Abstract
In the current study, the results of an intradermal tuberculin test and a gamma interferon (IFN-γ) release assay were compared. IFN-γ release assay is based on the detection of IFN-γ production after in vitro stimulation with Mycobacterium avium subsp. avium–specific antigen for the discrimination of pigs naturally infected with M. avium subsp. hominissuis. Fifty-five clinically healthy pigs were used in the study. Three of these were proven by culture and real-time quantitative polymerase chain reaction methods to be infected with M. avium subsp. hominissuis (2 animals) and Mycobacterium xenopi (1 animal). No animals were positive by the tuberculin test. Both M. avium subsp. hominissuis–positive pigs were evaluated as positive by the IFN-γ release assay. Bacteriologically negative and M. xenopi–positive pigs were unresponsive in the IFN-γ release assay, indicating the specificity of the method. The results suggest that the IFN-γ release assay has a higher sensitivity than the tuberculin test and that the assay can be used for diagnosis of M. avium infections in live, naturally infected pigs.
In swine, mycobacterial infection usually occurs without clinical manifestation; however, the infection causes economic losses for farmers because meat from infected animals is considered inappropriate for human consumption.1,12 Moreover, mycobacterial infections can be a cause of abortions and reduced growth rates in pig herds in some cases. 15 The most frequent species isolated from porcine lymph nodes are members of the Mycobacterium avium complex.9,12,13 The majority of these isolates was identified as M. avium subsp. hominissuis (MAH; characterized by the presence of the insertion sequences IS1245 and lacking IS901) and approximately 10% of isolates were identified as M. avium subsp. avium (MAA; IS901+, IS1245+). 4
Diagnosis of the infection is usually possible only during postmortem examinations of pigs by the detection of granulomatous lesions. This approach is, however, nonspecific for mycobacteria because other bacteria species such as Rhodococcus equi have been isolated from granulomatous lesions in pigs.7,12 Thus, bacteriological cultivation or molecular biological approaches are required for a confirmation of mycobacterial infection.
To date, only the tuberculin test has been established as a screening method for the detection of mycobacteria-positive pigs prior to slaughter, but this method shows low sensitivity. 5 Therefore, there has been an effort to establish more reliable screening methods for the discrimination of infected animals. Recently, serological detection of mycobacteria-specific antibodies was described. 3 In terms of cell-mediated immunity, the gamma interferon (IFN-γ) release assay was shown to be a useful tool for detection in domestic pigs as well as wild boars (both belonging to Sus scrofa species) experimentally infected with MAA.6,14 These studies, however, used pigs experimentally infected with MAA and do not include MAH, which seems to be the most frequent M. avium subspecies isolated from naturally infected pigs. 4 The aim of the present study was to ascertain whether the IFN-γ release assay is useful also for individual diagnosis of naturally infected pigs.
Fifty-five clinically healthy pigs (Large White breed), both sexes, aged 4–5 months (approximately 90 kg of body weight) were used in the study. The pigs, originating from 3 different herds with overall good herd health, were utilized from October 2006 to March 2007 in feeding experiments at the Veterinary Research Institute in Brno, Czech Republic.
One month before slaughter, samples of heparinized blood were taken from the pigs. Each sample was divided into 2 parts: 1 ml of the blood was stimulated in vitro with 50 µl of MAA-specific antigen (preparation contained 28,000 IU of MAA proteins, strain D4 ER), a and 1 ml of blood served as the unstimulated control sample. Both samples were incubated for 20 hr at 37°C. Gamma interferon production was quantified using a commercially available enzyme-linked immunosorbent assay kit b in accordance with the manufacturer’s instructions. The detection limit of the kit was 2 pg/ml. Values below the detection limit are marked as 0 pg/ml in Figure 1. Absorbance was read on a multidetection microplate reader. c
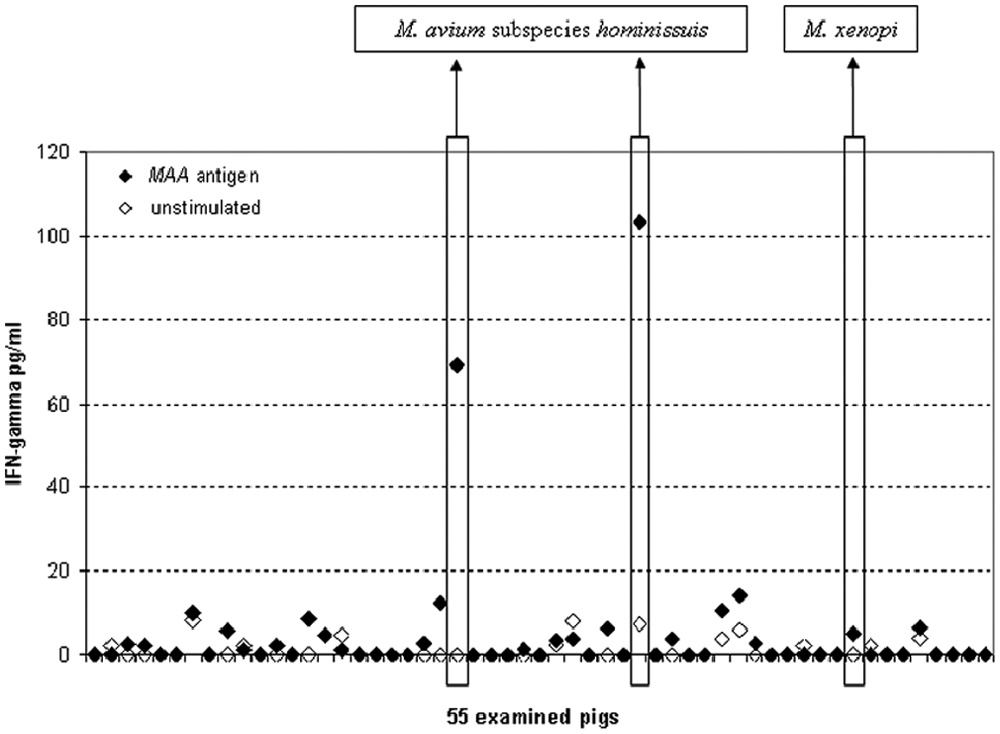
Individual results of gamma interferon (IFN-γ) release assay in pigs naturally infected with Mycobacterium avium subsp. hominissuis. MAA = M. avium subsp. avium.
Simultaneously, skin testing (i.e., an intradermal administration of 0.1 ml of the antigen designed for this type of test a ) was performed. Based on the manufacturer’s recommendations, local lesions were evaluated 48 hr after administration. In the slaughterhouse, detailed inspection was performed with a focus on macroscopic lesions. In parallel, samples of various organs, including mesenteric lymph nodes, tonsils, and spleen, were taken and examined by the culture method for the presence of mycobacteria. Isolates were identified by conventional polymerase chain reaction for the detection of species-specific insertion sequences IS900, IS901, and IS1245. 10
Among the 55 pigs used in the study, no animal was positive by the tuberculin test (i.e., no local reaction at the site of injection exceeded 20 mm in diameter 48 hr after antigen administration). No macroscopic lesion was detected at slaughter. However, using the culture method, 3 pigs were positive for mycobacteria: 2 animals were positive for MAH, and 1 animal was positive for M. xenopi. The bacteria were detected in samples from mesenteric lymph nodes.
The concentrations of IFN-γ detected in the 2 MAH-positive pigs were 103 and 69 pg/ml versus 8 and <2 pg/ml in the antigen-stimulated and unstimulated samples, respectively (Fig. 1). The level of IFN-γ from bacteriologically negative pigs ranged between 2 and 20 pg/ml and between 2 and 4 pg/ml in the antigen-stimulated and unstimulated samples, respectively. The pig that was positive for M. xenopi showed results from the IFN-γ release assay comparable with bacteriologically negative animals. When the results (2 pigs positive in bacteriological examination as well as in IFN-γ release assay, and 53 pigs negative in both tests) were examined using the Fischer exact test, d the difference was statistically significant (P < 0.01).
The IFN-γ release assay is based on specific stimulation of lymphocytes in the blood.2,11 The difference between the antigen-stimulated and unstimulated samples is crucial for evaluation of the assay. In uninfected animals, the level of IFN-γ should be comparable in both stimulated and unstimulated samples. Conversely, mycobacteria-infected animals have specific Th1 lymphocytes in the blood, and these lymphocytes are responsible for IFN-γ production after in vitro stimulation with mycobacterial antigen. As a result, there should be a significant difference between the antigen-stimulated and unstimulated samples from these animals. In the current study, differences between the antigen-stimulated and unstimulated samples were detected only in MAH-positive pigs, which indicated the specificity of the IFN-γ release assay. Additionally, the tuberculin test showed negative results in both MAH-positive pigs, which indicated the higher sensitivity of the IFN-γ release assay in comparison to the tuberculin test. In a recent study, 8 skin testing was used in wild boar (Sus scrofa) with known mycobacterial status and found to have a low sensitivity (43%–57%) in bacille Calmette-Guérin–immunized boars but a high sensitivity (75%–100%) in culture-positive animals. Differences between this work and the current study could be explained by the fact that MAH is a weaker pathogen when compared to MAA or Mycobacterium bovis with lower intensity of Th1 response. In that case, the amount of produced IFN-γ could be too low to induce a local reaction in skin testing.
In the case of the M. xenopi–infected pig, this animal was tuberculin test negative and the level of IFN-γ was similar to that detected in negative animals. One possible explanation for these results is that the MAA antigen was used in both methods and the immunodominant antigens in different mycobacterial species may not be cross-reactive. In conclusion, results of the current study suggest that there is a higher sensitivity of the IFN-γ release assay in comparison to tuberculin test and that the assay can be used for diagnosis of M. avium infections in live, naturally infected pigs.
Footnotes
Acknowledgements
The authors would like to thank Mrs. Eva Kotlarova for her excellent technical help.
a.
Avitubal, Bioveta Inc., Ivanovice na Hané, Czech Republic.
b.
BioSource International Inc., Camarillo, CA.
c.
Synergy™ 2, BioTek Instruments Inc., Winooski, VT.
d.
Prism, GraphPad Software Inc., La Jolla, CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This work was supported by the Ministry of Agriculture of the Czech Republic (MZe 0002716202, 1B53009) and Ministry of Education, Youth and Sports of the Czech Republic (CZ.1.05/2.1.00/01.0006, ED 0006/01/01 AdmireVet).
