Abstract
Viral plaques are well recognized skin lesions of cats. They are thought to be caused by papillomavirus infection; however, the causative papillomavirus is uncertain. In the current study, polymerase chain reaction using 2 consensus primer sets and 1 primer set specific for Felis domesticus papillomavirus 2 (FdPV-2) was used to amplify DNA from a series of 14 feline viral plaques. The FdPV-2 sequences were detected in all 14 viral plaques by the specific primers but in only 1 of 14 feline cutaneous trichoblastomas. Papillomavirus DNA was amplified from 8 plaques using the consensus primers. Sequences from FdPV-2 were amplified using the consensus primers from 4 plaques. In addition, 3 plaques contained papillomavirus DNA sequences from Felis domesticus papillomavirus sequence MY1, and a previously unreported papillomavirus DNA sequence was amplified from 1 plaque. As FdPV-2 was consistently present within the plaques, this suggests that this papillomavirus is the likely etiologic agent. Feline viral plaques can undergo neoplastic transformation to Bowenoid in situ carcinomas (BISCs). As FdPV-2 DNA is frequently present within BISCs, this suggests that FdPV-2 induces viral plaque formation and then remains detectible after neoplastic transformation.
Viral plaques are uncommon feline skin lesions that are thought to be caused by papillomavirus infection. 7 Although some plaques prove to be of little clinical significance, a proportion progress to Bowenoid in situ carcinomas (BISCs) and, subsequently, to invasive squamous cell carcinomas (SCCs). 7,11,20 Previous studies have revealed that feline viral plaques consistently contain histologic and immunohistochemical evidence of papillomaviral infection. 20 However, only 2 viral plaques have been investigated to determine which papillomavirus species is associated with these lesions. In one of the viral plaques, Felis domesticus papillomavirus 1 (FdPV-1) was identified, 18,19 while 3 papillomaviruses, including FdPV-1, Felis domesticus papillomavirus 2 (FdPV-2), and a novel papillomavirus, designated Felis domesticus papillomavirus sequence MY1 (FdPV-MY1), were detected in swabs of the other. 14 In the present study, polymerase chain reaction (PCR) was used to amplify papillomavirus DNA from a series of feline viral plaques. The consistent detection of a single papillomavirus species within the plaques would provide evidence of a causal relationship.
Fourteen viral plaques and 14 trichoblastomas were identified by retrospective surveys of databases at Massey University (Palmerston North, New Zealand), New Zealand Veterinary Pathology Ltd. (Palmerston North, New Zealand), Cornell University (Ithaca, New York), and Gribbles Veterinary Pathology Ltd. (Hamilton, New Zealand). The diagnosis within the databases was confirmed using histologic examination of a hematoxylin and eosin-stained section and previously reported diagnostic criteria. 7 Briefly, all plaques consisted of focal moderate to marked epidermal hyperplasia with orthokeratosis and prominent keratohyaline granules. The plaques contained variable numbers of keratinocytes that had increased quantities of blue-gray fibrillar cytoplasm, cytoplasmic vacuoles, and dark shrunken nuclei that were often peripherally displaced. Keratinocytes within the deeper layers of the affected epidermis often contained a centrally placed dark shrunken nucleus that was surrounded by a clear cytoplasmic halo (koilocytes). Neither intranuclear viral inclusions nor cellular dysplasia was visible within any of the viral plaques. The average age of the cats with viral plaques was 9.5 years, although individual cats ranged from 2 to 14 years old. None of the cats was reported to have clinical evidence of immunosuppression or concurrent disease.
As previously described, 13 DNA was extracted from formalin-fixed, paraffin-embedded tissue. Three sets of PCR primers were used to amplify sections of the papillomavirus L1 gene from the viral plaques. Both FAP59/64 and MY09/11 are consensus primers that amplify DNA from papillomaviruses in multiple species. 2,5 In contrast, the JMPF/R primers were designed to specifically amplify DNA from FdPV-2. 12 The reaction conditions for the primer sets have been previously described. 12 All PCR reactions were carried out in duplicate. Positive controls for the FAP59/64 and MY09/ 11 primers were DNA extracted from a canine oral papilloma and a bovine fibropapilloma, respectively, while the negative control did not contain template DNA. The DNA extracted from a BISC was used as a positive control for the JMPF/R primers while DNA extracted from a canine oral papilloma and DNA extracted from a bovine fibropapilloma were included as negative controls. All DNA amplified by the consensus primers and from the 4 viral plaques by the JMPF/R primers was sequenced as previously described. 13
Age, breed, and papillomaviruses detected within 14 cats with viral plaques. *
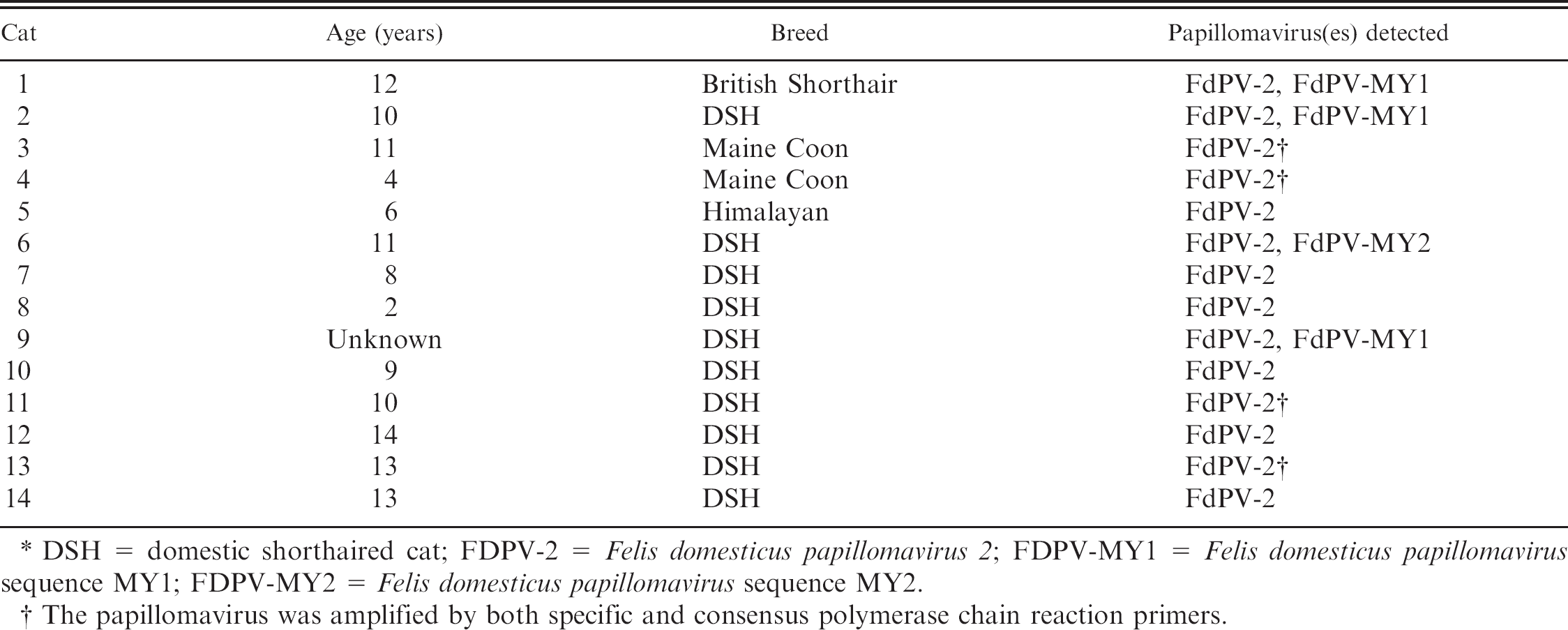
DSH = domestic shorthaired cat; FDPV-2 = Felis domesticus papillomavirus 2; FDPV-MY1 = Felis domesticus papillomavirus sequence MY1; FDPV-MY2 = Felis domesticus papillomavirus sequence MY2.
The papillomavirus was amplified by both specific and consensus polymerase chain reaction primers.
The DNA was amplified using the JMPF/R primers from all 14 feline viral plaques (Table 1), but just 1 of 14 trichoblastomas. Sequencing of DNA amplified from 4 of the plaques revealed greater than 99% similarity to FdPV-2. Additional sequencing of the DNA amplified by the JMPF/R primers was considered unnecessary because of the consistent results in these 4 cases and the previously demonstrated specificity of these primers for FdPV-2. 15 Although DNA was amplified from the BISC, the specific primers did not amplify DNA from either the canine oral papilloma or the bovine fibropapilloma.
The MY09/11 primers amplified papillomavirus DNA from the positive control and from the 4 viral plaques. Sequencing the DNA from the plaques revealed the presence of 2 different papillomavirus sequences. Three of the plaques contained sequences that were greater than 99% similar to the previously described FdPV-MY1. 14 The 375-base pair (bp) sequence was 66% similar to human PV-80, but only 60% similar to FdPV-2 and 59% similar to FdPV-1. One viral plaque contained a papillomavirus DNA sequence that had not been previously reported; it was most similar to FdPV-MY1 with 68% similarity between the 2 DNA sequences. The novel sequence was 65% similar to human PV-120, but only 59% and 58% similar to FdPV-1 and FdPV-2, respectively. This sequence was designated Felis domesticus papillomavirus sequence MY2 (FdPV-MY2; GenBank accession no. HM130736). Examination of the 4 plaques that contained DNA sequences from multiple papillomaviruses did not reveal any additional histologic features.
Using the FAP59/64 primers, papillomavirus DNA was amplified from the positive control and from the 4 viral plaques. Sequencing revealed that the DNA amplified from the plaques was 98% similar to FdPV-2. The small amount of variation within the sequences was considered to be due to sequencing errors rather than evidence of the presence of different papillomavirus variants. No additional histologic features were visible within the samples from which both the consensus and specific primers amplified FdPV-2.
Feline viral plaques typically appear as multiple, up to 8 mm in diameter, pigmented or nonpigmented, raised lesions. 7,20 They can develop anywhere on the body and have been reported in cats from 7 months to 15 years old. 17 Although there is some evidence that immunosuppression may predispose cats to plaque formation, 4,6,14 plaques also develop in cats without any detectible immunosuppressive disease. 20 Although Persian cats were initially thought to be predisposed, 4 none of the currently described plaques and none of the plaques within a previous series of affected cats were of this breed. 20
Feline viral plaques are thought to be caused by papillomavirus infection. 7 Supporting a papillomavirus etiology, viral plaques consistently contain viral cytopathic changes, including koilocytosis, cells with increased quantities of blue-gray cytoplasm, and clumped keratohyaline granules. 7,20 Additionally, papillomavirus antigen is detectible using immunohistochemistry in most viral plaques. 20 The presence of viral cytopathology and papillomavirus antigen indicates that viral replication is present within the plaques. 9 To the authors' knowledge, only 2 viral plaques have been investigated to determine the papillomavirus present. One plaque was found to contain FdPV-1 DNA, 18,19 while swabs of the second plaque contained DNA from 3 papillomaviruses, including FdPV-1, FdPV-2, and FdPV-MY1. 14 Because of the small number of plaques investigated and the identification of multiple papillomaviruses within these 2 plaques, it was not possible to determine the causative papillomavirus. In the present study, FdPV-2 DNA was consistently present within all 14 plaques, but it was only detected in 1 of 14 trichoblastomas. This suggests that FdPV-2 is the cause of feline viral plaques.
Felis domesticus papillomavirus 2 was initially detected in a series of BISCs. 13 By using specific primers, FdPV-2 DNA was detected in 20 of 20 BISCs, but in only 3 of 17 non-SCC feline skin lesions, indicating a significant association between BISCs and FdPV-2. 12 Bowenoid in situ carcinomas are thought to develop as a result of neoplastic transformation of a viral plaque. 20 Therefore, the results of the present study suggest that FdPV-2 induces plaque formation and then remains detectible within plaques that have undergone neoplastic transformation. Whether or not FdPV-2 influences the progression of a viral plaque to a BISC remains uncertain. 11 However, both viral plaques and BISCs contain altered tumor suppressor protein expression, suggesting a possible oncogenic action of FdPV-2. 10 The complete sequence of FdPV-2 was recently determined, 8 suggesting that the papillomavirus should be classified within a newly proposed genus, Dyothetapapillomavirus. 3
Although these results indicate that FdPV-2 is likely to be the cause of feline viral plaques, the pathogenesis of this disease is uncertain. Feline viral plaques show many histologic and clinical similarities to human epidermodysplasia verruciformis (EV), 7 a rare skin disease that develops because the body is unable to make an immune response against cutaneous papillomavirus infection. 16 Most people are asymptomatically infected by cutaneous papillomaviruses. 1 However, in people with EV, the loss of immunity allows papillomaviruses that normally result in asymptomatic infection to induce clinical lesions. 16 Similarly, FdPV-2 DNA is detectable in skin swabs of approximately 50% of clinically normal cats. 15 However, viral plaques develop in only a small proportion of infected cats. Human EV develops most frequently due to inherited defects within the EVER1 or EVER2 genes. 16 Further investigation is required to determine why asymptomatic FdPV-2 infections progress to viral plaques in some cats.
In contrast to earlier studies, FdPV-1 was not detected within any plaque. This could suggest that FdPV-1 is an uncommon cause of plaque development. Alternatively, FdPV-1 DNA was detected within swabs of clinically normal feline skin, 14 suggesting that this virus may have been previously detected as an asymptomatic infection rather than the cause of the viral plaques. However, FdPV-1 cannot be excluded as the cause of the viral plaques, though it was not detected. The consensus primers used in the current study have previously amplified FdPV-1 14 and canine oral papillomavirus, which is closely related to FdPV-1, 18 and was used as a positive control in the current study. However, the sensitivity of the consensus primers for FdPV-1 is unknown, which suggests that small quantities of DNA may not have been detected within the plaques. Interestingly, FdPV-1 has not been amplified from a BISC, suggesting that if FdPV-1 is the cause of viral plaques, the papillomavirus DNA does not remain detectible after neoplastic transformation.
Of the 14 viral plaques, 4 contained multiple papillomavirus types. Three viral plaques contained sequences from FdPV-MY1, and FdPV-MY1 sequences have previously been amplified from a swab of a viral plaque and a swab of clinically normal skin from a cat that was infected with Feline immunodeficiency virus. 14 One viral plaque contained a papillomavirus sequence (designated FdPV-MY2) that has not been previously reported. Papillomaviruses are classified using the L1 gene. 3 In the present case, as only a segment of the L1 gene was amplified, definitive classification of FdPV-MY2 is not possible. However, this papillomavirus appears to be most closely related to FdPV-MY1 and dissimilar to FdPV-1 and FdPV-2. The role of FdPV-MY1 and FdPV-MY2 in the development of viral plaques is uncertain. While it is possible that they could influence plaque formation, their inconsistent detection suggests that neither FdPV-MY1 nor FdPV-MY2 is necessary for plaque formation. As FdPV-MY1 sequences were previously detected within a swab of clinically normal skin, 14 both papillomaviruses could asymptomatically infect feline skin and be unrelated to plaque formation.
In the present study, the FAP59/64 consensus primers amplified FdPV-2 DNA from 4 of the 14 plaques. In contrast, primers designed specifically to detect FdPV-2 DNA amplified DNA from all 14 plaques. The greater sensitivity of the specific primers could be due to the increased similarity and subsequent higher affinity of the specific primers for FdPV-2 DNA. Alternatively, the FAP59/64 primers amplify a 480-bp segment of DNA while the JMPF/R primers amplify just 177 bp. 12 Formalin fixation can result in fragmentation of the DNA, 14 and the longer the section of DNA amplified, the greater the chance that amplification will be prevented by the presence of fragmented DNA. It is unknown if FdPV-2 was amplified by the consensus primers in 4 samples because these samples contained a higher concentration of papillomavirus DNA or contained less fragmentation within the DNA.
In conclusion, the current report is the first time multiple feline viral plaques have been evaluated to determine which papillomavirus are present. The results revealed that feline viral plaques consistently contained FdPV-2 DNA, which suggests that FdPV-2 is likely to be the cause of feline viral plaques. Four plaques contained sequences from multiple papillomaviruses, including 1 papillomavirus that has not been previously reported. However, as these additional papillomaviruses were inconsistently present, their role in viral plaque development is uncertain. As FdPV-2 commonly infects feline skin asymptomatically, 15 most cats that are infected with this papillomavirus do not develop viral plaques. Additional research is required to determine why FdPV-2 infection results in plaque formation in a small proportion of cats.
