Abstract
Cardiac mesothelial hyperplasia forming pale plaque lesions on the epicardial surface is a common incidental finding in the hearts of aged humans. A similar phenomenon with a more papillary appearance has also been reported as an incidental finding in dogs and mice. These lesions are believed to occur in response to friction between the epicardium and overlying pericardium. We investigated this lesion in adult cattle, a phenomenon that has been associated with bovine leukemia virus infection and epicardial lymphoma. We examined 73 hearts from adult cattle, predominantly of dairy breeds: 53 from a rendering facility and 20 from a state diagnostic laboratory. Cardiac mesothelial hyperplasia was much more prevalent in cattle than in other reported species (97% of examined hearts). The most common distribution was overlying the great vessels in a dark red papillary pattern. Cardiac mesothelial hyperplasia was also variably observed on all 4 cardiac chambers and the pericardium. Occasionally these lesions took on a smooth plaque-like appearance resembling those observed in humans. The lesions varied from 0.25 cm2 to covering 90% of the epicardial surface. No association was observed between cardiac mesothelial hyperplasia and bovine leukemia virus infection or cardiac lymphoma. Cardiac mesothelial hyperplasia was a common incidental finding in bovine hearts that must be distinguished from neoplasia and acute or chronic inflammation.
Introduction
Fibrous plaques on the epicardial surface of the heart, also known as soldier patches or milk spots, are common findings in aged humans.8,11 Such lesions appear as raised smooth white plaques most common on the anterior epicardial surface of the right ventricular myocardium. The frequency of these lesions increases with age and cardiac enlargement, or with skeletal abnormalities that lead to increased contact between the pericardial and epicardial surfaces. These lesions are considered incidental but are important to recognize in order to distinguish from pericardial disease. Similar lesions have been recognized as an incidental finding in dogs, and in mice with sternal dislocations.1,2,5,6,9 Initial reports in dogs were as an incidental microscopic finding in experimental Beagles; however, more recently, grossly discernible epicardial proliferations have been identified in dogs with cardiac abnormalities.2,5,6,9 These lesions are thought to occur in response to friction between the epicardial surface of the heart and the overlying pericardium.1,5,8
A morphologically similar epicardial proliferation has been reported in cattle in association with cardiac lymphoma. 10 The occurrence of hemorrhagic pericardial effusion in association with fibrous and papillary thickening of the epicardium has been reported as a precursor to a distinct epicardial presentation of bovine leukemia virus (BLV)-associated lymphoma. 7 The occurrence of cardiac mesothelial hyperplasia (CMH) has not been reported previously in clinically healthy cattle, to our knowledge. We determined the prevalence of CMH in adult cattle and investigated the association between CMH and BLV infection and/or BLV-associated lymphoma.
Materials and methods
We examined bovine hearts from 2 different sources: 53 adult cattle submitted for rendering, and 20 cattle >1 y old submitted to the Tulare branch of the California Animal Health and Food Safety (CAHFS) laboratory for autopsy. Rendering submissions were examined from October 2017 to January 2018, and the diagnostic submissions were examined from October 2017 to October 2018. Selection of animals was based on convenience. For the diagnostic submissions, age was determined from submission forms provided with the carcass. For the rendering submissions, adult status was assigned by the rendering facility based on size of the carcass, and only the hearts were available for direct examination. For the diagnostic submissions, the breed and diagnoses were determined (Table 1). For all submissions, the epicardial surface of all 4 cardiac chambers and the proximal great vessels was examined for CMH. For the diagnostic submissions, the parietal pericardium was also examined for CMH, and the presence of excessive pericardial fluid was noted. For the rendering submissions, hearts were removed from the carcasses prior to examination and therefore the pericardium was unavailable for examination, and quantification of pericardial fluid was not possible.
Descriptive data for diagnostic cases of cardiac mesothelial hyperplasia.

The approximate size, color, and morphology of any CMH lesions were recorded for 66 hearts (detailed morphometrics of the CMH were not performed on the first 5 diagnostic cases). Size of the observed lesions was either measured in 2 dimensions or, in severe cases, estimated as a percentage of the surface affected. In addition, all 4 chambers of the heart were opened, and the chambers, valves, and great vessels were examined for lesions. Tissue sections were examined histologically on a subset of 8 hearts. Masson trichrome for collagen was performed as well as CD31 and pancytokeratin immunohistochemistry to highlight endothelial and mesothelial cells, respectively. Submissions with cardiac lymphoma were stained with B- and T-cell markers CD79a and CD3, respectively.
When available in diagnostic submissions, blood was tested for BLV by ELISA (VMRD, Pullman, WA) that recognizes antibodies to BLV glycoprotein 51. For all other submissions, fluid from a piece of frozen and thawed spleen was used as a blood substitute for ELISA testing. With the exception of using splenic fluid as a sample, testing was performed according to the manufacturer’s directions. BLV is a retrovirus, and a positive ELISA result was interpreted as indicating infection.
To confirm the validity of using spleen fluid as a blood substitute, especially in the autolyzed tissues from rendered animals, paired blood and spleen fluid samples were tested in a subset of 13 submissions. Only animals that arrived alive and were euthanized on site were used for this subset. Five pieces of spleen were collected and placed in individual Whirl-Pak bags (Nasco, Fort Atkinson, WI). Spleen pieces were exposed to 1 of 5 different handling conditions: immediate freezing, 24 or 48 h at room temperature (~21°C), 24 or 48 h at 37.5°C. Following these steps, the spleen samples were frozen at −20°C for 5–30 d, thawed, manually macerated, and then free fluid was collected.
Results
CMH was grossly discernible in 71 of 73 (97%) hearts. Lesions were observed on the epicardial surface of all 4 heart chambers, as well as over the great vessels and pericardium. Detailed morphometrics were collected from 66 of the hearts with CMH. The number of lesions per heart varied from 1 to 17, and 33 hearts (50%) had lesions involving >1 region. The most common location was over the great vessels (
Distribution of cardiac mesothelial hyperplasia and bovine leukemia virus status in 73 bovine submissions.
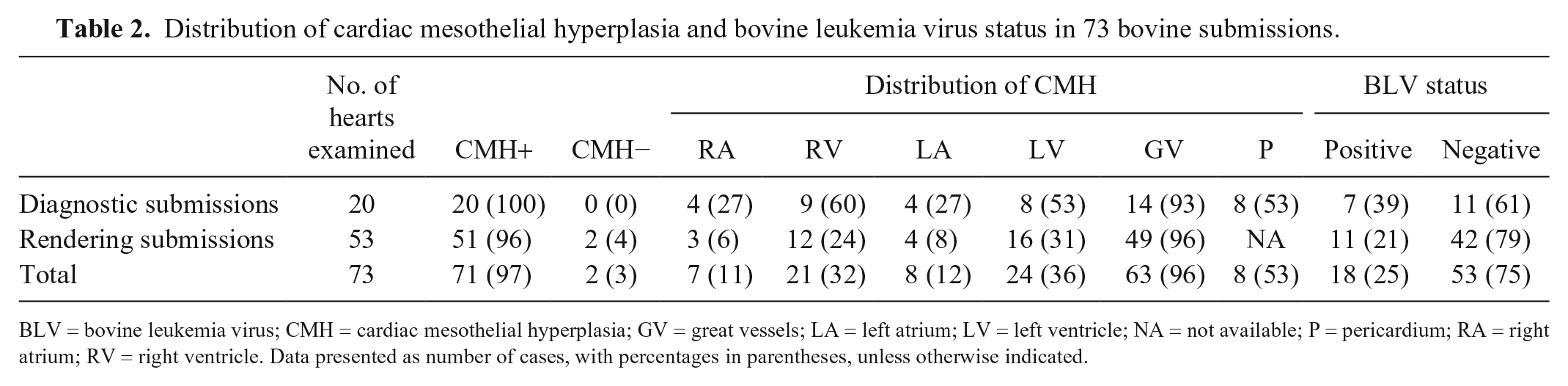
BLV = bovine leukemia virus; CMH = cardiac mesothelial hyperplasia; GV = great vessels; LA = left atrium; LV = left ventricle; NA = not available; P = pericardium; RA = right atrium; RV = right ventricle. Data presented as number of cases, with percentages in parentheses, unless otherwise indicated.
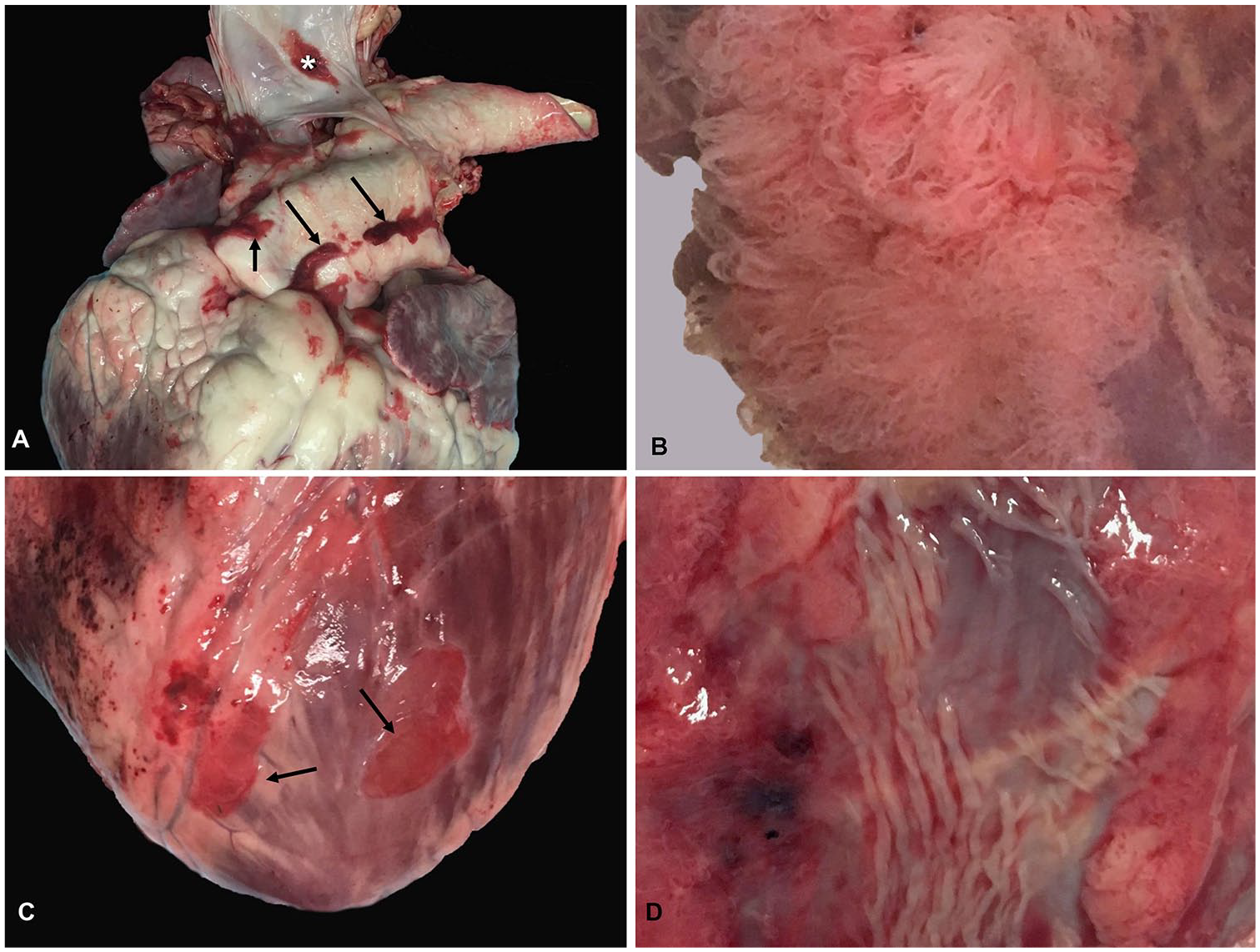
Cardiac mesothelial hyperplasia (CMH) in adult cattle.
Histologically, bovine CMH consisted of papillary fronds of variably dense fibrovascular tissue covered by a single layer of variably plump-to-attenuated mesothelial cells (Fig. 2A, 2B). Small numbers of lymphocytes and plasma cells infiltrated the collagenous stroma; neutrophils and macrophages were rare. Plaque lesions consisted of a variably thick band of fibrous tissue covered by a single layer of mesothelial cells (Figs. 2C, 2D). Immunohistochemistry for pancytokeratin and CD31 was performed to highlight mesothelial cells and blood vessels, respectively (Fig. 2E, 2F). Immunohistochemistry was also performed on one of the cases of cardiac lymphoma, confirming B-cell origin.
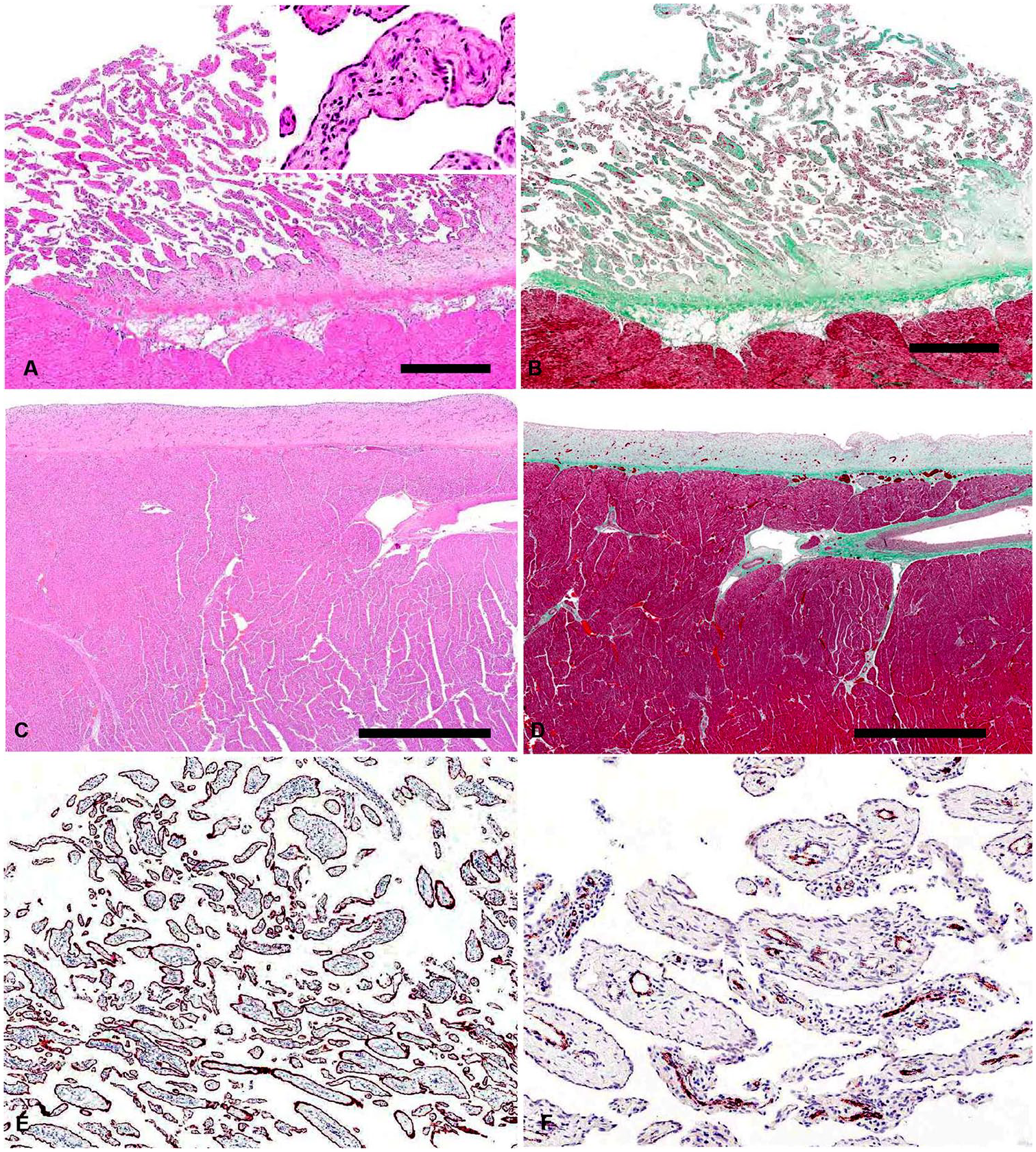
Histologic appearance of cardiac mesothelial hyperplasia (CMH) in cattle.
Spleen fluid ELISA results were consistent across all handling conditions. There was no significant difference in BLV ELISA results between blood and spleen fluid samples (Fisher exact test,
Additional cardiac lesions were only observed in 10 of 73 hearts (14%) within the 2 examined groups. The most commonly observed lesions were areas of localized endocardial fibrosis known as jet lesions (
Discussion
The 97% prevalence of CMH that we observed in cattle in our convenience sample is much greater than reported in dogs (microscopic incidence of 17%). 6 The predilection of CMH observed for the great vessels of cattle is distinct from the distribution of CMH in humans and dogs, which shows a predilection for the epicardium of the right ventricle and atria, respectively.5,8 The morphology of CMH in cattle exhibited similarities to both dogs and humans with the presence of both papillary and plaque lesions. Only papillary lesions have been reported in dogs; plaque lesions are the most common presentation in humans.5,11 Plaque CMH lesions in cattle appear to occur more commonly on the epicardial surface of the ventricular myocardium, as is the case in humans. This may suggest that the morphology of CMH is partially dependent on location on the heart and available space between the epicardium and the overlying pericardium. CMH lesions in cattle varied in color from dark-red to white. This may be the result of different vascularity between lesions.
CMH in humans, dogs, and mice is believed to be a chronic response to friction between the epicardium and overlying pericardium.1,5,8 This would explain why CMH appears to be more severe in conditions that increase the contact between these surfaces such as cardiac chamber enlargement or skeletal deformities such as sternal dislocations, pectus excavatum, or lordosis. Although friction may explain the occurrence of CMH on the epicardial surface of the ventricles and atria, it does not easily explain the occurrence of CMH around the great vessels in cattle. Epicardial stretching was proposed as an alternative cause of CMH in dogs. 5 Given the dynamic nature of the great vessels, this may help to explain the occurrence of CMH in this location. Alternatively, the absence of pericardial contact in this area may allow for proliferation of mesothelial tissue once stimulated.
Previous reports of CMH in cattle have suggested an association with lymphoma and BLV infection.7,10 Specifically, CMH was suggested as a precursor to a unique epicardial variant of BLV lymphoma. We found no correlation between CMH and BLV infection, or between CMH and cardiac lymphoma. BLV infection was observed in 25% of our samples; CMH occurred in 97% of hearts examined. We observed one case resembling the reported epicardial variant of BLV lymphoma. 7 In this case, CMH was particularly prominent because of infiltration of the fibrovascular tissue by neoplastic cells. It is possible that the highly vascular nature of CMH predisposes it to neoplastic infiltration in cattle with neoplastic lymphocytosis. In addition, CMH lesions that are enlarged by neoplastic infiltration may be more likely to be perceived and noted at autopsy.
Pericardial effusion has occasionally been reported in conjunction with both bovine and canine CMH cases.5,7 Two cases with pericardial effusion were observed of the 20 diagnostic cases that we examined. In both of these cases, CMH was very pronounced, occurring on all 4 chambers, the great vessels, and the pericardium. Perhaps the movement of excess pericardial fluid stimulated mesothelial cell proliferation. Alternatively, the highly vascular and delicate nature of CMH may lead to easy traumatization and predispose animals to the development of pericardial effusion. Idiopathic pericardial effusion is a common finding in cattle.3,4,7
CMH appears to be a common lesion in the hearts of adult cattle; we found CMH in 20 of 20 diagnostic submissions and 51 of 53 rendered carcasses. Although there is, to date, no evidence in cattle or other species to suggest that CMH is clinically significant, it is an important lesion to recognize to prevent misdiagnosis. Differentials for CMH in cattle and other species include neoplasia, specifically hemangiosarcoma or mesothelioma, and fibrinous epicarditis or fibrosis secondary to chronic inflammation. Histology may be required to distinguish CMH from neoplasia. Endothelial cells of CMH are bland relative to those of hemangiosarcoma, and although mesothelial cells of CMH may be plump and reactive, dysplasia is minimal and there is no evidence of infiltration into the underlying stroma or myocardium. CMH may be grossly distinguished from fibrin because it cannot be detached easily from the epicardial surface. It may be distinguished from chronic inflammation by the absence of adhesion to the overlying pericardium that occurs with chronic fibrosis and restrictive pericarditis. However, it is possible that a subset of CMH cases is the result of resolved mild epicarditis without the formation of adhesions. Given the prevalence of respiratory disease in cattle, associated subclinical pericarditis and epicarditis is likely common. Histologic examination of CMH generally reveals only mild mononuclear inflammation. All of these features help distinguish CMH from other clinically significant cardiac lesions. CMH should be considered a distinct cardiac entity of cattle and other species for which there is currently no evidence of an underlying infectious or neoplastic etiology.
Footnotes
Acknowledgements
We thank Kevin Keel and the University of California-Davis, Virtual Microscopy Laboratory, for help with slide scanning and image capture and editing. We also thank the USDA, cooperating rendering facilities, and Megan Parsons for help with sample collection.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
