Abstract
Mycobacterium fortuitum and at least 1 unidentified species of soil mycobacteria were isolated from lymph nodes from 4 of 5 African buffalo (Syncerus caffer) that had been culled because of positive test results using the Bovigam assay. The buffalo were part of a group of 16 free-ranging buffalo captured in the far north of the Kruger National Park (South Africa) assumed to be free of bovine tuberculosis. No Mycobacterium bovis was isolated. To investigate the possible cause of the apparent false-positive diagnosis, the Mycobacterium isolates were inoculated into 4 experimental cattle and their immune responses monitored over a 13-week period, using the gamma interferon assay. The immune reactivity was predominantly directed toward avian tuberculin purified protein derivative (PPD) and lasted for approximately 8 weeks. During that period 3 of 4 cattle yielded positive test results on 1 or 2 occasions. The immune responsiveness was boosted when the inoculations were repeated after 15 weeks, which led to 2 subsequent positive reactions in the experimental animal that did not react previously. Including an additional stimulatory antigen, sensitin prepared from M. fortuitum in the gamma interferon assay, showed that it was able to elicit a detectable gamma interferon response in all 4 experimentally inoculated cattle when applied in parallel with bovine and avian tuberculin PPD for the stimulation of blood samples. The implications of occasional cross-reactive responses in natural cases of infection with environmental mycobacteria in the diagnosis of bovine tuberculosis in African buffalo and cattle in South Africa are discussed.
Mycobacterium fortuitum is an environmental, non-tuberculous mycobacterium that has been repeatedly isolated from cattle. Previously, infections with other atypical mycobacteria were shown to result in positive skin tests in cattle. 1,2,5,13 Since it must be assumed that buffalo are abundantly confronted with environmental mycobacteria, such exposure may bias immunoreactivity towards infection with Mycobacterium bovis. It may also influence diagnostic assays for M. bovis, including both the skin test and the gamma interferon (IFNg) assay. The associated risk of encountering false-positive diagnoses in buffalo is unacceptably high when measured against the economical and ethical consequences. It is therefore important to identify a test strategy able to distinguish between specific, truly infected, and nonspecific reactors. The commercial Bovigam kit is an in vitro IFNg assay for the diagnosis of M. bovis infection in bovines that has proven extremely useful as an ancillary assay to the skin test in different countries. 4,10,11 Its use in free-ranging buffalo populations is preferred over the intradermal tuberculin test because it requires only a single manipulation of the animals. 9 In infected buffalo herds in the Kruger National Park (KNP; South Africa), the IFNg assay proved suitable to correctly identify infected individuals based on their immune response towards bovine purified protein derivative (PPD). 7 The aim of the present study was to describe various cases of natural M. fortuitum infection in buffalo and to perform a pilot experimental infection with M. bovis diagnostic follow-up.
Five buffalo from a group of 16 animals captured in the far northern tuberculosis-free region of KNP reacted strongly to bovine tuberculin PPD a in the gamma interferon assay. The bovine absorbance values were more than 0.1 greater than the absorbance values of avian PPD. The remaining 11 buffalo tested negative. Results for the positive and negative control samples included in the kit b as well as the nil controls of the test samples were within acceptable ranges, and according to the manufacturer's recommendations for interpretation of test results, these animals were classified as test positive. The buffalo were euthanized, and pooled lymph node samples from the head, thorax, and mesenterium were cultured according to standard procedures. 3 No visible lesions were observed at necropsy. Bacterial culture yielded fast-growing Mycobacterium spp. from 4 of 5 reactor buffalo and a presumptive diagnosis of M. fortuitum complex was made for 2 isolates based on biochemical characteristics (growth at 25°C production of arylsulphatase and nitrate reductase). The diagnosis was confirmed in a line probe assay 8 performed at the Mycobacteriology Unit of the Prince Leopold Institute of Tropical Medicine (Antwerp, Belgium). The 2 unidentified Mycobacterium isolates comprised unclassified soil mycobacteria. No isolation of M. bovis was made from any samples collected from the test-positive buffalo.
The potential role of environmental mycobacteria, including M. fortuitum as the cause of the false-positive reactions in buffalo, was investigated by way of experimental infection. Four IFNg-negative and skin test-negative cattle (10-month-old oxen of mixed breed) were inoculated intravenously with 2 ml of phosphate buffered saline (PBS) containing a mixture of the Mycobacterium isolates grown from the lymph nodes of the test-positive buffalo at a final concentration of 10 7 bacteria/ml. Two skin test-negative and IFNg-negative control animals were inoculated with PBS and kept on the farm, separate from the experimental group. Blood in heparin was collected in intervals of 1–2 weeks for a period of 13 weeks. Inoculation was repeated for the experimental group after 15 weeks, while monitoring of the control animals was stopped for operational reasons. Blood samples were processed and tested according to the standard IFNg protocol provided by the manufacturer. b Starting in week 5 postinoculation, an additional aliquot of 1.5 ml of whole blood was stimulated with 50 μg of Fortuitum, b a sensitin derived from M. fortuitum.
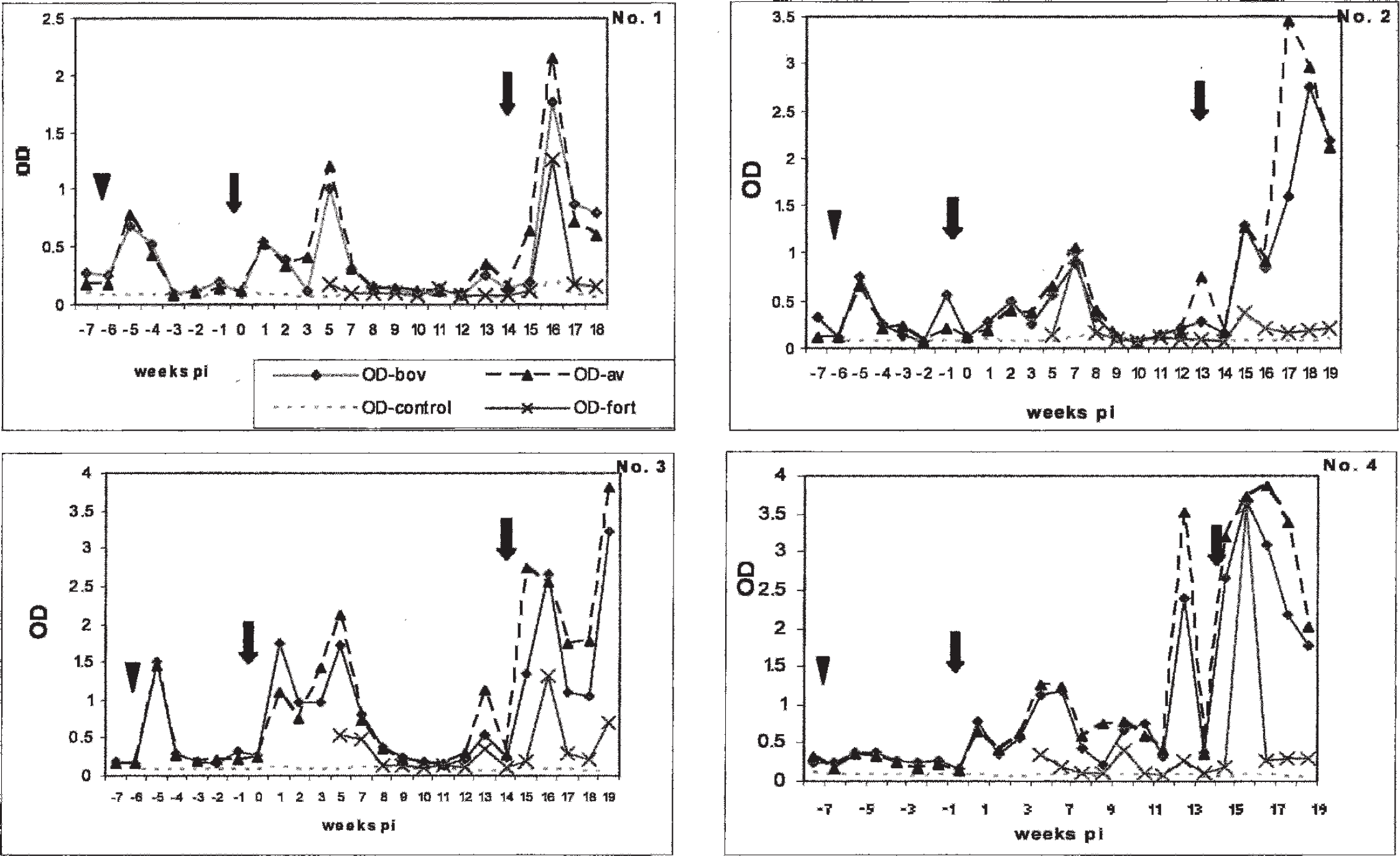
Kinetics of the early gamma interferon response of 4 cattle inoculated with a mixture of environmental mycobacteria. Panels 1–4 illustrate the gamma interferon responses of 4 experimental cattle to inoculation with environmental mycobacteria. OD: optical density; p.i.: postinfection; arrowhead: skin test; arrow: inoculation with environmental mycobacteria.
The kinetics of the IFNg responses of the experimental cattle are illustrated in Figure 1 and show that inoculation with environmental mycobacteria resulted in an at least 2-fold increase of the bovine and avian absorbance values within 1 week in animals 1, 3, and 4, and within 2 weeks in animal 2. For most of the experiment, the IFNg response was primarily directed against avian tuberculin. However, all experimental animals showed episodes when bovine and avian absorbance values were equally strong and 1–2 test occasions when the IFNg response to bovine tuberculin exceeded that of avian tuberculin by at least 0.1, resulting in a positive test outcome (Fig. 1). Animal number 1 tested positive on the last 2 test occasions in weeks 17 and 18, while animals no. 2 and 3 did so in week 1 and week 2. Animal no. 4 showed 1 peak of bovine IFNg reactivity in week 1 and a second peak in week 11. Eight weeks after inoculation, the IFNg responses of the experimental animals had normalized to preinoculation levels in 3 of 4 experimental animals. Following the second inoculation with the Mycobacterium cocktail, the IFNg reactivity increased markedly (3- to 10-fold) in all experimental oxen and remained high for the remaining period of the experiment. In the IFNg assay, the control cattle kept separately did not show any significant immune response to stimulation with any of the antigens, except for 2 occasions when one of the animals mounted a sporadic, short-lived response to avian tuberculin PPD, not exceeding an optical density (OD) value of 0.53 (data not shown).
In summary, following natural or experimental exposure to environmental mycobacteria including at least M. fortuitum, buffalo and cattle may become sensitized to stimulation with bovine and avian tuberculin PPD when tested in the IFNg assay. The resulting cross-reactive immune responses can lead to a reduction in IFNg test specificity and occasional misclassification of animals as test positive. Since both infected and uninfected buffalo populations are exposed to the same environmental mycobacteria, it is not surprising that similar patterns of IFNg reactivity have also been observed occasionally in a small number of buffalo in naturally infected populations. 7 No indication for the induction of IFNg reactivity to Fortuitum by M. bovis has, however, been found in subsequent studies in infected buffalo (A. L. Michel, unpublished data).
The findings of the current study have shown that stimulation of whole blood with Fortuitum has led to the production of detectable amounts of IFNg in sensitized cattle, indicating that Fortuitum may be a suitable antigen for the detection of exposure to related environmental mycobacteria when using this diagnostic tool. A previous study on sensitisation of cattle by nontuberculous mycobacteria in South Africa found that skin test reactivity to Fortuitum was the third most frequently encountered sensitization in “problem herds” (history of nonspecific reactors) after that of M. avium and M. kansasii. 12 While the most probable source of natural infection of buffalo and cattle with M. fortuitum would be soil and water, it has actually been shown that oral administration of M. fortuitum in cattle led to skin reactivity to mammalian and avian tuberculin as well as Fortuitum. 6 A previous study 12 of experimentally sensitized cattle with 5 different nontuberculous mycobacteria measured their skin reaction to intradermal injection of sensitins prepared from the same strains. It found that the homologous sensitin caused distinctly larger reactions than heterologous sensitins. 12 It is therefore relevant in this context to investigate the diagnostic value of including Fortuitum as an additional stimulatory antigen in the IFNg assay protocol to improve the specificity of the IFNg assay in buffalo and cattle under South African conditions. This is of significance because buffalo are of extremely high commercial value in South Africa with a resulting need for a higher specificity than generally accepted in cattle.
Acknowledgements. The author wants to thank Prof. F. Portaels for kindly identifying the Mycobacterium strains isolated from the buffalo, as well as Central Commonwealth Serum Laboratories, Australia, for providing the Fortuitum. The study would not have been possible without the excellent technical support from the staff at the Onderstepoort Veterinary Institute Tuberculosis Laboratory and the Animal Provision Unit led by Mr. C. van Vuuren.
Footnotes
a.
Institute for Animal Science and Health, Lelystad, The Netherlands.
b.
Commonwealth Serum Laboratories, Victoria, Australia.
