Abstract
Adapting the gamma interferon (IFNγ) assay for tuberculosis screening at points-of-concentration of cattle would improve global efforts to eradicate bovine tuberculosis. Two separate studies were conducted to evaluate if transportation of cattle, the time of blood collection, and total lymphocyte count affects the retention of a positive IFNγ assay result during slaughter of cattle experimentally sensitized with inactivated
The bTB gamma interferon (IFNγ) assay is a diagnostic assay that measures IFNγ released by lymphocytes in response to antigen stimulation. 12 In the United States, the IFNγ assay is an official supplemental diagnostic test for bTB (http://www.aphis.usda.gov/animal_health/animal_diseases/tuberculosis/downloads/tb-umr.pdf) and has been used on live cattle that tested positive to the caudal fold tuberculin test during herd surveillance. 11 In an effort to evaluate the adaptability of the IFNγ assay for a POC screening of bTB, a previous study demonstrated that cattle that tested positive for bTB on the IFNγ assay before slaughter remained positive at exsanguination. 10 However, blood collected during exsanguination has been reported to have a decreased IFNγ production upon antigen stimulation. 13 The factors associated with, and the rate of the measurable decline in IFNγ production from whole blood after exsanguination, are not known.
The objectives of the current study were to determine the rate of measurable decline in IFNγ production following transportation of cattle and if the total lymphocyte count changes at each time of blood collection after commencement of exsanguination of cattle at slaughter. An understanding of these factors would support the development of an optimal use of the IFNγ assay for POC screening of cattle for bTB at slaughter.
The Michigan State University (MSU; East Lansing, Michigan) Institutional Animal Care and Use Committee approved the present study (approval no. 08/09-125-00). Two separate studies were conducted. Five clinically healthy, nonpregnant adult Holstein cows (2–3 years of age) and 5 clinically healthy Angus steers (1.5 years of age) were used in study 1 and study 2, respectively. The 5 Holstein cows were part of a larger previous study looking at the IFNγ response to tuberculosis in blood collected at commencement of exsanguination at slaughter from cattle sensitized with
In study 1, after a 1-week acclimation period, 4 of the Holstein cows were randomly selected and then immunologically challenged with 0.1 ml of inactivated
Blood samples for the IFNγ assay were collected from each animal at the housing facility (W), after a 30-min transport of the cows to the slaughter facility (X), at approximately 15 hr post-transport and immediately before stunning (Y), at commencement of exsanguination (Z0), and at 5 min postexsanguination commencement (Z5). All blood samples (6–8 ml) were collected from live cattle with a 20-gauge needle into a 10-ml evacuated tube containing sodium heparin. Samples of blood were collected from the jugular vein at the housing facility and from the coccygeal vein or artery of live cattle at the slaughter facility. Cows were stunned via captive bolt and exsanguinated in accordance with the standard operating protocols of the slaughter facility. A blood sample was collected from each cow immediately after the jugular and carotid arteries were severed and blood was flowing freely. The time from stunning to the first blood collection at commencement of exsanguination ranged from 60 to 90 sec. Another sample of blood was collected 5 min after exsanguination commenced. Each blood sample obtained at exsanguination was collected into a 10-ml syringe with the plunger removed; the syringe plunger was replaced and the blood was immediately transferred with an 18-guage needle into two 10-ml evacuated tubes containing sodium heparin. Blood samples were delivered in an iced cooler to the MSU-DCPAH for sample testing within 1 hr after collection.
Five Angus steers were used in study 2. The sensitization protocol and method of blood sample collection and delivery to the laboratory was the same as described for study 1 but the time of blood sample collection differed. After the steers were scheduled for slaughter, blood was collected at the following time points: W, Z0, at 1 min postcommencement of exsanguination (Z1), and at 2 min postcommencement of exsanguination (Z2). In addition to performing the IFNγ assay on the blood samples for bTB evaluation, a total lymphocyte count was conducted. The blood sample (3–5 ml) for the lymphocyte count was collected into a 7-ml evacuated tube containing ethylenediamine tetra-acetic acid and delivered within 1 hr of sample collection to the MSU-DCPAH for sample testing and analysis.
The IFNγ assay was performed according to the manufacturer’s instructions.
b
Briefly, 1.5-ml aliquots of each blood sample were dispensed to each of 4 wells of a 24-well tissue culture plate. The wells contained 100 µl of sterile phosphate buffered saline (negative antigen control), 100 µl
The total lymphocyte count was measured via a hematology system.
e
The lymphocyte count was derived from the automated differential values when routine acceptance criteria were met; otherwise 100-cell microscopic differential count was performed manually by board-certified clinical pathologists. Statistical analyses on the changes in the IFNγ productions and the lymphocyte count were performed using the general linear mixed model (GLMM); time of sampling was the fixed effect, and measures were repeated on the 4 sensitized cows. Statistical significance level was set at
In study 1, the categorical interpretations of the bTB IFNγ assay at each time point were as follows: (W) all 4 sensitized cows were positive, (X) 3 cows (75%) were positive, (Y) all 4 sensitized cows were positive, (Z0) 2 cows (50%) were positive, and (Z5) no cow was positive. The nonsensitized cow was negative for bTB throughout the study. The results are presented in Figure 1. The residual plots suggested that the residuals of B-C and B-A do not appear normally distributed, hence log transformation was performed. Spatial power was used as the error structure in the GLMM analyses of both measurements. The time of blood collection was significantly associated with both log transformed B-C (
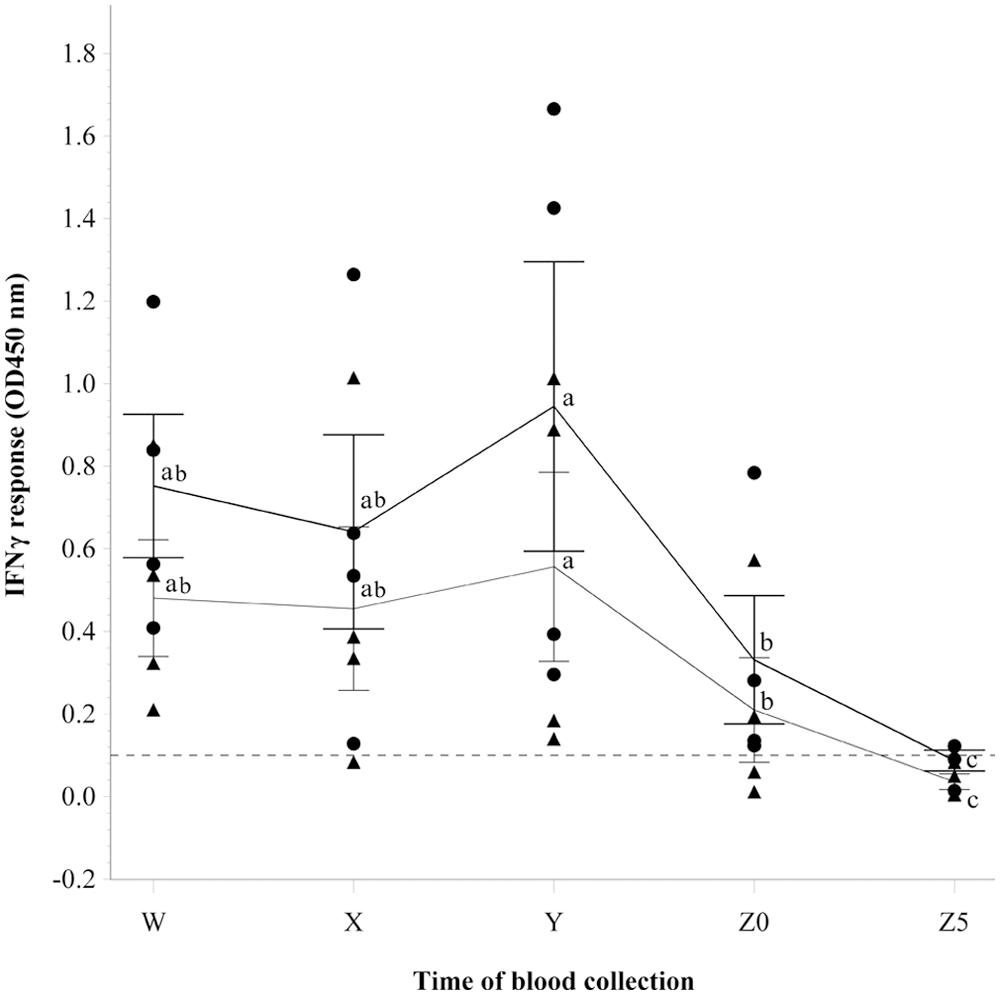
Differences in the gamma interferon (IFNγ) responses observed for blood samples collected before and during slaughter from 4 cows sensitized by subcutaneous injection with inactivated
In study 2, the categorical interpretation of bTB results using the IFNγ assay at each time point were as follows: (W) all 4 sensitized steers were positive. Subsequently 2 animals (50%) were positive at Z0, Z1, and Z2. The nonsensitized steer was negative throughout the study. The residual plot and the Shapiro–Wilk test suggested that the residuals of B-A (
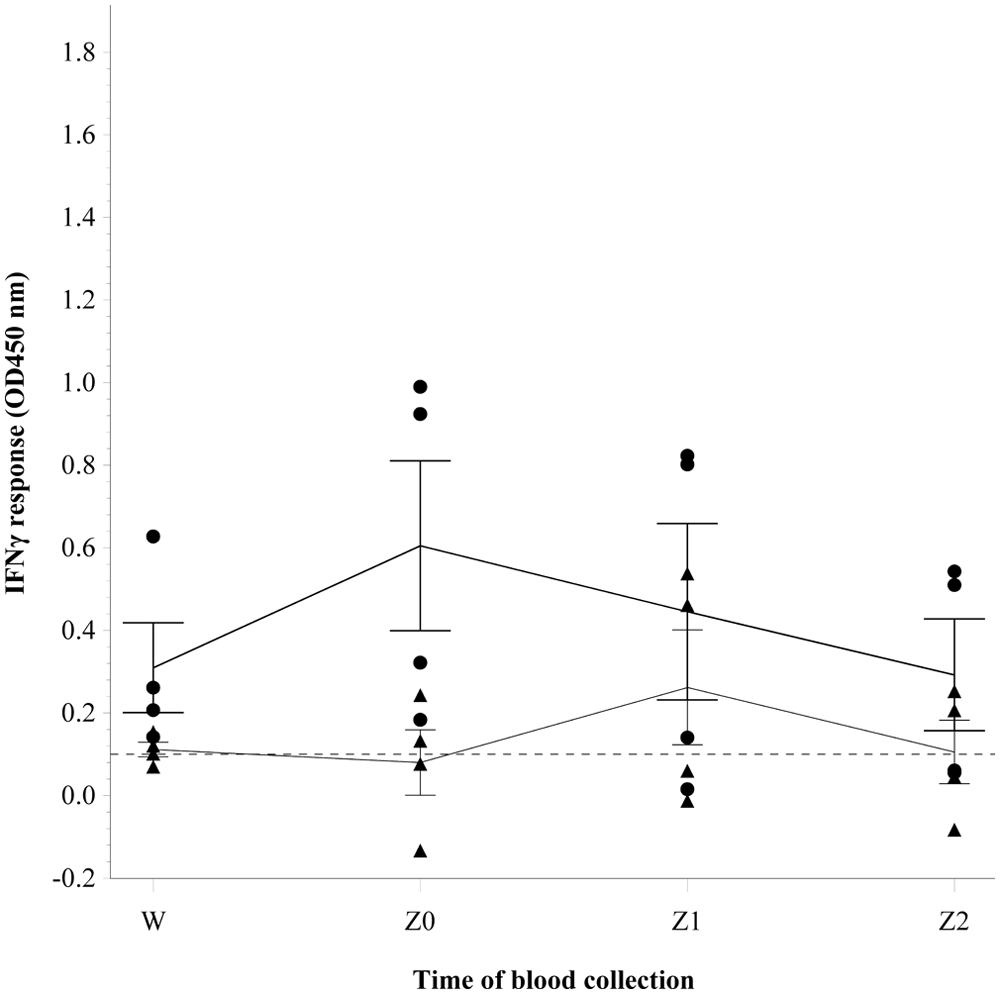
Differences in the gamma interferon (IFNγ) responses observed for blood samples collected before and during slaughter from 4 steers sensitized by subcutaneous injection with inactivated
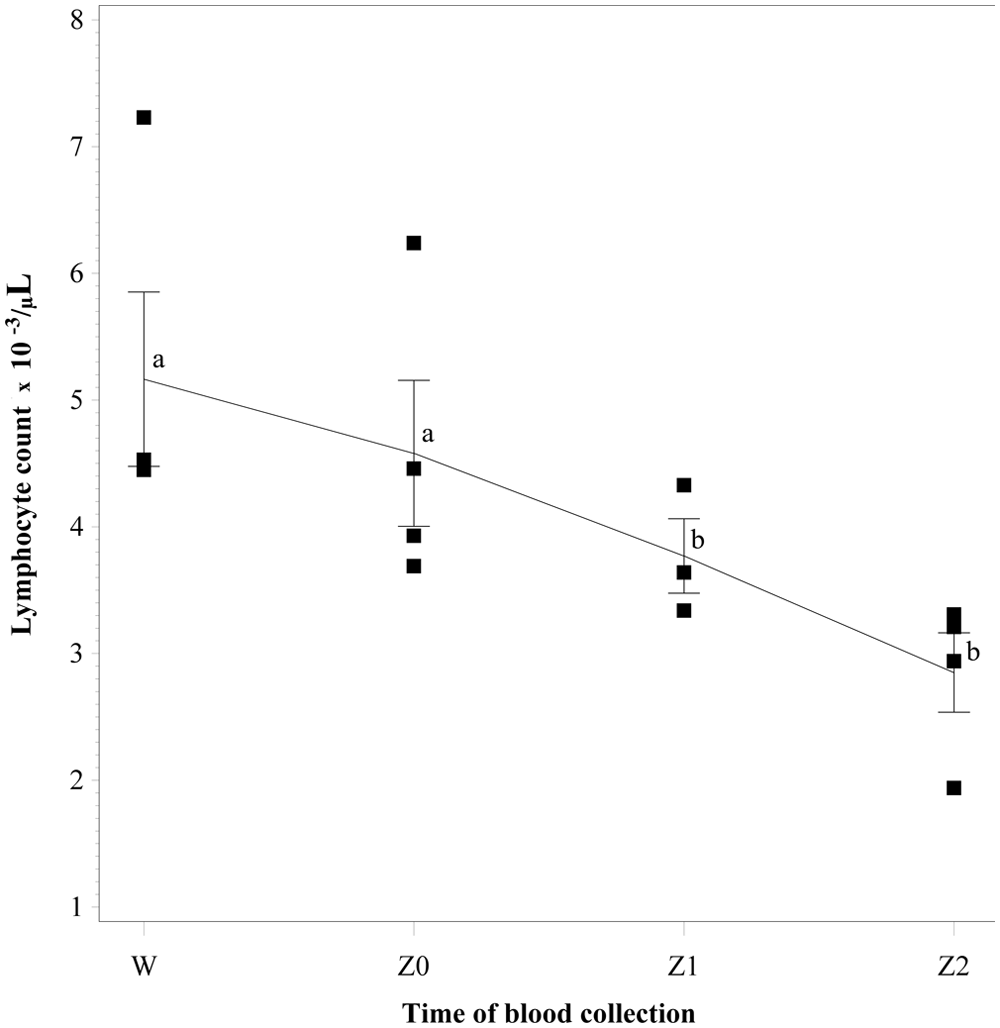
The total lymphocyte count for blood samples collected before and during slaughter from 4 steers sensitized by subcutaneous injection with inactivated
The current study demonstrates that time of blood collection after commencement of exsanguination is critical to obtaining reliable bTB results using the IFNγ assay. Based on the results of the present study, IFNγ production declines with time following exsanguination. At 5 min postcommencement of exsanguination, all cattle, which previously had positive results for bTB, were negative by IFNγ assay using the same established interpretation criteria. The decline in measurable IFNγ production in response to antigen stimulation following exsanguination is in agreement with results of previous similar studies.10,13 The findings of the current study highlight the need for timely collection of blood postexsanguination if the IFNγ assay is to be implemented as part of a bTB surveillance program at slaughter.
Transport of cattle for 30 min resulted in a transient drop in IFNγ production. However, after an overnight rest, the corrected ELISA values for measurable IFNγ were similar with those obtained prior to transport. The decline in IFNγ production following transport could be due to a transport-associated transient suppression of lymphocyte count and functions. Although not measured after transport of animals in the current study, lymphocyte count and functions have been shown to be transiently suppressed after transport of cattle. 14 In addition, short-term transportation (approximately 45 min) of cattle has been reported to induce reduction in lymphocyte count. 9 Hence, the IFNγ assay with its present bTB interpretation criteria may not be ideal for bTB surveillance in slaughtered animals that are not well rested after transport to the slaughter facility.
The statistical power of the present study is less than optimal because of the limited number of subjects used. At certain time points of blood collection, although declines in IFNγ response were not found to be significant, changes in the categorical classification of the assay were observed. Therefore, strong emphasis on statistical significance may undermine the biological importance of the declines in IFNγ response observed in the current study.
The decline in total lymphocyte count could be responsible for the corresponding decline in IFNγ responses observed in the current study. It was observed that between collection of blood before stunning and at exsanguination, as well as with each passing minute postexsanguination, there was a parallel decline in total lymphocyte count. Because IFNγ is a cytokine produced by T lymphocytes, 1 it is likely that a decrease in absolute lymphocyte count would also cause a decline in specific stimulated IFNγ production. The factors responsible for the declining lymphocyte count were not studied. One possible explanation is the accumulation and adhesion of leukocytes to the epithelial cells of blood vessel walls at the site of injury in the early phases of inflammation, also called “margination.” 1 Another possible explanation for declining lymphocyte count at exsanguination could be stress. Stress associated with a 45-min transportation of cattle has been found to induce reduction in lymphocyte counts. 9 In rats, an hour of acute stress involving vigorous shaking of whole body has been demonstrated to result in a significant decrease in absolute number of leukocytes.5,6 In the rat studies, the decrease in blood leukocyte numbers represents a redistribution of leukocytes from the blood to organs such as the bone marrow and skin.3,4 Assuming that stunning causes similar lymphocyte redistribution as seen in short-term transported cattle or in acutely stressed rats, any decrease in absolute number of lymphocytes would decrease IFNγ productions as well. It is also possible that stunning could have affected lymphocyte function as was observed after 72 hr of transportation of cattle. 14
Although the goal of the current study was not to measure sensitivity or specificity of the assay in exsanguinated cattle, a change in the cutoff value used to categorize cattle as bTB positive or negative would have increased the number of sensitized animals reported as positive for bTB at the measured points. For example, if the criteria for interpretation of an animal as being positive for bTB was that the difference between the bovine PPD and no-antigen control responses was ≥ 0.1 and the difference between the bovine PPD and avian PPD responses was ≥ 0.05, the sensitized cow in study 1 that tested negative for bTB at time point X would have been classified as positive for bTB at both X and Z0, and 1 sensitized cow would have given a positive result for bTB at Z5. With this criterion, a total of 2 steers in study 2 would have given positive results for bTB at Z1 and Z0. In all of these, the nonsensitized cattle would never be classified as positive for bTB.
Useful data were obtained in the current study, although the study was limited in terms of number of cattle used and time points chosen for collection of samples of blood for testing. First, it was shown that under experimental conditions, blood obtained from cattle at commencement of exsanguination for IFNγ assay evaluation was more likely to remain positive for bTB than blood collected at successive time points; hence the time of blood collection is crucial to obtaining a useful IFNγ assay result for bTB at slaughter. Second, the number of total lymphocytes in flowing blood drops quickly after the exsanguination process commences, which likely contributes to the drop in IFNγ production observed with blood obtained at various time points during exsanguination. Finally, a relatively short period of transportation was sufficient to cause a reduction in IFNγ production when blood obtained after transport was compared with blood obtained immediately before transport. This last finding is important if the IFNγ assay is to be used to test cattle for bTB at POC. Based on these findings, specific protocols could be developed to improve the reliability of the bTB IFNγ assay for adoption as a surveillance tool at slaughter. For example, to compensate for the decline in lymphocyte count, an adjusted ELISA OD value cutoff for categorizing animals as either bTB positive or negative could be developed for blood collected at exsanguination. To understand the most appropriate cutoff criteria, testing of more cattle at slaughter would be useful, and the use of cattle under natural conditions of
Footnotes
Acknowledgements
The authors are grateful to the staff of the Michigan State University Beef Cattle Research and Teaching Center for their assistance in housing and caring for the animals; the staff of the Michigan State University Department of Animal Science Meat Laboratory for assistance in slaughtering the animals; Dr. Joseph Gardiner of the Department of Epidemiology and Biostatistics, Michigan State University for his assistance in formatting the figures presented in this article; and William Sears of the Department of Population Medicine, University of Guelph, Canada, for his assistance in the data analyses.
a.
Sensitinogen, U.S. Department of Agriculture Animal and Plant Health Inspection Service, National Veterinary Services Laboratories, Ames, IA.
b.
Bovigam, Prionics AG, Zurich, Switzerland.
c.
Prionics USA Inc., La Vista, NE.
d.
Sigma-Aldrich, St. Louis, MO.
e.
Advia, Siemens Healthcare Diagnostics Inc., Tarrytown, NY.
f.
SAS/STAT 9.2, SAS Institute Inc., Cary, NC.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received funds from the U.S. Department of Agriculture National Institute of Food and Agriculture Special TB Grant (agreement no. 2009-34427-19959) and the Edward and Roberta Sterner Fund for Bovine Health for the research and publication of this article.
