Abstract
Mesothelial papillary hyperplasia (MPH) has been described as an incidental finding on the epicardial surface of clinically normal laboratory Beagle dogs. We describe MPH in 4 dogs diagnosed with acute cardiac tamponade (1 case) or chronic cardiac disease (3 cases). Cardiac MPH appeared as distinct, soft, irregular villous plaques on the epicardial surface of the auricles and occasionally the ventricles. Histologically, areas of MPH were composed of multiple papillary fronds arising from the epicardial surface and projecting into the pericardial space. Fronds were covered by cuboidal and occasionally vacuolated mesothelial cells and were supported by loose fibrovascular stroma with various degrees of edema and inflammation. Although these may represent incidental findings with no clinical significance, the gross appearance warrants differentiation from other conditions. Additional insight into the pathogenesis of MPH is needed to fully understand its significance in the face of concurrent cardiac disease.
Cardiac mesothelial papillary hyperplasia (MPH), also known as epicardial fronds, has been described as an incidental finding on the atrial epicardial surface of clinically normal Beagle dogs under experimental settings.1,5,9 No definitive sex predisposition has been confirmed in the few reported cases, although a slight predilection for males has been noted by one research group. 1 Gross anatomic changes are characterized by irregular villous plaques on the epicardial surface of the right or left cardiac atrium. Histologically, lesions consist of multiple papillary fronds that are covered by cuboidal mesothelial cells and supported by a loose collagenous stroma.1,5,9 The pathogenesis of MPH remains unknown, but these changes may be related to a chronic response to friction between the atrial epicardial surface and the pericardium. 5 Cardiac MPH is an uncommon pathologic finding in dogs, and its overall frequency is currently unknown, making it difficult to determine if these changes have any relationship with other underlying cardiac changes and thus pose an additional risk to affected individuals. We describe herein the occurrence of cardiac MPH in non-Beagles in a diagnostic setting. We also provide detailed gross images of the affected epicardium and underscore the need to include this lesion in the differential diagnosis of other common cardiac changes.
Case 1 was an 8-y-old male Pug with acute onset of dyspnea followed by cardiorespiratory arrest and death. The referring veterinarian performed the autopsy, and the formalin-fixed heart was sent to the Athens Veterinary Diagnostic Laboratory (AVDL; Athens, GA) for evaluation. Reported gross findings included oral and conjunctival mucosal pallor, hemothorax, and hemopericardium. Gross inspection of the formalin-fixed heart revealed extensive areas of epicardial hemorrhage in the coronary groove and pericardial sac. The right and left auricles were covered by distinct, soft, irregular villous plaques (Fig. 1). No changes were observed in the cardiac valves. The cause of death was determined to be hemorrhagic diathesis of unknown etiology, with cardiac arrest as a result of acute hemopericardium.
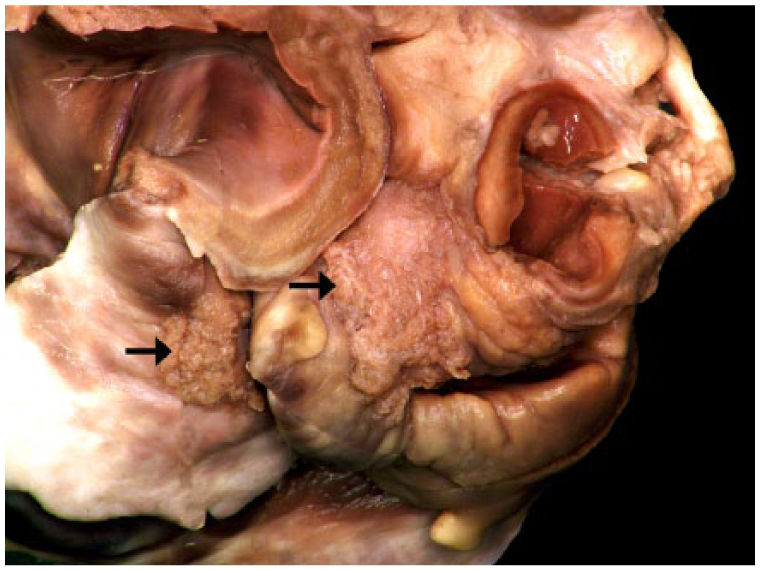
Extensive areas of mesothelial papillary hyperplasia (arrows) cover the epicardial surface of the right auricle of the formalin-fixed heart of dog 1.
Case 2 was an 18-y-old male neutered Cairn Terrier that was submitted for autopsy at the AVDL following acute onset of dyspnea, cyanosis, and death. Gross findings consisted of a cyanotic tongue and an enlarged right cardiac auricle, which was thickened by extensive, soft, irregular villous plaques that were partially covered by clotted blood and fibrin (Fig. 2). The mitral valve leaflets were thickened by smooth, tan, firm nodules (myxomatous degeneration). The caudal vena cava was distended, and the lungs were diffusely dark red to brown. The liver was moderately enlarged and friable, with a dull capsular surface. The cause of death was attributed to left-sided cardiac insufficiency, as evidenced by the microscopic findings of pulmonary alveolar histiocytosis with erythrophagocytosis. Hepatomegaly was the result of hepatic glycogenosis.
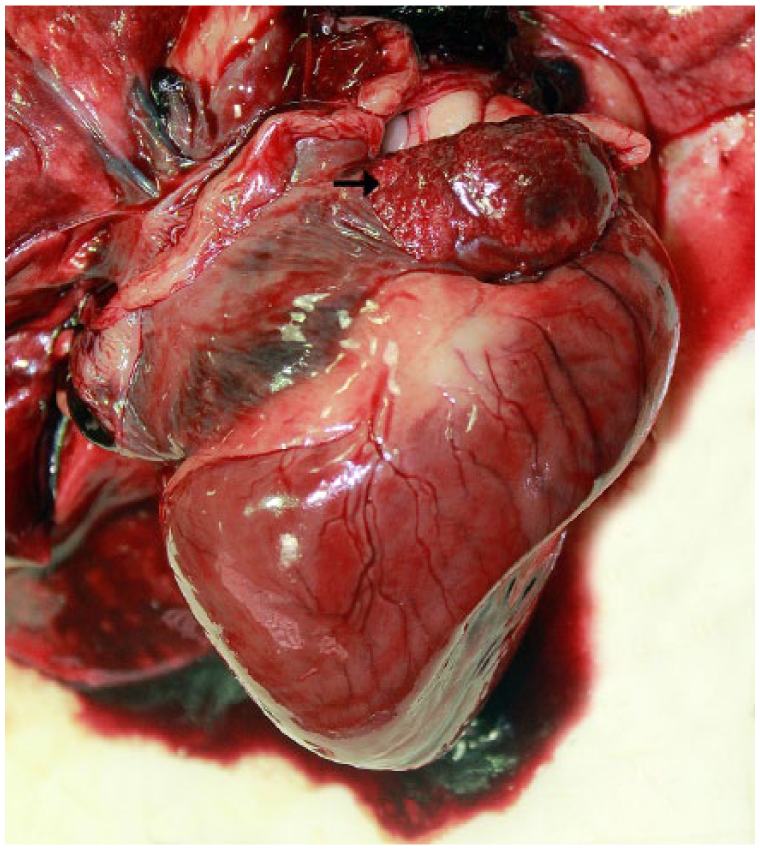
An area of mesothelial papillary hyperplasia thickens the epicardial surface of the right auricle (arrow) of dog 2. Blood and strands of fibrin cover the lesion.
Case 3 was a 14-y-old male mixed-breed dog that was submitted for autopsy at the AVDL after a 2-y history of progressive cardiac disease. Euthanasia was elected because of worsening clinical signs and poor prognosis. Gross findings were characterized by globoid cardiac enlargement and soft, irregular villous plaques that covered the left and right atrial epicardium, extending to the right ventricular epicardium. The mitral and tricuspid valve leaflets were thickened by smooth, tan, firm nodules (myxomatous degeneration), and there was biventricular cardiac dilation. The lungs were diffusely dark red. The cause of death was attributed to left-sided cardiac insufficiency as a result of bilateral, myxomatous valvular degeneration.
Case 4 was a 9-y-old neutered male Toy Poodle with a chronic history of a left-sided apical heart murmur with progression to clinical signs of congestive heart failure. Echocardiography revealed a left auricular aneurysm and significant mitral valve regurgitation. The dog underwent surgical resection of the auricular aneurysm, which was submitted for histologic evaluation at the Department of Pathology (University of Georgia, Athens, GA). Gross inspection of the submitted sample revealed a markedly thin, left auricular myocardium that was covered by soft, irregular villous plaques.
Histologically, areas of MPH were composed of multiple, 0.8–2 mm tall papillary fronds arising from the epicardial surface and projecting into the pericardial space (Figs. 3, 4). Fronds were covered by cuboidal and occasionally vacuolated mesothelial cells and were supported by loose fibrovascular stroma. In cases 1 and 2, the stroma was expanded by edema and infiltrated by minimal to small numbers of neutrophils, macrophages, lymphocytes, and rare plasma cells. In case 2, the affected right auricular epicardium was thickened by MPH and covered with blood clots and fibrin, and the mesothelial covering was occasionally ulcerated. Cases 3 and 4 did not exhibit inflammation. There was no evidence of bacterial, fungal, or protozoal infection in Gram-, periodic acid–Schiff (PAS)-, and hematoxylin and eosin (H&E)-stained sections. Masson trichrome (MT) and picrosirius red (PR) stains were performed in all cases to determine the extent of and orientation of collagen fibers within the hyperplastic foci, respectively. MT stain revealed that all cases had variable amounts of collagen within the fibrovascular stroma (Fig. 3). PR stain showed various amounts of disorganized, green and yellow birefringent collagen fibers within the stroma, which was in stark contrast to the thick, layered bundles of subepicardial collagen (Fig. 4).
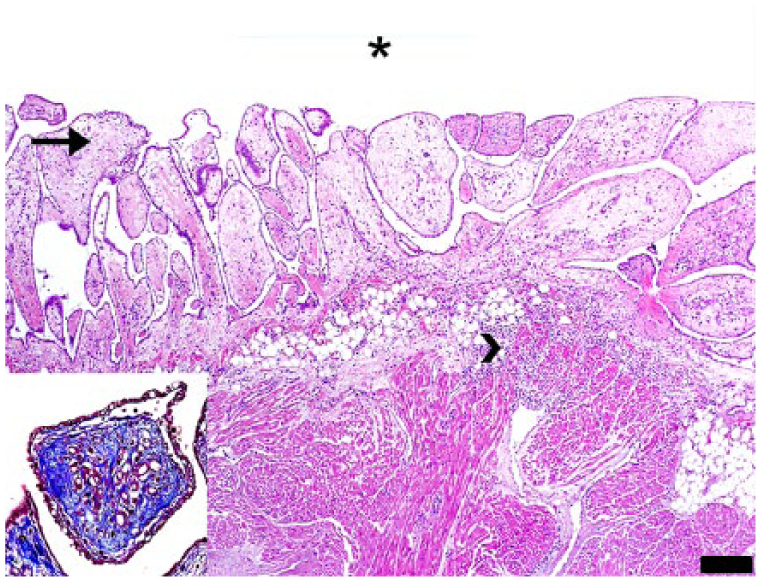
The epicardial surface is covered by variably sized, mesothelial cell–covered, papillary fronds that project into the pericardial space (asterisk) in dog 1. The papillary projections are supported by an edematous fibrovascular stroma (arrow) and infiltrated by variable numbers of neutrophils and fewer lymphocytes and plasma cells that extend into the underlying myocardium (arrowhead). H&E. Bar = 200 μm. Inset: Masson trichrome stain highlights the variably dense fibrous stroma.
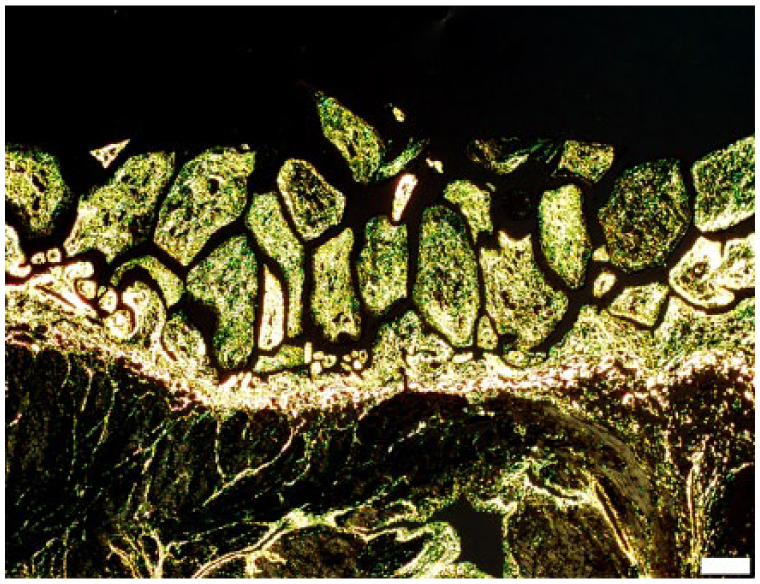
Papillary epicardial fronds in dog 1 are supported by a core of disorganized collagen fibers that exhibit variable, green-to-yellow birefringence under polarized light. Picrosirius red stain, polarized light. Bar = 200 μm.
Hyperplastic epicardial plaques in humans typically occur on the right atrium or right ventricle. 7 These changes are referred to as “milk spots” or “soldiers’ spots” and have been associated with advanced age and underlying cardiac insufficiency related to atrial or ventricular enlargement. 7 Lesions are thought to occur as a result of chronic physical irritation caused by constant friction between the affected epicardial surface and the pericardium because of underlying cardiac lesions.1,5,7 These changes can also result from chronic contact between the epicardium and the sternum or the vertebral bodies in cases of cardiomegaly and kyphoscoliosis, respectively. 7
The epicardium is a dynamic structure, and the mesothelium has been shown experimentally to proliferate as a sequel to cardiac damage, such as myocardial infarct in humans.2,4,7 In zebrafish that underwent experimental cardiac ventricular apex resection, there was induction of epicardial expression of Raldh2 in the adjacent ventricular epicardium. 4 The induction of Raldh2, which encodes the rate-limiting enzyme for retinoic acid synthesis, was followed by mesothelial cell activation and proliferation, which created a protective covering for the exposed myocardium. In zebrafish, epicardial Raldh2 expression was first enhanced in the area of the outflow tract and atria by 6–12 h post-ventricular resection, followed by the ventricular epicardium by 24 h post-resection. 4 Based on our findings and other published cases, the most severe MPH changes appear to be present over the atrial and auricular surface, suggesting that this may be an area of early mesothelial cell proliferation in dogs. In future studies, it would be interesting to examine mesothelial cell Raldh2 expression by immunohistochemistry in dogs with MPH.
Cases 2–4 in our study had myxomatous degeneration of the mitral and/or tricuspid valves with clinical, gross, and histologic evidence of cardiac insufficiency. Although establishing a causal relationship between MPH and underlying cardiac disease is not possible in the described cases, it is interesting to consider whether chronic stretching or friction of the epicardium caused by myocardial hypertrophy or dilation may have resulted in MPH, similar to the pathogenesis of hyperplastic epicardial plaques in humans. 7 One major limitation of our study is that fresh tissue was not available for culture to rule out an infectious process. However, the inflammation present in cases 1 and 2 was mild and may be secondary to friction between the auricular epicardial surface and the pericardium. The MPH lesion in case 2 also had surface-associated fibrin, which may have resulted from vascular leakage from the fibrovascular core as a result of frond ulceration. The absence of systemic signs, such as fever, the absence of bacterial, fungal, or protozoal organisms on Gram stain and PAS reaction on multiple tissue sections, and the fact that these changes are identical to those previously described in Beagles indicate that MPH is a reactive epicardial response rather than an infectious process. Cases 3 and 4 did not contain inflammatory cells in the MPH lesions, further suggesting that an infectious etiology is unlikely.
It is possible that the presence of chronic cardiac changes in cases 2–4 could have played a role in the pathogenesis of MPH in these dogs. However, the presence of MPH in case 1 (acute hemopericardium and no other cardiac changes) also indicates that these hyperplastic changes may occur as an incidental finding, a hypothesis supported by the high incidence of MPH in laboratory Beagles.1,5 Additional investigation into the pathogenesis and overall frequency of canine MPH is necessary to elucidate the significance and relationship, if any, of MPH to cardiac disease in dogs.
Regardless of its clinical significance and whether MPH occurs as a primary or secondary cardiac change, the location and gross features of the epicardial lesions in our cases warrant their gross and histologic differentiation from fibrinous epicarditis or neoplasia.7,8,10 Fibrinous epicarditis in dogs is typically associated with septicemia, and the fibrinous exudate should detach easily from the epicardial surface and not firmly adhere to it, as was observed in our cases. 8
Mesothelioma and cardiac hemangiosarcoma were suspected in cases 1 and 2, respectively, based on the gross appearance of the lesions. Cytologic differentiation of mesothelioma from MPH may be difficult given the similar morphologic appearance of neoplastic and reactive mesothelial cells, and histologic examination of the epicardial tissue may be required for a definitive diagnosis. In the case of mesothelioma, histopathology would reveal a densely cellular neoplasm with variable cellular morphology, which can be classified as epithelioid, sarcomatoid, or biphasic, as opposed to papillary fronds covered by a single layer of mesothelial cells in the case of MPH. 6 Cardiac hemangiosarcoma is typically composed of poorly differentiated spindle cells, which form variably sized blood-filled channels. Although densely cellular hemangiosarcoma may be confused with other neoplastic spindle cell growths, histologic examination of MPH would easily distinguish it from hemangiosarcoma given the single layer of mesothelium and overall low cellularity within the fibrovascular core of the papillary projections.
MT and PR stains were useful to highlight the degree of fibrosis within the stroma in areas of MPH. Although PR has historically been used to evaluate the types of collagen within lesions, the ability of this stain to discern between collagen types under polarized light has been refuted. 3 In the PR-stained sections, the presence of loose, green birefringent collagen bundles within the fibrovascular cores, compared to the dense, yellow birefringent collagen of the subepicardium, may suggest active remodeling of the epicardium in these cases.
The 4 current cases were diagnosed between 2013 and 2016. During this period, 1,549 dogs were submitted for autopsy at the AVDL. A search through our web-based system using key words such as cardiac mesothelial papillary hyperplasia, epicardial hyperplasia, mesothelial hyperplasia, and mesothelial plaques did not reveal any entries, indicating a low prevalence of MPH in dogs in our routine caseload. However, these results must be interpreted carefully because MPH may have been considered an incidental finding or overlooked and thus not recorded during autopsy.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
