Abstract
Bovine viral diarrhea virus (BVDV) 1b was isolated from tissues of a term bovine fetus with petechial hemorrhages noted throughout the body and placenta at autopsy. Fresh lung, kidney, thymus, and liver tissues were examined by direct fluorescent antibody testing and were positive for BVDV antigen and negative for bovine herpesvirus 1 antigen. An organ pool of fresh tissues was positive for noncytopathic (NCP) BVDV-1 by virus isolation. BVDV-1b was identified by sequencing of the 5’-UTR region of the genome. Fixed brain, placenta, thymus, lymph node, lung, kidney, skeletal muscle, liver, and bone marrow were positive for BVDV antigen by immunohistochemistry. Although BVDV hemorrhage and/or thrombocytopenia has been associated historically with NCP strains of BVDV-2, this case adds to more recent reports of BVDV-1 infections and hemorrhage in cattle. This BVDV-1b isolate should be investigated for its potential to cause hemorrhage in postnatal cattle.
Bovine viral diarrhea viruses (BVDV; family Flaviviridae, genus Pestivirus), represented by BVDV-1 and -2, can produce a variety of clinical presentations: acute or transient, respiratory, digestive tract, thrombocytopenic/hemorrhagic form, mucosal disease, reproductive and fetal, and persistently infected (PI) cattle. 7 The hemorrhagic and thrombocytopenic form was identified in different regions in the United States3,19; using molecular tests available at the time, BVDV-2 was identified in these cases.4,20 In a 2005 review of BVDV manifestations, the hemorrhagic form was mostly attributed to BVDV-2. 12 However, there have been reports of BVDV-1 and, more specifically, the BVDV-1b subtype isolated from cattle with hemorrhage.1,5,14,16,23 The occurrence of BVDV infections with hemorrhages is possible in fetuses.
We describe a case of BVDV-1b infection of a term bovine fetus with extensive hemorrhages throughout the body and placenta. A thin, 22-kg, full-term, female fetus (Angus per history) and placenta (3.6 kg), with moderate postmortem autolysis, were presented for autopsy at the Oklahoma Animal Disease Diagnostic Laboratory (OADDL; Stillwater, OK). Maternal serum was not available, and no vaccination history was given. At autopsy, the subcutis, major muscle groups, oral cavity, larynx, pharynx, and multiple internal organs contained numerous 1–3 mm irregularly round petechiae (Figs. 1, 2). The placenta contained similar petechiae, and several cotyledons were diffusely reddened. The medulla and cortex of both kidneys was hemorrhagic, and round pale-tan discolored areas of 1–2 mm diameter were scattered irregularly.
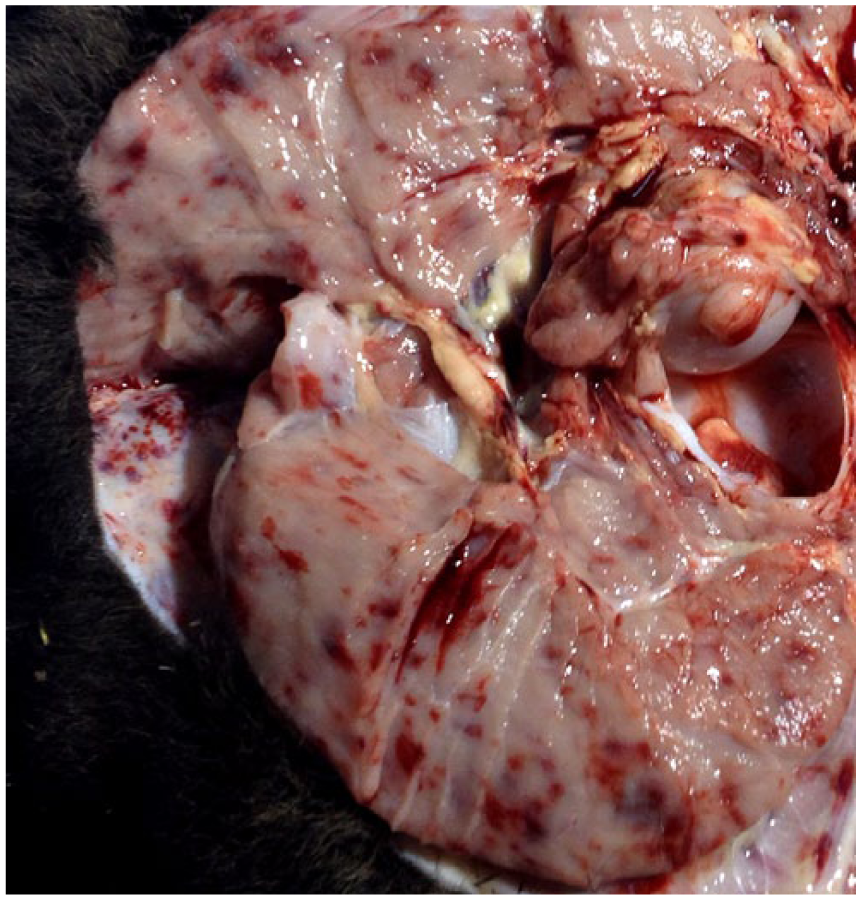
Intramuscular and subcutaneous hemorrhage around the coxofemoral joint of a bovine viral diarrhea virus–infected fetus.
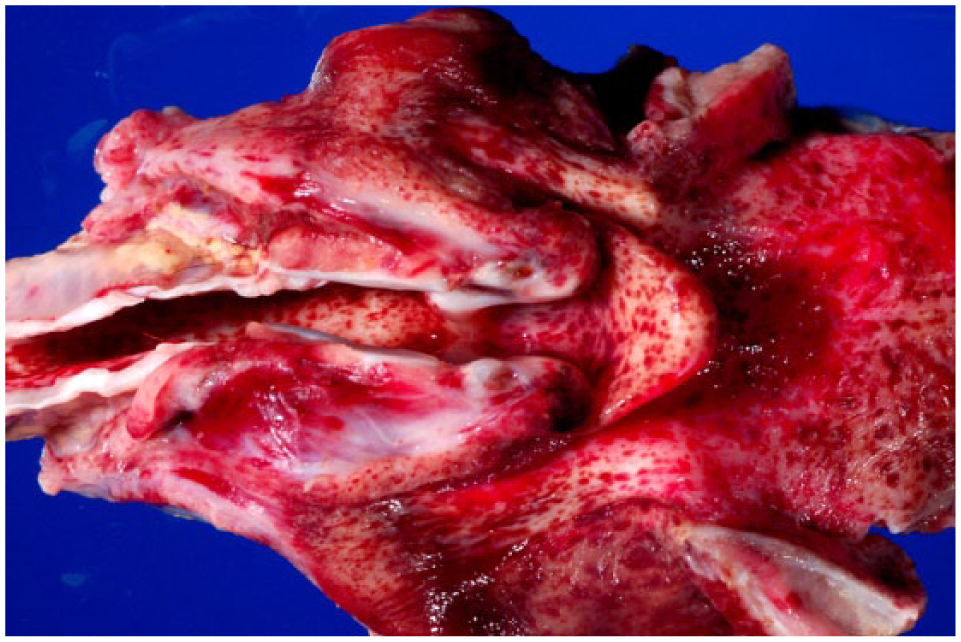
Petechial hemorrhages in larynx, trachea, and base of tongue of a bovine viral diarrhea virus–infected fetus.
Upon histologic examination, the liver exhibited marked multifocal-to-coalescing hepatitis, with mixed inflammatory cell infiltrates, frequently of perivascular distribution. The kidney had similar marked multifocal-to-coalescing nephritis with regionally extensive medullary hemorrhage. Pulmonary vessel lumens contained platelet clumps. The connective tissue around multiple large vessels and airways was hemorrhagic. Placental chorionic villi were occasionally covered by a thin layer of red blood cells mixed with fibrin. Thymus, brain, tongue, spleen, and esophagus were focally hemorrhagic.
Samples were submitted for bacterial culture, fetal serology, and direct fluorescent antibody testing (DFAT). Lung and stomach contents were negative for Brucella spp. Placenta and tissue pool were negative for Campylobacter fetus. The placenta was positive for Pseudomonas spp. (likely a contaminant). Salmonella culture of the tissue pool of lung and stomach contents was negative. Fetal serum was negative for antibodies to Brucella abortus (card test), BVDV-1, bovine herpesvirus-1 (BoHV-1), Leptospira interrogans serovar Canicola, Leptospira. kirschner serovar Grippotyphosa, Leptospira interrogans serovar Hardjo, Leptospira interrogans serovar Icterohaemorrhagiae, and Leptospira interrogans serovar Pomona (all leptospiral microscopic agglutination test titers <100), and Neospora (N. caninum antibody test kit, cELISA, VMRD, Pullman, WA). Fresh samples of lung, kidney, thymus, and liver were submitted for DFAT. The fresh tissues were positive for BVDV antigen and negative for BoHV-1.
Subsequent to the BVDV-positive, DFAT-positive test, an organ pool of lung, kidney, thymus, and liver was submitted to the research laboratory of the Department of Veterinary Pathobiology of Oklahoma State University. Tissue homogenates were inoculated onto confluent Madin–Darby bovine kidney monolayer cultures and incubated for 6 d9,10; no cytopathic effects were observed. The cultures were frozen and subsequently tested by a gel-based PCR. 7 Uninfected negative controls were included. The MDBK infected cultures were positive for BVDV-1. The positive samples were submitted to the U.S. Department of Agriculture, National Animal Disease Center (Ames, IA), and the virus was subtyped using a region of the 5’ untranslated region (UTR).10,11,13,21,22 The virus was subtyped as BVDV-1b (Fig. 3).
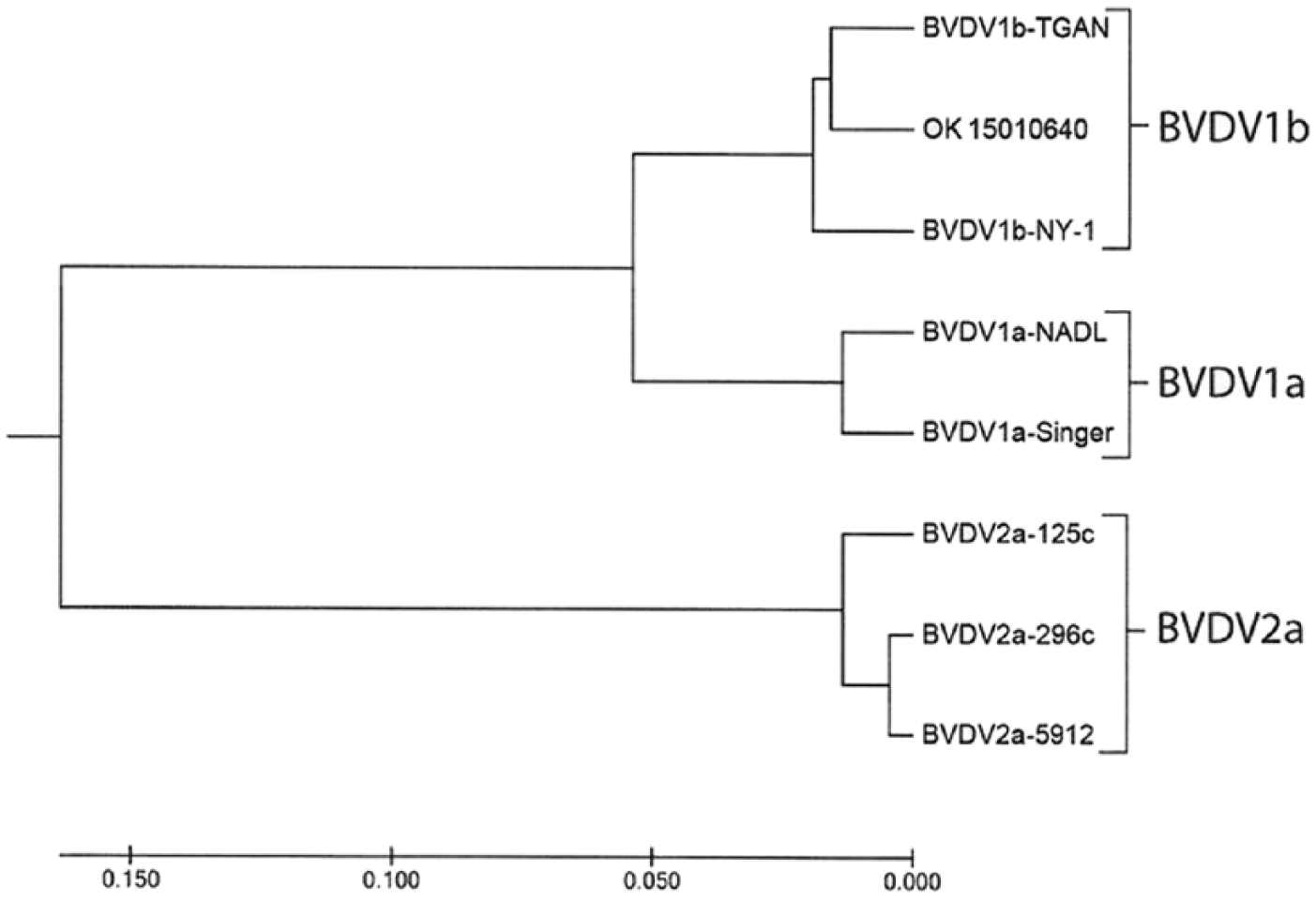
Dendrogram of bovine viral diarrhea virus (BVDV)-1b from fetal hemorrhage (OK 15010640). The evolutionary history was inferred using the UPGMA method. 21 The optimal tree with the sum of branch length (= 0.44689717) is shown. The tree is drawn to scale, with branch lengths in the same units as those of the evolutionary distances used to infer the phylogenetic tree. The evolutionary distances were computed using the maximum composite likelihood method 22 and are in the units of the number of base substitutions per site. The analysis involved 8 nucleotide sequences derived from the 5’-UTR. All positions containing gaps and missing data were eliminated. There were 228 positions in the final dataset. Evolutionary analyses were conducted in MEGA7. 13
Fixed tissues were submitted for BVDV immunohistochemistry (IHC). Sections of tongue, liver, kidney, lung, thymus, lymph node, skeletal muscle, bone marrow, brain, and placenta were examined by BVDV IHC as described previously. 6 Primary antibody was an IgG1 monoclonal antibody in ascites, designated 3.12F1 (OSU Saliki, Oklahoma State University, Stillwater, OK), that was diluted 1:75,000 in phosphate-buffered saline solution containing polysorbate 20. Secondary and tertiary antibodies were rabbit anti-mouse IgG1 antibody (H+L; Zymax [ref 816740, lot 1373247A], Invitrogen, Frederick, MD) diluted 1:500 and biotinylated goat anti-rabbit IgG (H+L; [ref BA-5000, lot W0327], Vector Laboratories, Burlingame, CA). Streptavidin–horseradish peroxidase and Nova red substrate ([catalog SK-4805], Vector Laboratories) were used to develop the color reaction. For positive control, a section of ear from a PI calf was stained as described above. For negative controls, duplicate sections of tissues from the fetus and a PI calf were treated with normal mouse IgG prior to treatment with secondary and tertiary antibodies, and treated for color development. Intensity of the BVDV signal was graded from 0 (no signal) to 4+ (intense uniform staining of cell cytoplasm).
The strongest BVDV-positive IHC signals were in neurons (Fig. 4) in multiple brain sections, cerebral endothelium, placental epithelium, and within large cells—presumed to be macrophages or dendritic cells—throughout the thymus (Fig. 5). In addition to 4+ antigen intensity of antigen by IHC in brain and thymus, 4+ staining was observed in the placenta (Fig. 6). In other organs, BVDV-positive macrophages were randomly scattered throughout interstitium or perivascular connective tissue. The bone marrow had randomly scattered positive mononuclear cells, which appeared to be both myeloid and erythroid precursors; however, megakaryocytes were uniformly negative for BVDV antigen (Table 1).
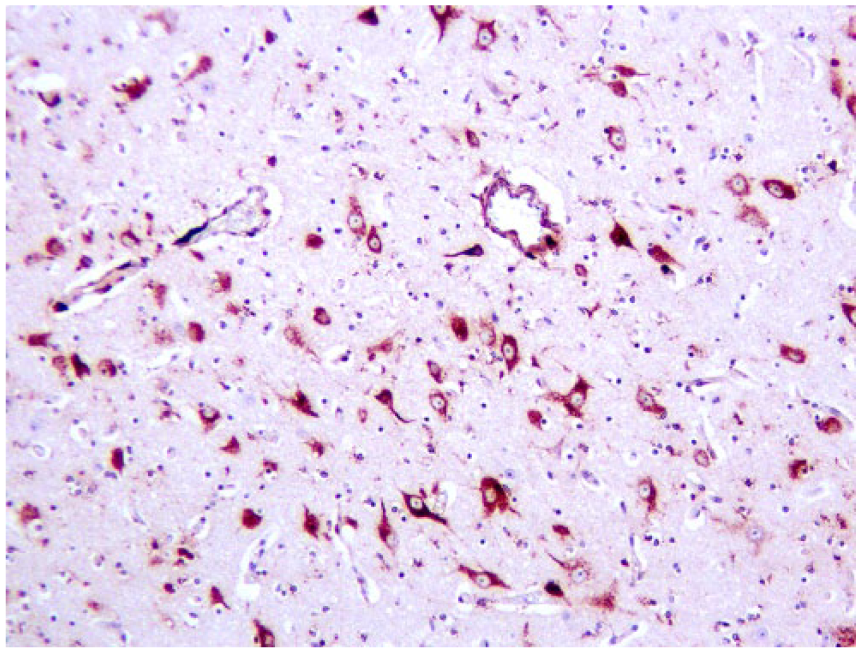
Neurons, endothelium, and perivascular cells in cerebral cortex stain positively for bovine viral diarrhea virus by immunohistochemistry; glia are negative.
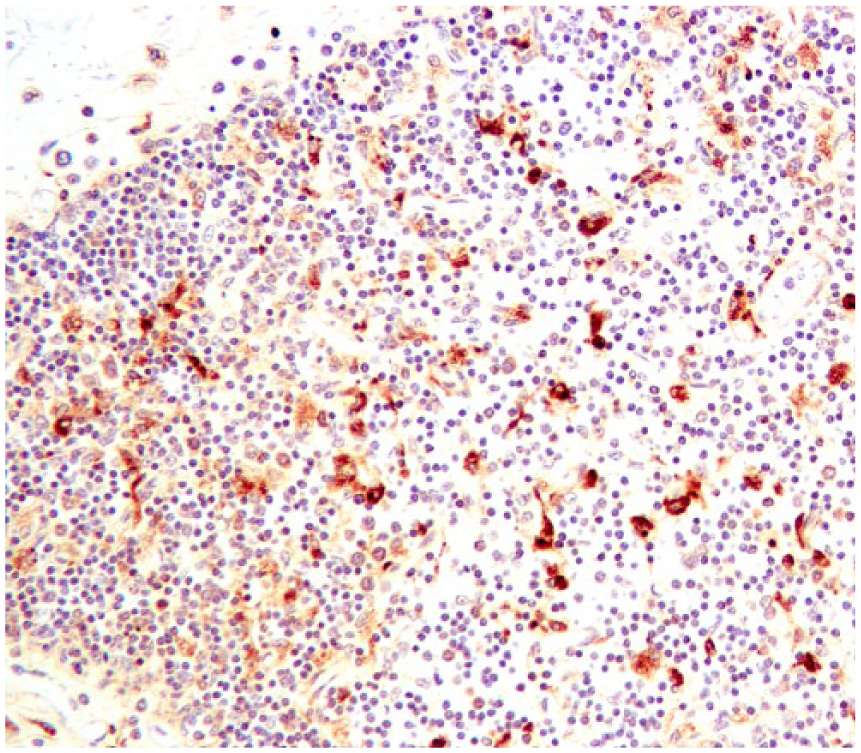
Large cells, most likely macrophages, stain positively for bovine viral diarrhea virus (BVDV) in fetal thymus; lymphocytes are mostly negative. BVDV immunohistochemistry.
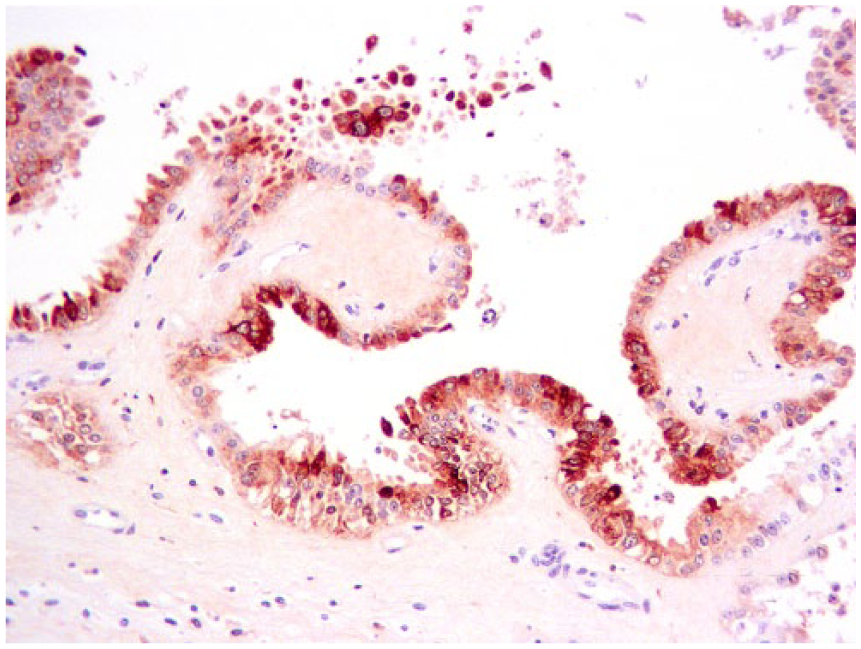
Placental trophoblastic epithelium stained immunohistochemistry positive for bovine viral diarrhea virus.
Bovine viral diarrhea virus (BVDV) antigen intensity and distribution in fetal tissues, detected by immunohistochemistry (IHC).
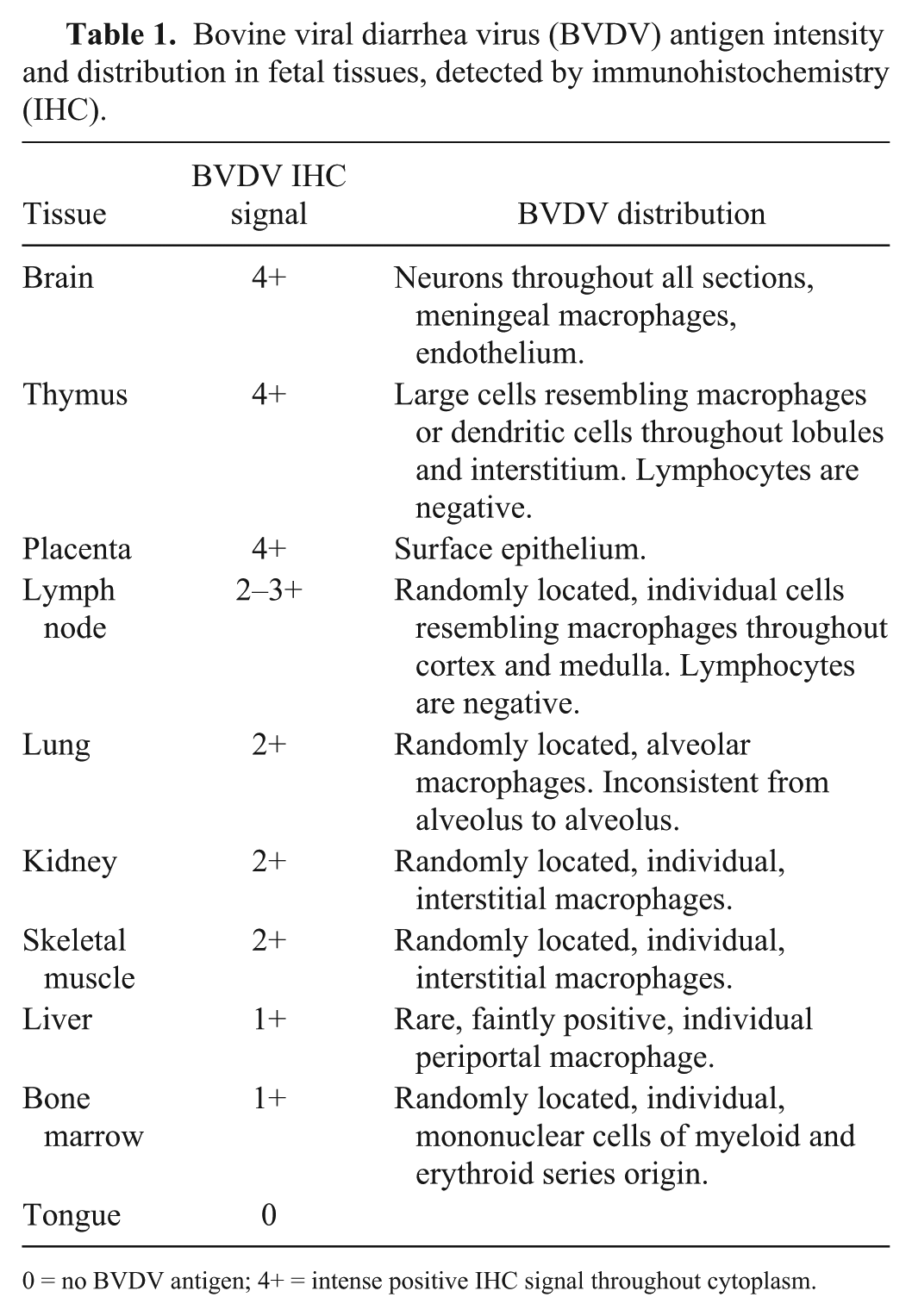
0 = no BVDV antigen; 4+ = intense positive IHC signal throughout cytoplasm.
The finding of BVDV-1b in a case of severe and extensive hemorrhage in a term fetus indicates that this strain has particular virulence. Initial DFAT testing for BVDV on the fresh tissues indicated that BVDV should be pursued further in this case. Using cell culture to propagate the virus and its confirmation sequencing led to the characterization of noncytopathic (NCP) BVDV-1b. The submitting veterinarian was notified to alert the owner to possible fetal BVDV infections in the herd. Testing for other affected fetuses as well as for PI calves was warranted and encouraged. Also encouraged was the implementation of an effective biosecurity program and a BVDV vaccination program to prevent fetal infections.
The finding of numerous tissues positive by BVDV IHC underscores the increasing role of IHC in the diagnosis of infections. 8 The 4+ lesion scoring for BVDV IHC in the brain, thymus, and placenta demonstrates the value of selecting these tissues for BVDV confirmation. BVDV antigen has been identified in the brains of PI calves 17 and a heifer with meningoencephalitis. 2 A prior study found extensive regions of the brain positive for BVDV antigen via IHC in infected fetuses. 18 The placenta and thymus were also positive for BVDV by IHC in a prior study detecting infected fetuses in a vaccine challenge study. 15 The detection of NCP BVDV-1b in a hemorrhagic case is further evidence that the hemorrhagic form is not limited to BVDV-2 viruses.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The research was supported by McCasland Foundation Endowed Chair for Food Animal Research (RW Fulton).
