Abstract
Bowenoid in situ carcinoma (BISC; papillomavirus-associated squamous cell carcinoma in situ) is an uncommon skin neoplasm of cats that can result in euthanasia because of the development of multiple lesions or because of progression to invasive squamous cell carcinoma. BISCs are currently thought to be caused by Felis catus papillomavirus 2 (FcaPV-2). The presently described cat developed a single 0.5 cm in diameter interscapular mass. Over the following 18 months, the mass doubled in size; no additional lesions developed. The mass was surgically excised and histologically diagnosed as a BISC. However, in contrast to previously reported BISCs, neither prominent thickening of the deep aspects of the follicular infundibula nor marked cell dysplasia were present. Furthermore, ~50% of the keratinocytes in the affected epidermis had prominent PV cytopathic changes that included shrunken angular nuclei and elongated basophilic cytoplasmic inclusions. As the histopathology was not typical for FcaPV-2 infection, polymerase chain reaction was performed and revealed only DNA sequences from Felis catus papillomavirus 3 (FcaPV-3). No further BISCs developed in this cat 6 months postremoval, hence surgical excision appeared to be curative. Results from this case suggest that, although FcaPV-2 appears to be the predominant cause of BISCs in cats, infection by FcaPV-3 can also cause these neoplasms. BISCs caused by FcaPV-3 appear to have unique histologic features that allow the causative PV type to be predicted. Results from this single case suggest that BISCs caused by FcaPV-3 may have a more benign clinical course than those caused by FcaPV-2.
Papillomaviruses (PVs) are highly species-specific epitheliotropic DNA viruses. As part of their normal replication, PVs produce oncoproteins that disrupt normal cell regulation. 1 The vast majority of PV infections either do not result in the development of a visible lesion or cause a self-resolving hyperplastic wart. 7 However, the oncogenes produced by a small number of PV types can predispose an infected cell to neoplastic transformation. 1
Disease resulting from PV infection is considered uncommon in cats. 8 To date, the disease that is most strongly associated with PV infection in cats is bowenoid in situ carcinoma (BISC; papillomavirus-associated squamous cell carcinoma in situ).9,13 This neoplasm typically occurs as single or multiple scaly or crusting lesions within haired skin, especially around the head and neck. 4 Cats with BISCs may require euthanasia either because of the development of multiple BISCs or because of progression of a BISC to a squamous cell carcinoma (SCC). Devon Rex, and the closely related Sphinx, cats may be predisposed to BISCs, and BISCs in cats of these breeds are often rapidly progressive. 15
A 17-year-old female Domestic Shorthair cat was presented to the primary care veterinarian because of a slightly raised 0.5 cm in diameter interscapular skin mass of uncertain duration. No tests were performed at this time, and a post-injection inflammatory reaction was suspected because the cat had received multiple subcutaneous injections at this location for the management of previously diagnosed diabetes mellitus and chronic renal failure. The mass was reported to have enlarged slowly, and 18 months later had doubled in size to 1 cm in diameter. The cat had no other skin lesions. The mass was surgically removed with 1-cm margins and submitted for histologic examination.
On histologic examination, normal epidermis merged abruptly with a well-demarcated focus of marked, irregular epidermal hyperplasia. There were multiple, broad, downward-projecting rete pegs confined by the basement membrane and accompanying mild parakeratosis, hyperkeratosis, and variable melanization. The epidermal hyperplasia also involved the superficial follicular infundibular epithelium (Fig. 1). Dysplasia was present and included: crowding of basal keratinocytes; loss of nuclear polarity; elongation of basal keratinocyte nuclei; and disorderly keratinization. Keratinocytes in the affected epidermis exhibited mild anisokaryosis and anisocytosis and variably large, prominent nucleoli. Mitoses were present at all levels in the affected epidermis. Numerous keratinocytes in the middle to superficial aspects of the affected epidermis were expanded by a large perinuclear vacuole that peripherally displaced a shrunken angular nucleus with retained nuclear architecture. The nucleus of these cells was often surrounded by a prominent, elongated, darkly basophilic cytoplasmic body (Fig. 2) that was interpreted to be condensed or flattened keratohyalin granules. A minority of the enlarged keratinocytes had a small quantity of intracytoplasmic, blue-gray fibrillar material. Dermal melanophagia was mild.
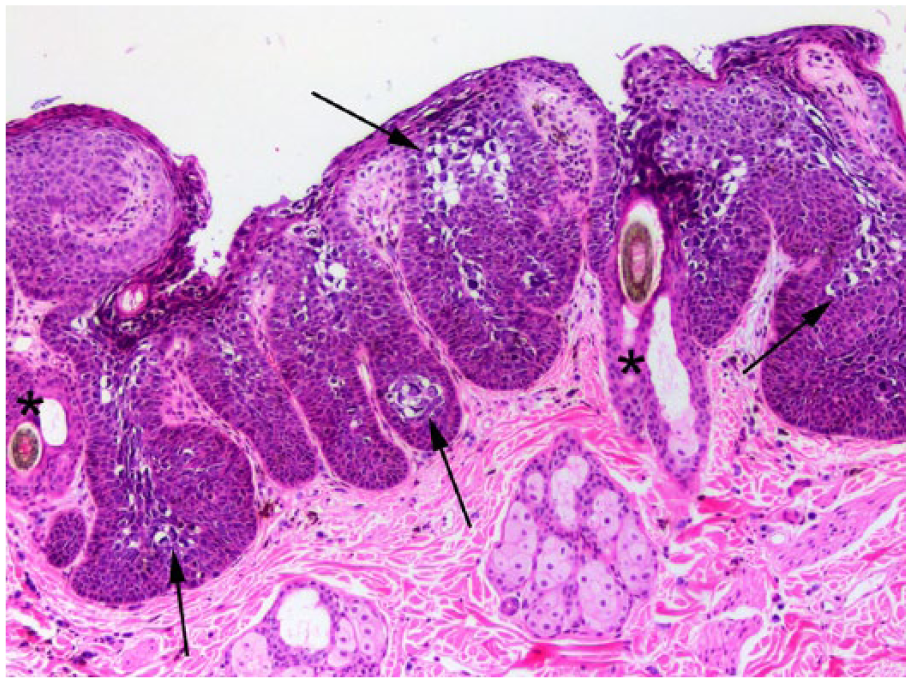
Bowenoid in situ carcinoma associated with Felis catus papillomavirus 3. Note marked epidermal hyperplasia with the thickened epidermis forming broad buds within the underlying dermis. Changes are not visible deep within follicular infundibula (*). Large numbers of cells with papillomavirus-induced cytopathic changes are visible (arrows). Hematoxylin and eosin.
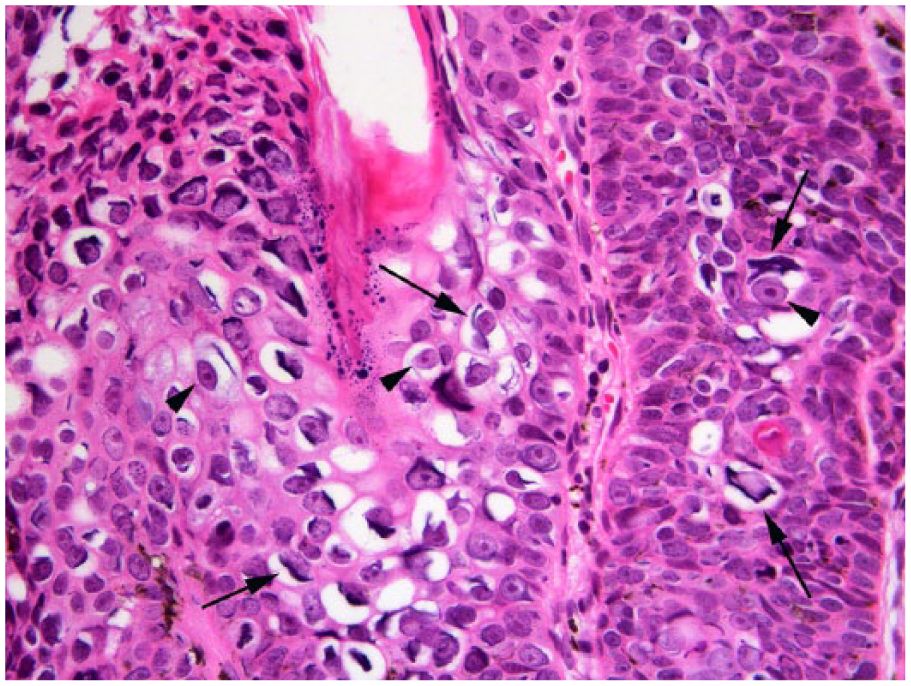
Bowenoid in situ carcinoma associated with Felis catus papillomavirus 3. Viral cytopathic changes include enlarged cells with clear cytoplasm that contain prominent elongated basophilic bodies (arrows) and angular shrunken nuclei that have retained visible nuclear architecture (arrowheads). Hematoxylin and eosin.
To investigate a possible PV etiology, polymerase chain reaction was used to amplify PV DNA sequences from DNA that was extracted from the sample as previously described. 14 Four sets of primers were used to ensure amplification of all 4 currently recognized Felis catus papillomavirus (FcaPV) types. These primers included FAP59/64 that has been previously used to amplify FcaPV-2 DNA, 14 MY09/11 that amplifies FcaPV-3 and -4 DNA, 10 and CP4/5 that amplifies FcaPV-1 DNA sequences. 11 To maximize the sensitivity of detection of FcaPV-2, primers specific for this PV type were also used. 13 Positive controls included DNA extracted from a feline BISC that contained FcaPV-2 DNA for the FAP59/64 and specific primers, DNA extracted from a feline sarcoid for the MY09/11 primers, and DNA extracted from a feline oral papilloma for the CP4/5 primers. No template DNA was added to the negative controls. In the present case, PV DNA was only amplified by the MY09/11 and CP4/5 primers. Sequencing of both amplicons revealed that the amplified DNA sequences were identical to those of FcaPV-3. No DNA was amplified by either the FAP59/64 primers or the primers specific for FcaPV-2.
Immunostaining for p16 has been reported to be consistently increased in feline BISCs, presumably because of degradation of the retinoblastoma (pRB) protein by FcaPV-2 oncoproteins. 13 In the present case, p16 immunostaining was performed as described previously, 12 but revealed only mild intensity of immunostaining within scattered keratinocytes both in the thickened epidermis and in the surrounding histologically normal epidermis.
The final diagnosis was a histologically unusual BISC associated with FcaPV-3 infection. Six months after excision, lesion recurrence or additional lesions have not been observed.
The skin lesion in our report was consistent with a SCC in situ. SCCs in situ are subdivided into BISCs that are associated with PV infection, and actinic keratosis caused by chronic sun exposure. In the present case, a diagnosis of BISC was favored over actinic keratosis given the well-demarcated focus of full-thickness epidermal hyperplasia, presence of melanin pigment, and characteristic loss of nuclear polarity.2,4,19 Three unique histologic features were observed in the BISC in this cat. First, the follicular infundibula were only mildly thickened, and this thickening was confined to the superficial aspects of the follicular infundibular epithelium. The second novel feature was the mild cell dysplasia that was present despite marked epidermal thickening. In the experience of 2 of the authors (JS Munday and R Fairley), a BISC with the amount of epidermal thickening seen in the present case would be expected to contain significant cell dysplasia. The third unique histologic feature was the presence of numerous keratinocytes containing PV-induced cytopathic changes. Although recently developed BISCs have been reported to contain cytopathic changes in a small proportion of keratinocytes, 19 PV-induced cytopathic changes were visible in ~50% of the keratinocytes in the present case.
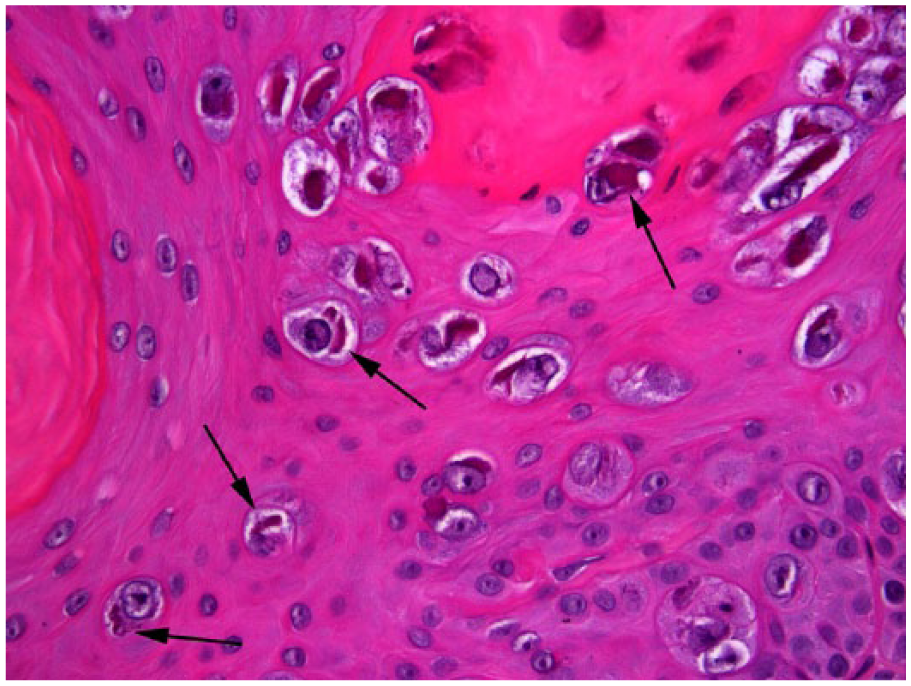
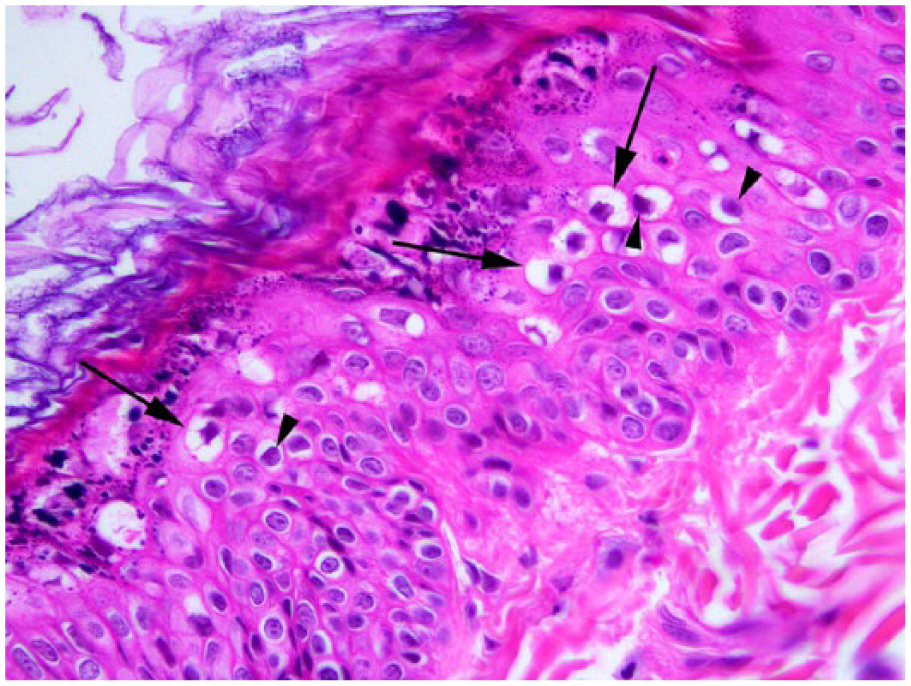
The appearance and extent of the PV-induced cytopathic changes were unusual. In cats, the 3 PV types that have been associated with visible lesions are of different PV genera, and infection by each PV type is expected to result in a different histologic appearance. FcaPV-1 is a genus Lambdapapillomavirus and results in characteristic brightly eosinophilic cytoplasmic bodies (Fig. 3).16,17 FcaPV-2 is a genus Dyothetapapillomavirus and results in cells with shrunken nuclei surrounding a rim of clear cytoplasm without any cytoplasmic bodies (Fig. 4).2,6,19 FcaPV-3 is most closely related to genus Taupapillomavirus (TauPV) 10 and evidence from the present case suggests that this PV results in the presence of an angular shrunken nucleus that retains nuclear architecture. Elongated basophilic bodies were often within the cytoplasm adjacent to the nuclei. FcaPV-3 is closely related to canine papillomavirus 2, a common cause of canine cutaneous papillomas (warts). 5 As cutaneous warts in dogs almost always resolve spontaneously, it is possible that TauPVs are only weakly carcinogenic. This is consistent with the mild dysplasia and absence of progression seen in the presently described FcaPV-3–associated BISC.
Feline oral papilloma associated with Felis catus papillomavirus 1. Viral cytopathic changes include enlarged keratinocytes that contain mildly enlarged nuclei with prominent eosinophilic cytoplasmic bodies (arrows). Hematoxylin and eosin.
Bowenoid in situ carcinoma associated with Felis catus papillomavirus 2. Viral cytopathic changes are present within all layers of the thickened epidermis and include mildly enlarged cells that contain a shrunken nucleus surrounded by a rim of clear cytoplasm. Cells in the superficial epidermis are more greatly enlarged (arrows). However, in contrast to the cytopathic changes in Figure 2, there is reduced nuclear detail in affected cells (arrowheads), and no cytoplasmic bodies are visible. Hematoxylin and eosin.
Previous studies have amplified FcaPV-3 from a small proportion of feline BISCs and SCCs.10,12 However, as this detection was often as a coinfection with FcaPV-2, FcaPV-3 was considered most likely to be an “innocent bystander” in the lesions. One BISC that contained only FcaPV-3 DNA has been reported 10 ; however, as FcaPV-2 was subsequently detected in other samples from the cat in the previous report, the role of FcaPV-3 in lesion development was uncertain. In our case, the failure to detect other FcaPV types, the unusual histologic appearance of the BISC, and the unique viral cytopathic effects in the keratinocytes in the affected epidermis are all consistent with FcaPV-3 being the cause of the BISC. Because PVs are often present asymptomatically on the skin surface, it cannot be excluded that the lesions contained an undetected PV type and FcaPV-3 was present simply as an incidental infection.
In our case, the BISC enlarged from 0.5 cm to 1 cm in diameter over an 18-month period. Although the clinical behavior of BISCs is variable, the slow lesion progression over a long period of time is unusual.3,4 The cat in our report developed a single BISC and did not develop any additional lesions. In contrast, multiple lesions were consistently present in a previous study of 19 cats with BISCs. 19 In a different study in which cats with BISCs were observed for 12 months, 9 of 9 cats developed additional BISCs with a median time to additional lesion development of just 50 days. 3 In the present cat, the development of just 1 slowly progressive lesion over a 2-year period suggests that the cat had an unusually mild disease. As this is a single case, it is hard to draw firm conclusions, but our evidence suggests that BISCs caused by FcaPV-3 may have a more benign clinical behavior than those caused by FcaPV-2.
In cats, p16 immunostaining has been used in BISCs and SCCs to indicate loss of normal pRB function resulting from PV infection.9,12 The lack of immunostaining visible within the presently described BISC suggests that the oncoproteins produced by FcaPV-3 do not result in dysregulation in the pRB/p16 pathway.
Previous studies have revealed that PV infection of the skin of cats is common, but lesion development is rare. 18 The factors that predispose to the development of a BISC are uncertain. In the present case, the site of BISC development had been used to administer multiple subcutaneous injections. Whether or not the repeated injections predisposed to BISC development is unknown.
Footnotes
Author’s contributions
JS Munday contributed to interpretation of data; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. R Fairley contributed to analysis of data; critically revised the manuscript; and gave final approval. K Atkinson contributed to conception of the study; contributed to analysis of data; critically revised the manuscript; and gave final approval.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
