Abstract
Bowenoid in situ carcinomas (BISCs) are papillomavirus (PV)-induced skin neoplasms that are thought to be caused by Felis catus papillomavirus (FcaPV) 2. As BISCs are typically multiple and can become extensive, they can be difficult to treat. Herein we describe 4 cats that developed skin neoplasms that contained FcaPV-3 DNA. One cat developed multiple basal cell carcinomas (BCCs), 1 a BISC with unusual extension into hair follicles, and 2 developed a single typical-appearing BISC. All neoplasms contained prominent PV-induced cell changes and intense p16CDKN2a protein immunostaining. Results from these 4 cats provide evidence that FcaPV-3 could cause a proportion of feline skin cancers, albeit less frequently than FcaPV-2. Excision of the typical BISCs and the BCCs appeared curative. Although the cat with the unusual BISC was euthanized because of the large size of the lesion, evidence from these 4 cats suggests that skin neoplasms that contain FcaPV-3 DNA may have a less aggressive clinical behavior than those associated with FcaPV-2. A consistent feature of the neoplasms in all 4 cats was the presence of prominent basophilic intracytoplasmic inclusion bodies; these inclusions have not been reported in lesions caused by FcaPV-2, to our knowledge, and their detection may allow differentiation between the different PV types and could therefore be a useful prognostic feature.
Keywords
Studies within the last decade have suggested that papillomaviruses (PVs) cause viral plaques and bowenoid in situ carcinomas (BISCs) 7 and may cause a proportion of cutaneous squamous cell carcinomas in cats.1,19 To date, Felis catus papillomavirus 2 (FcaPV-2), which is classified in the Dyothetapapillomavirus genus, is thought to cause the majority of skin disease in cats.1,13,16 Herein we describe 3 cats that developed BISCs and 1 cat that developed multiple feline basal cell carcinomas (BCCs) in which FcaPV-3, which along with FcaPV-4 and -5 probably represent a novel PV genus, 9 was identified. Although a single case has been reported of a BISC that contained only FcaPV-3 DNA sequences, 11 the development of BISCs containing FcaPV-3 DNA in multiple animals provides evidence of a possible role of this PV type in the development of feline BISCs.
Case 1 was a 14-y-old male domestic shorthair (DSH) cat that developed three <1 cm intradermal skin masses on the ventral neck. Excisional biopsy was performed, and histology revealed that all were horizontally orientated elongated masses present within the superficial dermis. One neoplasm had multifocal contiguity with the overlying epidermis. No contiguity was visible in the other 2 neoplasms; however, atypical cells were visible expanding follicular infundibula adjacent to both neoplasms suggesting contiguity may have been present within non-sectioned areas. Neoplastic cells were visible infiltrating from the tumor into the surrounding dermis, and all 3 neoplasms were classified as BCCs. The neoplastic cells were arranged in variably sized aggregates that were divided into nests and trabeculae by a moderate highly cellular fibrovascular stroma (Fig. 1). Palisading of cells was visible around the periphery of some cell trabeculae, and large cystic cavities that contained only small quantities of eosinophilic cell debris were frequent within the neoplasms. Prominent clefts were visible between the cell trabeculae and the surrounding stroma in one tumor. The neoplastic cells exhibited mild pleomorphism with the cells generally small, dark, and polygonal, with small dark nuclei and little cytoplasm. No spindle-shaped cells were visible within any of the neoplasms, and the neoplasms were not surrounded by a fibrous pseudocapsule. Prominent within all 3 neoplasms were cells with enlarged pale foamy nuclei surrounded by a darkly basophilic elongated intracytoplasmic body that assumed the shape of the nucleus (Fig. 2). Less commonly, cells were visible that had a shrunken eosinophilic or basophilic nucleus that was surrounded by a clear cytoplasmic halo (koilocytes). Neoplastic cells did not extend to tissue margins.
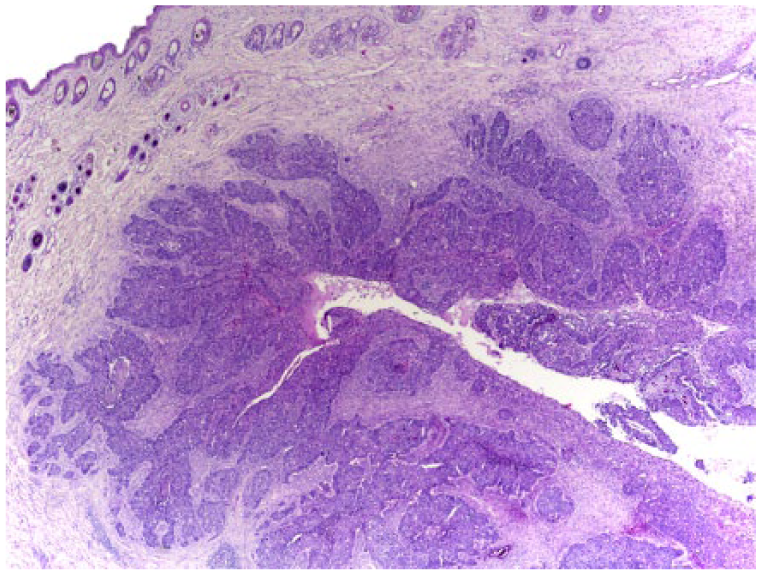
Basal cell carcinoma in case 1. The mass consists of a horizontally elongated proliferation of neoplastic epithelial cells arranged in nests and trabeculae surrounding multiple cystic cavities. Neoplastic cells are visible infiltrating the surrounding dermis. H&E.
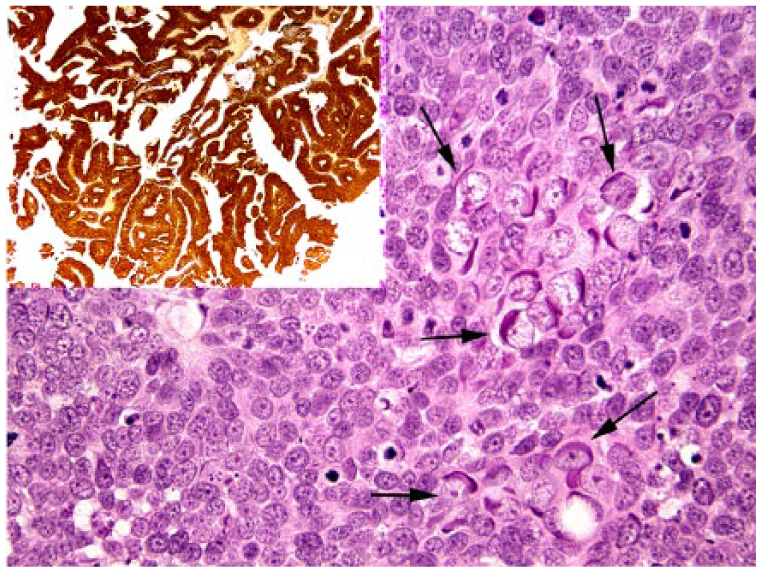
Basal cell carcinoma in case 1. Numerous cells that contain enlarged vesicular nuclei and prominent elongate basophilic intracytoplasmic bodies are visible within the neoplasm (arrows). H&E. The neoplastic cells consistently contain intense cytoplasmic and nuclear immunostaining using antibodies against p16CDKN2a protein (inset).
Given the histologic evidence of PV infection within the BCCs, DNA was extracted from formalin-fixed tissue as described previously. 14 To amplify PV DNA, the consensus PCR primers FAP59/64 and MY09/11 were used along with the JMPF/R primers that specifically amplify FcaPV-2 DNA.2,6,14 A BISC known to contain FcaPV-2 was used as the positive control for the FAP59/64 primers; a feline sarcoid containing bovine papillomavirus 14 was the positive control for the MY09/11 primers. No template DNA was added to the negative controls. DNA was only amplified from the feline BCCs by the MY09/11 primers, with sequencing subsequently revealing that all 3 neoplasms contained FcaPV-3 DNA sequences.
Immunostaining to detect p16CDKN2a protein (p16) was performed as reported previously 13 and revealed intense nuclear and cytoplasmic immunostaining within all of the neoplastic cells within the BCCs. Immunostaining was not visible within the surrounding epidermis or in histologically normal follicles within the sections.
Approximately 6 mo after the 3 BCCs were removed, an ulcerated 1.5-cm diameter mass was observed on the ventral neck. Histology revealed a BCC with large numbers of cell nests infiltrating the surrounding dermis. PV-induced cell changes that included cells with basophilic intracytoplasmic inclusions were prominent, and FcaPV-3 DNA was again the only PV type amplified. The neoplastic cells contained intense nuclear and cytoplasmic p16 immunostaining.
Three apocrine ductular adenomas were also excised from the limbs of this cat (case 1) at this time. These did not contain histologic evidence of PV infection, and no PV DNA could be amplified using the FAP59/64, MY09/11, or JMPF/R primers. Additionally, no increased p16 immunostaining was present. No additional masses have developed in the 1 y following the surgical excision of the BCC from the neck of case 1.
Case 2 was a 14-y-old male DSH cat that developed roughly symmetrical scabby lesions within both preauricular areas. Over the next 18 mo, the lesions slowly expanded, resulting in a raised plaque that extended from the pinna to the eyes bilaterally. The large size of the lesion prevented excision, but histologic examination of a sample of the lesion revealed thickening of the epidermis and underlying follicles. In contrast to the typical appearance of a BISC in which follicular changes are restricted to the infundibulum, thickening was present in the external root sheath extending to the level of the hair bulb (Fig. 3). Cells within the thickened follicles were supported by a minimal fibrous stroma that separated the cells into broad trabeculae and nests. Cells showed palisading along the periphery of the trabeculae, and the peripheral cells were smaller and darker than more centrally positioned cells. Cells within affected areas showed moderate-to-marked atypia, but were generally polygonal with small dark nuclei and moderate quantities of basophilic cytoplasm. Keratinization was not visible within the thickened follicles. Evidence of PV infection included numerous cells with enlarged pale vesicular nuclei with elongated basophilic cytoplasmic bodies. The lesion was classified as a BISC, although with unusual follicular changes.
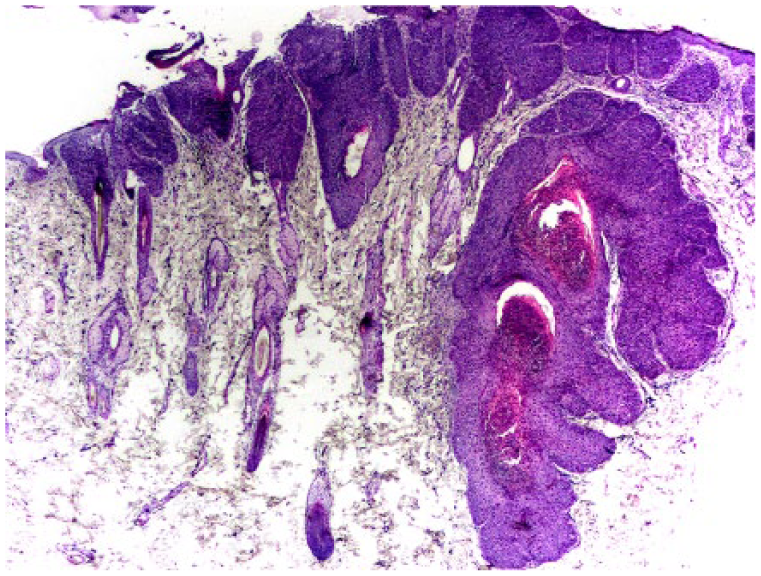
Bowenoid in situ carcinoma (BISC) in case 2. Consistent with previous reports of feline BISCs, thickening and dysplasia are visible within the epidermis. However, in contrast to typical BISCs in which follicular changes are confined to the infundibulum, thickening is present within the external root sheath extending close to the hair bulb. H&E.
Molecular and immunohistochemical analyses were performed as before and revealed that FcaPV-3 was the only PV present and that the cells had intense p16 immunostaining. Over the following 6 mo, the lesions continued to expand and became ulcerated. The cat was euthanized, but an autopsy was not performed.
Cases 3 and 4 were both 10-y-old male DSH cats that developed a single plaque-like scaly mass on the dorsal surface of the paw and nasal planum, respectively. Histology of both masses revealed well-demarcated foci of irregular thickening and dysplasia of the epidermis without significant follicular involvement. PV-induced cell changes, similar to those in the previous cases, were prominent within the affected epidermis (Fig. 4). The only PV detected in the lesions was FcaPV-3, and both lesions exhibited intense p16 immunostaining. Both were consistent with BISCs, although the basophilic elongated intracytoplasmic inclusions were not typical. Neither lesion recurrence nor additional lesion development has been observed in the 12 mo following excision.
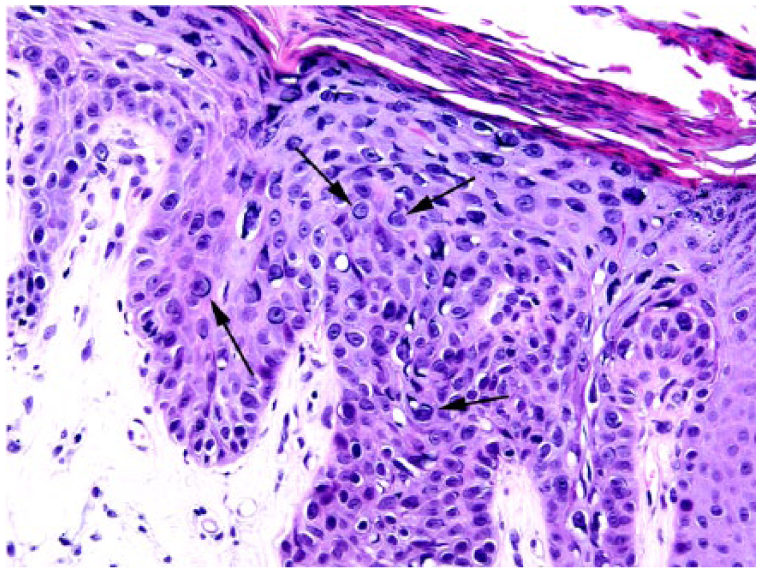
Bowenoid in situ carcinoma (BISC) in case 3. The epidermal changes observed are considered typical for a BISC. However, cells with enlarged vesicular nuclei and prominent basophilic elongate cytoplasmic inclusions, similar to those observed in the basal cell carcinoma, are visible (arrows). H&E.
Most skin disease caused by PVs in cats is currently thought to be caused by FcaPV-2. 7 Although a single case has been reported in which FcaPV-3 was the only PV type detected in a feline BISC, 11 the results from the 4 cats in our report provide additional evidence that FcaPV-3 could cause skin neoplasia in cats. Although the proportion of PV-induced skin lesions that contain FcaPV-3 DNA in cats is unknown, FcaPV-2 DNA has been consistently detected in large series of feline viral plaques and BISCs,14,15 suggesting that FcaPV-3 is comparatively rarely associated with skin disease in cats.
The lesions observed in the 4 cats ranged from the presence of mild BISC-like changes with little follicular involvement to the development of a BCC within the dermis. Interestingly, case 2 had lesions that included changes in both the epidermis as well as the deep segment of the hair follicle. The apparent ability of FcaPV-3 to infect cells within the epidermis and deeper within the follicles has not been previously reported, to our knowledge, and may explain why this PV type can be present in both BISCs and BCCs. In contrast, FcaPV-2 appears to be restricted to infecting cells in the surface epidermis and follicular infundibulum, and has not been associated with the development of BCCs in cats. 4 Given that PVs tend to be highly specific in the cells that they are able to infect, 17 it is perhaps unsurprising that FcaPV-3 and FcaPV-2 have small differences in the range of cells that they can infect.
Whether lesions that contain FcaPV-3 DNA have a different clinical behavior to those associated with FcaPV-2 is currently unknown. However, cats with BISCs, presumably caused by FcaPV-2, typically develop multiple lesions that are often too numerous to excise surgically. 3 In contrast, none of the cats in our report had >4 lesions, and single lesions were observed on 2 cats. Furthermore, in contrast to cats infected by FcaPV-2 that often rapidly develop additional BISCs, 3 surgical excision of the lesions that contained FcaPV-3 DNA appeared to be curative in 3 of the 4 cats described herein. The BISC in case 2 did result in euthanasia, suggesting that lesions that contain FcaPV-3 DNA can potentially be progressive and involve large areas of skin. However, given that the lesions in this cat had been present for 18 mo prior to diagnosis, it is possible that more prompt intervention could have been curative. Although additional cases are required, there is accumulating evidence that FcaPV-3 may cause skin neoplasms that have a less aggressive clinical course than those associated with FcaPV-2.
The histologic appearance of the lesions in the 4 cats was diverse. However, all contained cells with prominent basophilic intracytoplasmic inclusions. Such inclusions have been reported previously in a BISC that contained only FcaPV-3 sequences. 11 However, basophilic intracytoplasmic inclusions have not been reported in lesions associated with FcaPV-2. Therefore, histology is likely to enable differentiation between FcaPV-2 and FcaPV-3 and, if prognostic differences between lesions caused by the 2 PV types are proven, these basophilic inclusions could be an easily observed prognostic feature. As FcaPV-2 and FcaPV-3 are classified within different genera,5,10 it is unsurprising that infection by these 2 PV types produce different PV-induced cell changes within the lesions.
The neoplasms observed in case 1 were classified as BCCs. Although differentiation of a BCC from a follicular or apocrine gland neoplasm can be difficult, features that support a basal cell origin in this case include the horizontal orientation of the masses within the dermis. Additionally, the presence of prominent clefts between the cell trabeculae and the surrounding stroma has been reported to be a diagnostic feature of BCCs in people. 4 Furthermore, the absence of spindle-shaped cells or a fibrous pseudocapsule within the neoplasms is more consistent with a BCC than a neoplasm of apocrine gland or follicular origin. 4 BCCs with histologic evidence of PV infection have been described previously, and sequences from a novel PV type were reported in a feline BCC.4,12 However, the detection of FcaPV-3 within a feline BCC increases the spectrum of skin neoplasms that could be influenced by this PV type. To our knowledge, multiple BCCs have not been reported previously in a cat. Given that BCCs are considered to be rare neoplasms of cats, the development of 3 BCCs on the same animal suggests that a predisposing factor was likely to have influenced tumor development. Although it cannot be definitively confirmed that infection by FcaPV-3 was the factor that predisposed to development of multiple neoplasms in this cat, the presence of PV-induced cell changes supports the hypothesis of a causal association between FcaPV-3 and the multiple BCCs in this cat.
Given that PVs have co-evolved with their hosts over a long period of time, most PV infections are asymptomatic. 18 Therefore, the amplification of PV DNA from a lesion does not prove causality. In cats, FcaPV-2 is generally considered to cause BISCs not only because of the presence of PV-induced cell changes within a proportion of tumors, but because of the more frequent detection of FcaPV-2 in BISCs than normal skin, the higher FcaPV-2 viral loads in BISCs than normal skin, and the consistent detection of FcaPV-2 RNA in BISCs, but not normal skin samples.7,14,18,19 In addition, a role of FcaPV-2 in BISC development is supported by the increased p16 immunostaining observed in these lesions. 8 This protein increases as a result of the consistent action of the PV E7 protein–degrading retinoblastoma protein, which, in turn, increases p16 within a cell. 13 In the cats in our report, a role of FcaPV-3 in lesion development was supported by the presence of histologically detectible PV-induced cell changes within the neoplastic cells. In addition, FcaPV-3 DNA was detected in lesions from all 3 cats, but this PV was not identified in non-lesional skin from 1 of the affected cats, and this PV has been rarely detected in previous studies investigating the presence of PVs on the skin of cats. 8 Furthermore, the presence of intense p16 immunostaining in lesions from all 4 cats is consistent with PV infection influencing their development. Although the specificity of p16 for a PV etiology is unknown in cats, increased p16 immunostaining was not present in the apocrine tumors from case 1 that did not contain histologic evidence of PV infection or amplifiable PV DNA sequences. Our results support a role of FcaPV-3 in lesion development, but it is not possible to definitively differentiate between FcaPV-3 influencing neoplasm development and FcaPV-3 being present as a result of a more permissive environment.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
