Abstract
A group of Holstein first-calved heifers developed small pustules and ulcers on the vulva and in the vagina during the first 1–4 wk postpartum. The lesions varied from small red pinpoint foci to pustules and ulcers, 3–5 mm diameter. Some ulcers coalesced to form large ulcerated areas up to 15 mm diameter. In some animals, these ulcers progressed to become deep ulceration of the vaginal and vulvar mucosa with >50% of the mucosa involved. Vaginal biopsies from 4 heifers and vaginal individual swabs from 8 heifers for a combined sampling of 9 heifers were taken for clinical assessment. Six of the 9 heifers had parapoxvirus based on histopathology and/or PCR. Histologic examination of the biopsies of the pustules identified ballooning degeneration of the epithelium with degenerate epithelium containing eosinophilic intracytoplasmic inclusions consistent with a parapoxvirus in 3 of 4 biopsies. Testing for bovine herpesvirus 1, 2, and 4, bovine viral diarrhea virus, bovine papular stomatitis virus, and orf virus remained negative.
Multiple viral and bacterial causes of vulvovaginitis have been identified in cattle. Although most cases of vulvovaginitis are minor problems that can resolve quickly, some cases progress to serious mucosal damage. Infectious bovine pustular vulvovaginitis (IBPV) is a problem observed in heifers and is often associated with parturition. The disease is reported to spread by coitus. However, the virus is believed to also be transmitted by other means such as close contact with infected animals that shed the virus by aerosol or contact. It has also been postulated that the use of virally contaminated instruments or dairy personnel working with the infected animals may also spread the virus to naive animals. IBPV often involves multiple animals and can spread rapidly through a group of naive animals. This condition may also be associated with concurrent upper respiratory disease. Bovine herpesvirus 1 (BoHV-1) is the purported etiology of IBPV in cattle, and has been detected in infected animals reported in the literature.8,11,12 Bovine viral diarrhea virus (BVDV) has been reported to cause similar vulvar and vaginal lesions in a 14-mo-old heifer with a history of unresponsive bloody diarrhea. 6 BoHV-4 has also been isolated from cases of vulvovaginitis.2,3 BoHV-5 has been isolated from vaginal lesions in a group of cows artificially inseminated with virus-contaminated semen. 9 Mycoplasma bovigenitalium, Mycoplasma canadense, and Ureaplasma diversum have been detected in cases with granulopustular vulvovaginitis.7,10,12 Sometimes these bacteria also coinfect with BoHV-1. 7 The anaerobic bacteria Porphyromonas levii has been associated with severe necrotizing vulvovaginitis in post-parturient cattle.1,3,5,13 To the best of our knowledge, vulvovaginitis caused by a parapoxvirus has not been reported previously.
In January 2013, a dairy farm with 2,500 lactating cows in the Central Valley of California experienced an increased incidence (30–50%) of vaginal and vulvar lesions in first-calving heifers, 1–4 wk after parturition. Lesions began as small pinpoint reddened areas in the vaginal and inner vulvar mucosa that progressed over the next 2–4 d to small pustules (up to 5 mm diameter) that would then rupture, forming small ulcers. In many of the affected animals, the small ulcers would coalesce into larger and deeper ulcerated areas up to 15 mm diameter (Fig. 1). Animals with lesions that progressed to deep and extensive ulcers involving >50% of the vaginal mucosa would often be euthanized. The large deep vaginal ulcers in heifers that were not euthanized were slow healing and persisted for up to 4 wk after initial identification of the lesions. Heifers had been vaccinated with a modified live vaccine (BoHV-1, bovine parainfluenza virus 3, bovine respiratory syncytial virus; Inforce 3, Zoetis, Parsippany, NJ) and a leptospiral vaccine prior to breeding, and administered a clostridial vaccine at 6 mo of age and 1 mo prior to calving. The heifers on this dairy farm were artificially inseminated but were also exposed to bulls.
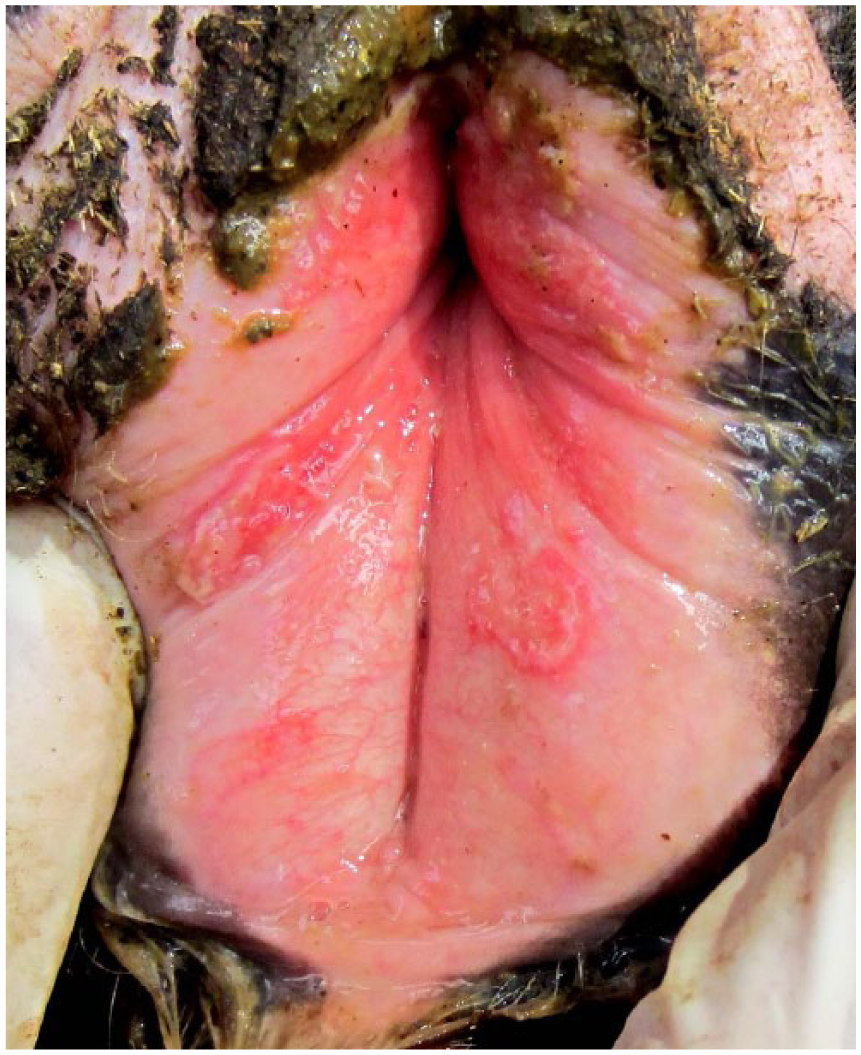
Large ulcers of the vaginal and vulvar mucosa of a Holstein cow.
The initial submission to California Animal Health and Food Safety Laboratory (CAHFS)-Tulare in January consisted of a vulvar biopsy in 10% neutral-buffered formalin from one heifer and a moist vulva swab on ice from a second heifer, which was placed in viral transport medium (VTM) consisting of buffered minimum essential media containing gentamicin and amphotericin B a few hours after collection. The biopsy had lesions compatible with a parapoxvirus (ballooning degeneration of the hyperplastic mucosal epithelium with affected cells demonstrating intracytoplasmic inclusion bodies). The swab was tested at CAHFS-Davis by PCR for BoHV-1, BoHV-2, and BVDV, initially. Based on histologic findings, PCR testing was added for pan-parapoxvirus, bovine papular stomatitis virus (BPSV), and orf virus (ORFV). Paraffin sections were also tested for BoHV-1 by immunohistochemistry (IHC). Only PCR for pan-parapoxvirus was positive. Two weeks later, in order to further rule out BoHV-1, the referring veterinarian (rDVM) submitted a pool of moist vulvar and/or vaginal swabs on ice from 4 affected heifers for BoHV-1 testing. Swabs were pooled in VTM on arrival, a few hours after collection; PCR tests for BoHV-1 and BVDV were negative. At the end of February, the herd continued to experience a 30% attack rate in heifers, and the rDVM submitted vulvar tissue biopsies on moist gauze and moist sterile swabs on ice from 3 affected heifers. Biopsies were placed in 10% formalin, and swabs were placed in VTM on arrival at CAHFS-Tulare within hours of collection. Two of the 3 biopsies had histologic lesions compatible with a parapoxvirus infection (Table 1). Swabs from the same 2 heifers were positive for pan-parapoxvirus by PCR and negative for the other 6 viruses (BoHV-1, -2, and -4, BVDV, BPSV, ORFV). The biopsy from the third heifer had granulation tissue in areas of ulcers with no active parapox-like epithelial lesions, and the swab from that heifer was negative by PCR for the 6 viruses including pan-parapoxvirus. This animal was not tested for BVDV. All 3 biopsies were again negative for BoHV-1 by IHC. In early April, in an attempt to isolate the virus, the rDVM submitted vaginal swabs in VTM from each of 4 affected heifers. Heifer 8 had a new lesion (blister that ruptured when swabbed). Swabs from all 4 heifers were tested by PCR for BoHV-2, BoHV-4, BPSV, ORFV, and pan-parapoxvirus (Table 1). Only pan-parapoxvirus testing was positive on 2 heifers, including the one with the new lesion. Virus isolation on all 4 heifers was performed on Madin–Darby bovine kidney cells and, following 3 passages 1 wk apart, no virus was isolated. Bacterial cultures were not requested by the rDVM and were not attempted on any of the samples submitted.
Vaginal biopsy findings and PCR results in an outbreak of parapoxviral vulvovaginitis in Holstein cows in California in 2013.
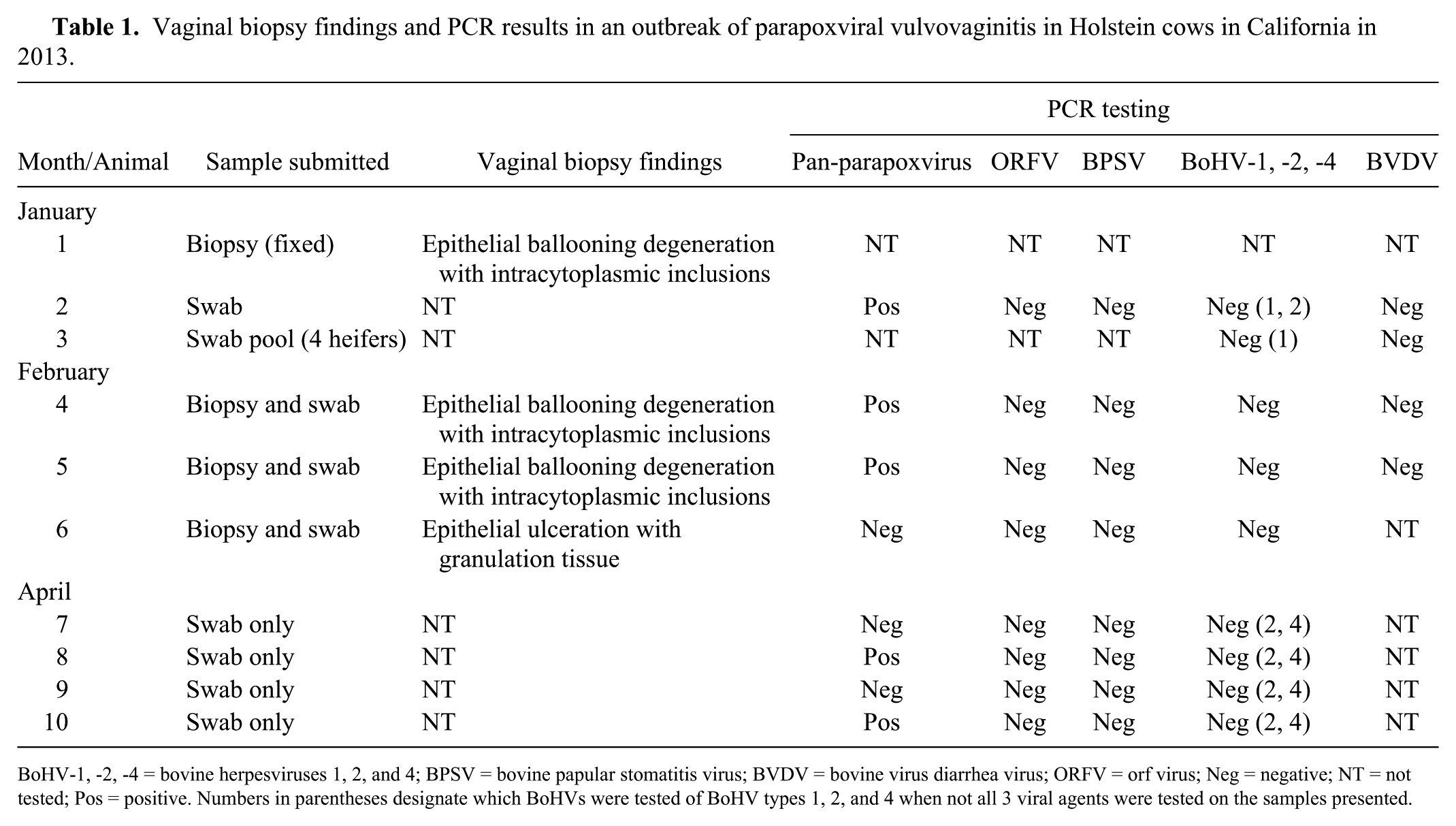
BoHV-1, -2, -4 = bovine herpesviruses 1, 2, and 4; BPSV = bovine papular stomatitis virus; BVDV = bovine virus diarrhea virus; ORFV = orf virus; Neg = negative; NT = not tested; Pos = positive. Numbers in parentheses designate which BoHVs were tested of BoHV types 1, 2, and 4 when not all 3 viral agents were tested on the samples presented.
All 4 biopsies were processed using routine histotechnology techniques, sections cut at 5 µm, and stained with hematoxylin and eosin. Paraffin blocks were further processed for BoHV-1 IHC.
Histologic examination of the biopsies identified lesions in various stages of development. In 3 of the 4 biopsies, the mucosal epithelium was thickened and undergoing mild-to-moderate mucosal epithelial hyperplasia and had multifocal clusters of superficial epithelial cells undergoing swelling and ballooning degeneration. Rare-to-small numbers of the swollen epithelial cells contained small (2–3 µm) eosinophilic intracytoplasmic inclusion bodies (Fig. 2). Minimal neutrophilic infiltrates into the affected epithelium were noted in these biopsies. In the stratum basale and adjacent submucosa, mild-to-moderate numbers of lymphocytes were present. The submucosa also had mild-to-moderate numbers of macrophages, plasma cells, and neutrophils. Many of the small blood vessels were dilated and had hypertrophied endothelium with intravascular neutrophils. Necrotic areas had loss of epithelium with abundant cellular debris and neutrophils. Epithelium at the edges of the ulcerated areas was hyperplastic, with mild proliferation and down-growth of the epithelium into the inflamed submucosa. The submucosa in the ulcerated areas contained variable numbers of neutrophils, lymphocytes, and macrophages. Submucosal blood vessels were prominent, proliferative, and congested. A few mixed bacteria were present in ulcers. The fourth biopsy had areas of epithelial necrosis, neutrophilic inflammation, active granulation tissue, and rare intravascular fibrin thrombi. Moderate numbers of neutrophils were present in the ulcers and adjacent submucosa. The areas of intact epithelium had moderate-to-marked hyperplasia and down-growth of epithelial pegs, with mild submucosal and perivascular edema, lymphoplasmacytic and histiocytic inflammation, and rare neutrophils.
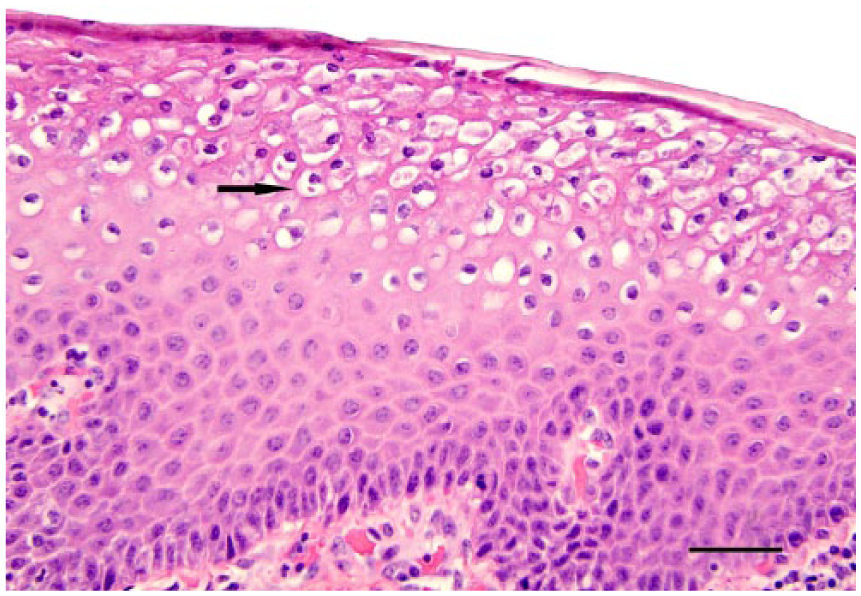
Hyperplasia of the vaginal mucosal epithelium with the epithelial cells undergoing swelling and ballooning degeneration. Some cells contain small (2–3 µm) eosinophilic intracytoplasmic inclusion bodies (arrow). H&E. Bar = 100 µm.
All PCR analysis was performed at CAHFS-Davis on vaginal and vulvar swabs placed in VTM. Fifty microliters of swab fluid were extracted (Max96 viral RNA isolation well kit, AM 1836-5, Life Technologies, Carlsbad, CA) to prepare samples for real-time PCR testing following the recommendations of the manufacturer (Path-ID multiplex one-step RT-PCR kit, Life Technologies; Applied Biosystems 7500 Fast real-time PCR system, Life Technologies). Real-time PCR was performed for the parapoxviruses using 3 different primer sets (kindly provided by Dr. K Kurth, WVDL, Madison, WI; primers and probe sets, unpublished data). A “pan-parapoxvirus” assay targeting a region in the B2L gene (viral envelope) was used as screening assay followed by the use of BPSV (Ankyrin) and ORFV assays (virion core) for typing. The thermocycler setting for all 3 assays was: stage 1 at 50°C for 10 min; stage 2 at 95°C for 10 min; and stage 3 at 95°C for 15 s; followed by stage 4 at 60°C for 1 min; stages 2–4 were repeated for 40 cycles
In 5 of the 8 animals tested (swabs), PCR analysis identified a parapoxvirus, but it was neither BPSV nor ORFV. No other viruses tested (BoHV-1, -2, and -4, BVDV, BPSV, and ORFV) were identified among the 8 individual animals. Three of the 4 biopsy samples had lesions compatible with parapoxvirus infection (Table 1). Two of the 3 with parapoxviral histologic lesions were positive for parapoxvirus by PCR on swabs; one was fixed tissue only. The one biopsy that did not have parapoxvirus-like lesions had granulation tissue consistent with an older lesion, and the swab from this animal was negative on pan-parapoxvirus PCR testing.
BoHV-1 was initially assumed to be the cause of pustular and ulcerative vulvovaginitis in postpartum heifers because it is the virus most commonly associated with this clinical presentation in California dairy farms (personal observation). In our case, vaccinating the heifers with a commercial intranasal attenuated BoHV-1 vaccine did not reduce the disease incidence, although this action has been effective in herds with BoHV-1 vulvovaginitis. Therefore, the rDVM in January 2013 initiated further testing. The additional investigation and subsequent biopsy of the lesions and PCR evaluation of vaginal swabs revealed the presence of a parapoxvirus and not a herpesvirus. PCR for BPSV was negative. Parapoxviruses, other than BPSV, that are known to cause lesions in cattle include pseudocowpox virus, a virus that is reported to affect the teats and mammary gland of postpartum cows. 4 No evidence of lesions was identified on udders or teats of the affected heifers in this herd. Why we were unable to identify the virus in all samples by PCR is unknown. It is possible that when swabs were taken, the virus was no longer present in the ulcers or tissues examined. Clinically, vaginal lesions were prevented and resolved by the use of a commercial sheep parapoxvirus vaccine (Contagious Ecthyma virus, Colorado Serum Company, Denver, CO) in all pre-breeding and currently pregnant heifers. Later, a parapoxvirus autogenous vaccine with BoHV-1 added had similar results in the herd.
How these animals became infected with this virus is unclear. It is possible that this virus was transferred from animal to animal by direct contact, or transmitted by fomites from dairy employees or their equipment when working with heifers after delivery. Insect vectors could potentially transmit the virus. However, at the time the cases occurred (late winter–early spring), flies were not a problem.
The frequency of vulvovaginitis caused by a parapoxvirus in dairy herds in California is undetermined. Our case represents the first outbreak by this unusual parapoxvirus reported in California. However, when small pustular epithelial lesions appear in a herd and rapid clearing of the problem does not occur after immunization with an attenuated intranasal BoHV-1 product, further investigation of the lesions with biopsy, PCR testing, and possibly bacterial cultures is warranted to determine if other agents are present.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
