattle were moved to a biocontainment unit where they grazed on pasture. Twelve of the heifers were randomly selected and challenged with 5,000 colony-forming units of
The experimentally and naturally infected cattle kept at the containment unit were skin tested by a licensed animal technician from AsureQuality by intradermal injection of bovine PPD
d
into the caudal fold. The skin tests were read 3 d postinjection (dpi), and the changes in skin thickness were measured with calipers. Any increase in skin thickness in comparison with the opposite caudal fold was regarded as a positive response. The experimentally infected cattle were skin tested 14 wk after inoculation with
Heparinized blood samples were taken on the day of tuberculin injection, 3 d later when the skin test was read, and 11–19 dpi. They were tested using procedures described previously. 12 The antigens used in the IFN-γ test were bovine and avian PPDs (22 μg/mL final concentration) and a combination of ESAT6 and CFP10 c (4 μg/mL final concentration). Phosphate-buffered saline was added to the blood sample for the negative control. The incubation of blood cultures was standardized to 18 h, and plasmas from the same animal were assayed for IFN-γ on the same enzyme-linked immunosorbent assay (ELISA) plate in order to eliminate any between-plate variation. Results are expressed as optical densities (OD) at 450 nm. The standard criteria used for designating positive responses were as follows: for the PPDs, bovine PPD minus avian PPD ≥0.100 OD450 (standard interpretation) and bovine PPD minus avian PPD ≥0.04 OD450 (modified interpretation); and ESAT6/CFP10 minus negative control ≥0.040 OD450 (standard interpretation) for the refined antigens (https://goo.gl/cBP3lQ).
Following the investigations, the animals were slaughtered and examined for bovine tuberculosis. All animal procedures were approved by the AgResearch Grasslands Animal Ethics Committee, permit 12378. Macroscopic lesions consistent with infection with
The statistical analyses of the IFN-γ data within each animal group were based on the bovine PPD minus avian PPD and the ESAT-6/CFP10 minus negative control OD450 values. There were insufficient animals to undertake a statistical analysis of differences in the proportions of animals responding positively within the groups. Comparisons of the IFN-γ responses were undertaken for blood samples collected from individual animals processed at different times and at different days; responses between groups (control, experimentally and naturally infected animals) were not undertaken because of the large between-animal variation. The IFN-γ data were analyzed by a mixed-effects model with fixed effects, day, time, and their interaction, random effects of animal, and day nested within animal. To match the normality assumption of the model, the ESAT-6/CFP10 minus negative control values for the control and experimentally infected animals were natural log transformed. The ESAT-6/CFP10 minus negative control values for the naturally infected animals and the bovine PPD minus avian PPD values for the experimentally infected animals were square-root transformed; no transformation was required for bovine PPD minus avian PPD values for the control and naturally infected animals. Multiple comparisons for the predicted means of the different time in each day and different day at each time were performed with a
None of the 12 animals from the uninfected control group had a positive skin test. All of the
The differential, bovine PPD minus avian PPD, and ESAT6/CFP10 minus negative control IFN-γ responses of the 12 uninfected controls and the 2 groups of
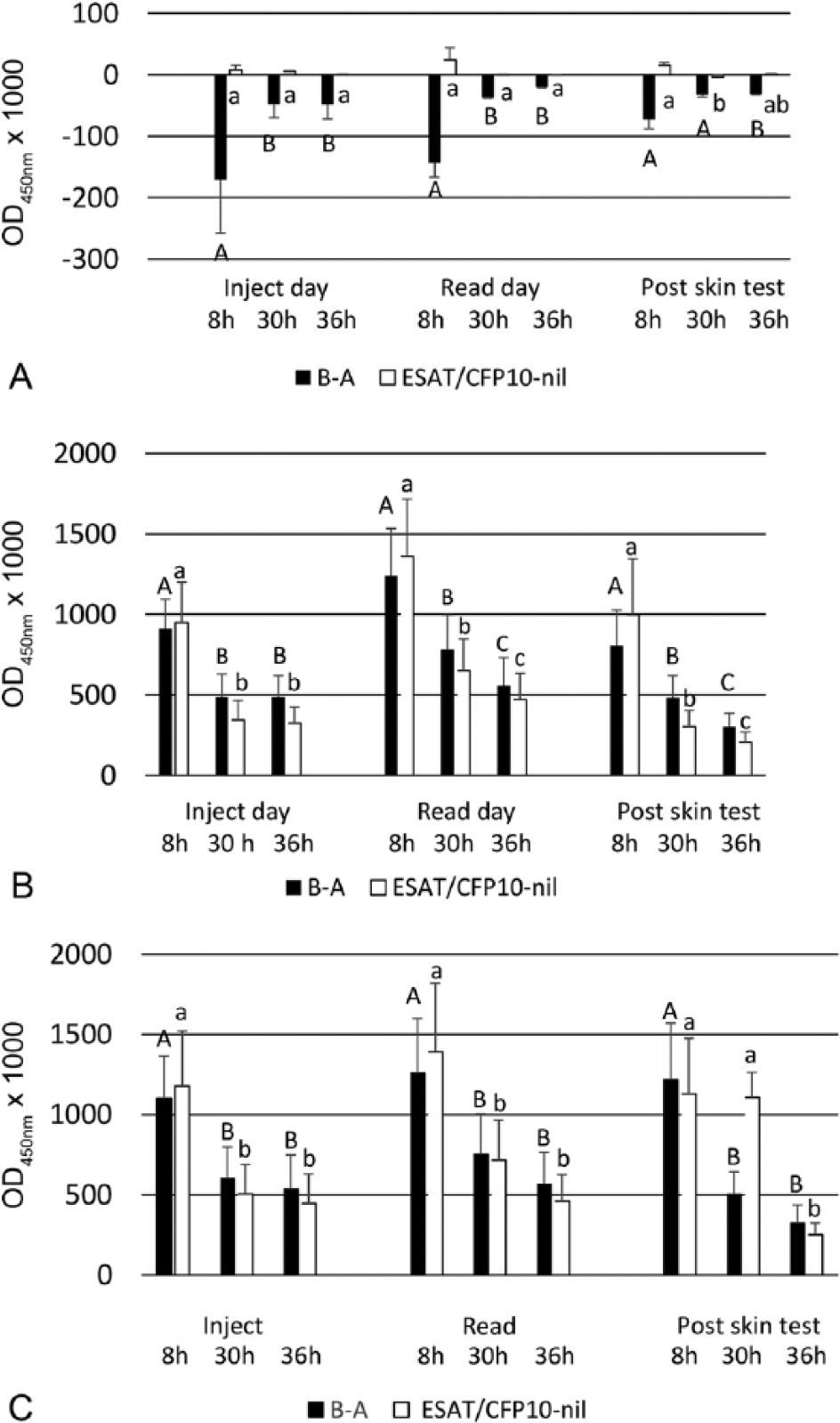
Gamma interferon responses shown as optical density at 450 nm (OD450) of bovine purified protein derivative (PPD) minus OD450 of avian PPD and OD450 of ESAT6/CFP10 minus OD450 of negative control. Values represent mean ± standard error of the mean for (
Summary of the proportion of uninfected controls (
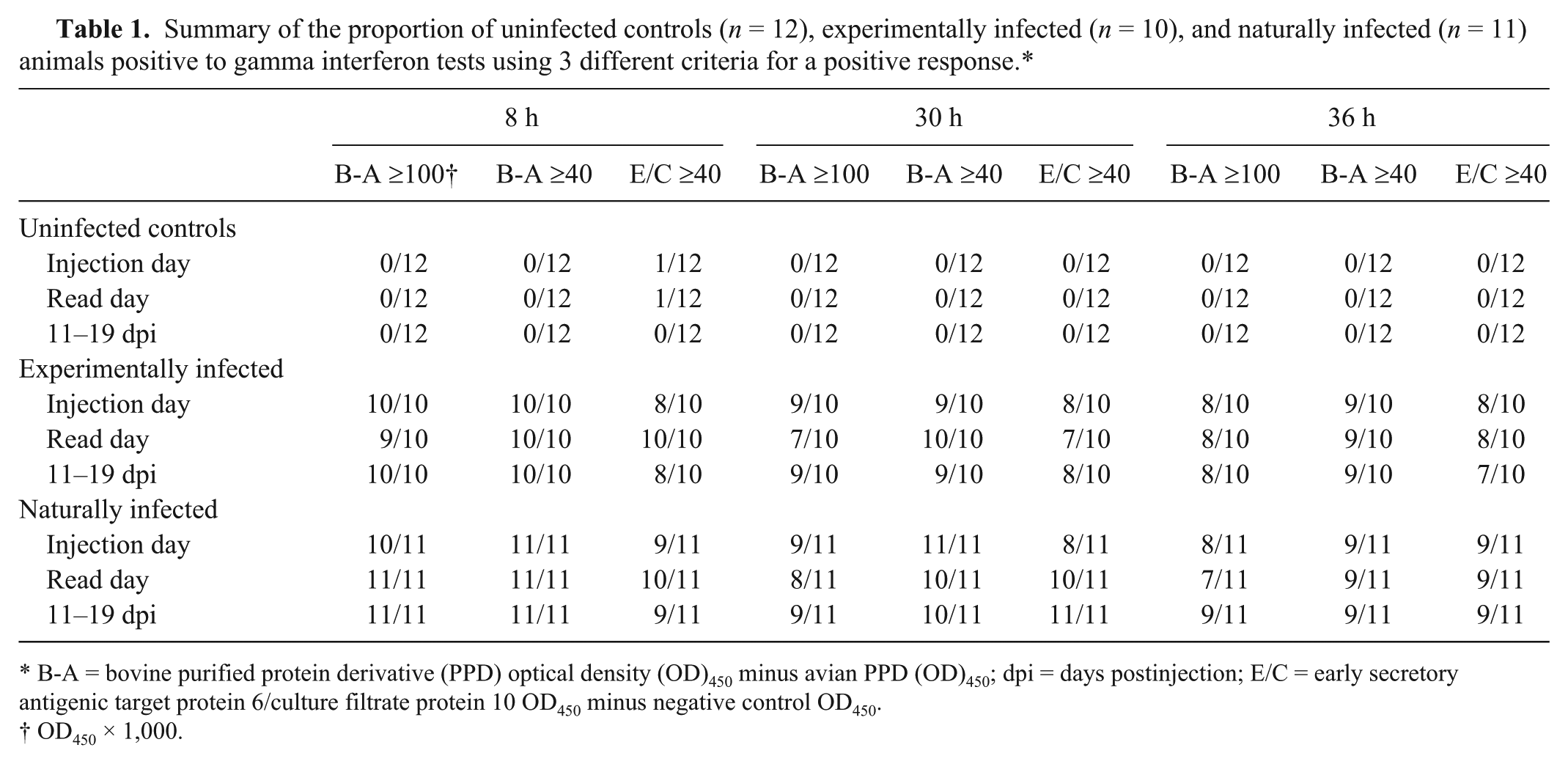
B-A = bovine purified protein derivative (PPD) optical density (OD)450 minus avian PPD (OD)450; dpi = days postinjection; E/C = early secretory antigenic target protein 6/culture filtrate protein 10 OD450 minus negative control OD450.
OD450 × 1,000.
The antigen responses were evaluated using 3 different cutoffs for designating a positive test: bovine PPD minus avian PPD ≥0.100 OD450, bovine PPD minus avian PPD ≥0.0.04 OD450, and ESAT6/CFP10 minus negative control ≥0.040 OD450 (Table 1). For the uninfected group, apart from 2 exceptions, all the tests were negative to the 3 different criteria for a positive response. The exceptions were positive responses in the same uninfected animal to ESAT6/CFP10 for the samples taken at the injection and read date of the skin test and processed at 8 h. Similar or slightly higher proportions of positive responses (differences of 0–3 positive responses) were observed for both naturally and experimentally infected groups of animals at the 8 h processing time compared with their respective 30- and 36-h processed samples. Differences between 30 and 36 h processing times were very small (0–2 positive responses) for the infected animals and a trend for slightly higher responses for the 30 h processed time. Larger group sizes would be required for a statistical evaluation of the differences between positive responses within groups.
One of the objectives of our study was to examine whether the currently used 30 h limit for processing blood samples used in New Zealand could be extended to 36 h, which would accommodate delays in the overnight delivery of samples to a laboratory. The decreases in the level of antigenic responses with increasing times to process blood samples recorded in our study were similar to those reported previously.
13
Significant decreases in responses were recorded with samples processed at 30 h versus those examined 8 h postcollection for both the PPD and ESAT6/CFP10 IFN-γ tests. Further significant decreases in the level of responses were observed between samples processed at 36 h versus 30 h postcollection at the injection and read days for the experimentally infected animals. Although these differences between 30 and 36 h were only observed in experimentally infected animals, a delay in processing beyond 30 h postcollection could have an effect on the ability to identify animals infected with
No major effects on IFN-γ responses, as determined by the magnitude of the responses, were attributable to caudal fold skin testing. This is in contrast to a study 5 in which IFN-γ responses to PPDs were elevated at the read day (3 dpi) of caudal fold skin testing. Similar, short-term boosting of IFN-γ responses by caudal fold skin testing were observed 10 in experimentally infected animals. However, boosting of IFN-γ responses has not been observed with comparative cervical skin testing, which uses both bovine and avian PPDs.4,7 Reasons for the differences observed in the effects of skin testing on IFN-γ results are unclear. Nevertheless, these differences highlight the complexities of the tuberculin response and the relationship to IFN-γ responses. The results of our study and previous investigations indicate that blood samples taken on the day that the caudal fold test is read can be examined without affecting the performance of the IFN-γ test. Such a change would eliminate the need for an extra farm visit required under the currently used protocols for IFN-γ testing in the New Zealand bovine tuberculosis control scheme, where blood sampling was restricted to 8–30 d post–skin testing. Based on the findings from our study, the New Zealand standards have been modified to allow blood collection for the IFN-γ test regardless of the timing of the skin test, but blood samples will not be processed if there is delay of >30 h for processing of samples (https://goo.gl/lScKNX).
Footnotes
Acknowledgements
We thank Gary Yates, Maree Joyce, and Kirstie Bland for the culture of postmortem samples; Peter Oden (AsureQuality) for carrying out the skin testing; Natalie Parlane for her help with the figure; and Dongwen Luo for his statistical advice and analyses.
Authors’ contributions
GW de Lisle contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. RS Green and BM Buddle contributed to design; contributed to acquisition, analysis, and interpretation of data; and critically revised the manuscript. BM Buddle gave final approval.
a.
Bovigam, Thermo Fisher Scientific, Melbourne, Australia.
b.
Bovine and avian PPD, Prionics, Schlieren-Zurich, Switzerland.
c.
ESAT6/CFP10, Serum Statens Institut, Copenhagen, Denmark.
d.
Observe bovine PPD (30,000 IU/mL), AsureQuality, Auckland, New Zealand.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this project was provided by TBFree New Zealand.
